9.1A: Free Radicals and Oxidative Stress
- Page ID
- 1509
Before you can understand what an antioxidant is, it is important to have an understanding of oxidants. As you have learned already, oxidation is the loss of an electron as shown below.
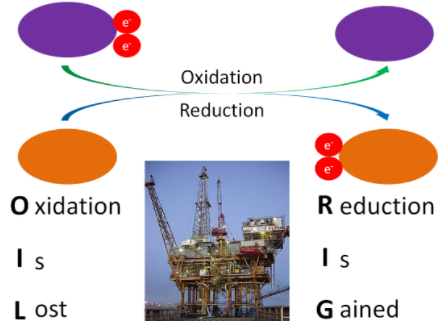
Figure 9.111 The purple compound is oxidized; the orange compound is reduced1
Some important terms to understand:
Free Radical - a molecule with an unpaired electron in its outer orbital.
The following example shows normal oxygen losing an electron from its outer orbital and thus, becoming an oxygen free radical.
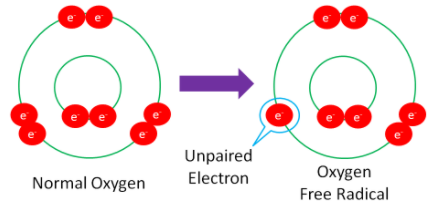
Figure 9.112 Normal oxygen is converted to an oxygen free radical by losing one electron in its outer orbital, leaving one unpaired electron
Free radicals are highly reactive because they actively seek an electron to stabilize the molecule.
Reactive Oxygen Species (ROS) - an oxygen-containing, free radical species.
Some of the most common ROS are (● symbolizes radical):
Superoxide (O2●)
Hydroxyl Radical (●OH)
Hydrogen Peroxide Radical (HO2●)
Peroxyl Radical (ROO2●)
Alkoxyl Radical (RO●)
Ozone (O3)
Singlet Oxygen (1O2)
Hydrogen Peroxide (H2O2)
Oxidative Stress - the imbalance between the production of ROS/free radicals and the body’s ability to quench them.
The following video does a good job illustrating how free radicals can be formed and quenched by antioxidants.
|
Web Link |
The following figure shows that inflammation caused by hitting your thumb with a hammer, exposure to UV light, radiation, smoking, and air pollution are all sources of free radicals.
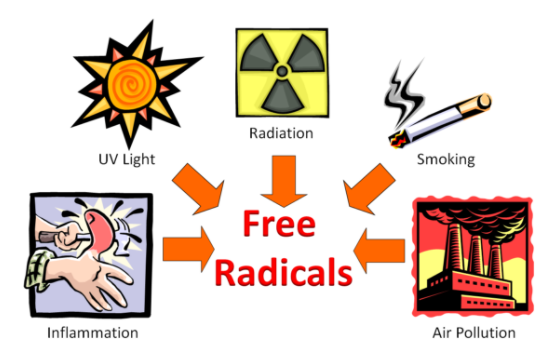
Figure 9.113 Some sources of free radicals
Free radicals can be generated by a variety of sources that can be classified as endogenous (within the body) and exogenous sources (outside the body). The link below is a figure that shows how ROS can be generated from each of these sources.
|
Web Link Sources and Cellular Responses to Reactive Oxygen Species |
So, we have these free radicals searching for an electron, what's the big deal? The problem arises if the free radicals oxidize LDL, proteins, or DNA as shown below.
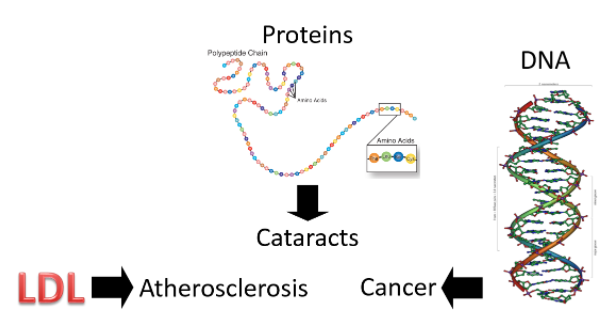
Figure 9.114 Free radicals can attack LDL, proteins, and DNA2,3
Oxidized LDL is more atherogenic, meaning it is more likely to contribute to atherosclerosis (hardening of the arteries) than normal LDL. Protein oxidation is believed to be involved in the development of cataracts. Cataracts are the clouding of the lens of the eye. If you would like to see what it looks like, see the link below.
|
Web Link |
If a nucleotide in DNA is attacked, it can result in a mutation. A mutation is a change in the nucleotide or base pair sequence of DNA. Mutations are a common occurrence in cancer.
References & Links
- en.Wikipedia.org/wiki/Image:G...e_Platform.jpg
- www.genome.gov/Pages/Hyperion...=amino%20acids
- http://www.genome.gov/Glossary/index.cfm?id=149
-
Videos
How Antioxidants Work - https://www.youtube.com/watch?v=lG3OOXIXvxw
Links
Sources and Cellular Responses to Reactive Oxygen Species - www.nature.com/nature/journal...8239a0_F1.html
Cataract Vision Simulator - https://www.aao.org/eye-health/disea...sion-simulator


