1.1: Management Principles/Guidelines for Head and Neck Cancer in Developing Countries
- Page ID
- 15426
OPEN ACCESS ATLAS OF OTOLARYNGOLOGY, HEAD & NECK OPERATIVE SURGERY
MANAGEMENT PRINCIPLES/GUIDELINES FOR HEAD AND NECK CANCER IN DEVELOPING COUNTRIES
Johan Fagan, Clare Stannard, Sameera Dalvie
Developing countries constitute the majority of the world’s landmass (Figure 1) and are home to more than 50% of its people.
Figure 1: Developing (blue) vs. developed world (tan)
Cancer poses a major public health crisis in the developing world. Developing countries accounted for >50% of newly diagnosed cancers in 2010; it is projected that this figure will increase to 70% by 2030 (Figure 2)1. This increasing percentage is attributed to population growth, reduced mortality from infectious diseases and an aging society1.
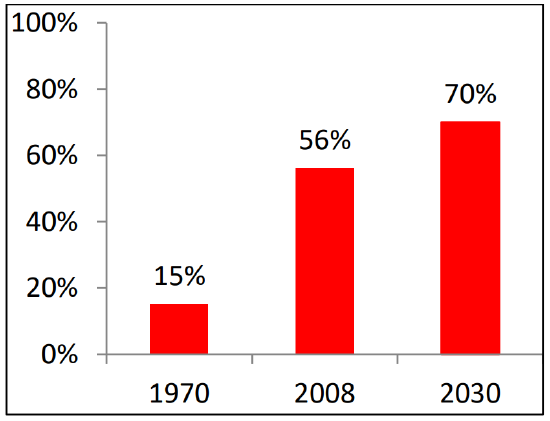
Figure 2: Increasing percentage of global burden of cancer in the developing countries1
There is a wide disparity in a cancer-related fatality which is aligned with income levels, ranging from 75% in low-income countries to 46% in high-income countries (Figure 3)1. Even though developing countries account for 67% of cancer-related deaths, they account for only 5% of cancer-related spending1.

Figure 3: Case fatality from cancer according to income levels of countries1
Therefore, it is apparent that it is essential that innovation, expertise, resources, teaching and research be directed to addressing cancer in the developing world if we are to improve cancer outcomes globally. Given the financial and infrastructural challenges, the cancer management also must be adapted to local constraints. (https://developingworldheadandneckcance rguidelines.com/)
HEAD & NECK CANCER IN DEVELOPING COUNTRIES
Rapid economic growth in developing countries has been accompanied by lifestyle changes associated with squamous cell cancer such as smoking, alcohol, and longevity. Two-thirds of oral and pharyngeal cancers (excluding nasopharynx) occur in developing countries2. Figure 4 illustrates the significant geographical variation for oral cancer; it is the most common cancer in males in high-risk areas such as Sri Lanka, India, Pakistan and Bangladesh where it accounts for up to 25% of all new cancers2. The principal causes of oral cancer are tobacco (smoked or chewed) and betel quid2. Cancer of the nasopharynx is also principally a developing world problem where it is related to Epstein Barr virus infection (Figure 5)3,4,5.

Figure 4: Countries (brown) with high incidence and mortality from oral cancer2.

Figure 5: Nasopharyngeal carcinoma: Estimated age-standardized incidence rate/100,000; GLOBOCAN 2008 (IARC)3.
HIV is also associated with malignancies of the head and neck. The prevalence of HIV is highest in developing countries; sub-Saharan Africa accounts for two-thirds of HIV positive people6. HIV is associated with Kaposi’s sarcoma and non-Hodgkin’s (and Hodgkin’s) lymphoma; with Kaposi’s sarcoma and lymphoma, surgery is restricted to obtaining a tissue biopsy. Squamous cell carcinoma of the conjunctiva is also associated with HIV7. Patients with advanced squamous cell carcinoma of the conjunctiva and metastases to parotid and cervical lymph nodes may require orbital exenteration, parotidectomy, neck dissection, and orbital reconstructive procedures. The association between HIV and mucosal squamous cell carcinoma of the upper aerodigestive tract is less clear-cut8.
FACTORS TO CONSIDER WHEN TREATING HEAD & NECK CANCER IN DEVELOPING COUNTRIES
One should guard against simply extrapolating management protocols from developed world centers to head and neck patients in developing countries.
1. Advanced cancers
Patients in developing countries are more likely to present with advanced cancer9,10; consequently, treatment is mostly palliative11. Even in a middle-income country like South Africa, 52% of patients undergoing total laryngectomy initially required an emergency tracheostomy12. Late presentation may be due to ignorance, poverty, poor access to health services, and patients consulting traditional healers and using traditional medicines.
The adverse consequences of delayed presentation are compounded by long waiting lists for surgery and irradiation. Frequently patients become inoperable while awaiting surgery or radiation; this complicates initial patient selection and treatment planning. Jensen et al (2007) reported that one month’s delay was associated with a 62% increase in tumour size, 20% new nodal metastases, and that cancers were upstaged (TNM) in 16% of patients studied; mean tumor volume doubling time was 3 months13. Some institutions administer “holding chemotherapy” (methotrexate or platinum-based drugs) in an attempt to slow tumor progression while patients await definitive treatment; yet there is no evidence that this improves outcomes.
2. Does HIV status matter?
Do HIV+ patients need to be managed differently? One may need to consider the following questions when managing HIV+ patients, especially when resources are limited:
Is radiotherapy accompanied by increased mucosal and cutaneous toxicity in HIV+ patients? Although there are many reports of radiotherapy-induced skin and mucosal toxicity with Kaposi sarcoma, the few reports of toxicity with other head and neck malignancies indicate good tolerance to radiation +/- chemotherapy14,15,16.
Should antiretroviral therapy be commenced to boost CD4 counts in immunocompromised patients prior to (chemo)radiation? Radiation can suppress CD4 counts. Yet even though it may seem reasonable to institute antiretroviral therapy to boost depressed CD4 counts prior to radiation, there are no controlled studies that address this question. Although interactions between antiretroviral therapy and radiation have not been well documented in the literature, there is a theoretical concern about the combination of the myelosuppressive effects of certain antiretroviral agents and myelosuppressive chemotherapeutic agents used with head and neck cancers such as platinum alkylators e.g. cisplatin and carboplatin16.
What is the anticipated life expectancy of an HIV+ patient? Adults that commence antiretroviral therapy before CD4 counts drop to < 200 cells/mm3 have about 80% of normal life expectancy; even severely ill HIV patients treated with antiretrovirals have at least an 80% chance of surviving 2 years17.
Do CD4 count and HIV status affect surgical outcomes? Even major surgery does not depress CD4 counts18, and HIV status per se does not increase the likelihood of early surgical complications19. A depressed CD4 count (<100 cells/mm3) has however been reported to be a predictor of postoperative sepsis20,21. Instituting antiretroviral therapy prior to surgery has the benefits of reducing the viral load of the patient (viral exposure to surgical team) and increases patients’ CD4 counts.
It may be concluded that there is insufficient evidence to modify treatment of “apparently healthy” HIV +ve patients with head and neck cancer with CD4 counts of >350 cells/mm3 16. There is also little reason to routinely determine the HIV status of healthy-looking head and neck cancer patients from an oncologic perspective alone; only when HIV infection causes ill health and immunosuppresssion may HIV status preclude patients from undergoing major surgery or chemoradiation.
3. How does one prioritize head and neck cancer patients?
Deciding who or who not to treat when the burden of cancer exceeds available treatment resources is perhaps the most difficult task that oncologists and surgeons in developing countries must deal with. Arriving at a decision involves ethical and practical considerations such as tumor stage, prognosis, palliation vs. cure, comorbidities, nutritional status, age, socioeconomic status, social support structure, distance from the closest treatment center, likelihood of regular follow-up, parental status, employment status, and the difficult ethical issue of possibly denying access to publicly funded treatment to patients originating from a foreign country without adequate treatment facilities.
However, when access to surgery and radiotherapy are the principal treatment constraints, it is reasonable to prioritize patients with the most curable (early stage) malignancies, especially when adjuvant radiation is not available or will be significantly delayed following resection of advanced malignancies.
4. Radiotherapy
Though central to treatment of head and neck cancer, radiotherapy is unavailable in much of the developing world. AbdelWahab et al (2013) reported that only 23/52 African countries had radiotherapy facilities, and that facilities were concentrated in the southern and northern parts of the continent (Figure 6); that brachytherapy resources were available in only 20 countries; and that, because only 2% of African countries had modern imaging equipment and treatment planning systems, simple, curative treatment is generally based on two-dimensional imaging and treatment planning11.

Figure 6: Radiation therapy services in Africa11
Tatsuzaki & Levin (2001) similarly reported significant unavailability of radiation facilities in Asia and the Pacific regions22; and Zubizarreta et al (2004) reported a major restriction to access to radiotherapy in 16/18 South American countries due to insufficient numbers of specialists23.
Consequently, most radiation services in the developing world are fairly basic and deliver mainly palliative care9. Radiation therapists also need to be cautious about extrapolating favorable treatment results emanating from modern radiation therapy centers of excellence to situations where reliance is placed on dated technology.
Patients that undergo radiation to the head and neck require long term follow-up to detect and manage delayed radiotherapy-related complications e.g. hypothyroidism increases over time and is present in 25% of patients at 5 years24. Therefore reliability for follow-up and the ability to monitor thyroid function and treat hypothyroidism must be considered when selecting patients for radiation.
5. Chemoradiation
In developed countries, chemoradiation (mostly chemotherapy used concurrently with radiotherapy / CCRT) is widely used as an organ-sparing treatment strategy with squamous cell carcinoma of the oral cavity, larynx, and oro-, hypo-, and nasopharynx. Compared to radiotherapy alone it has an 8% advantage in terms of locoregional control and survival rates25.
However, to achieve such favorable outcomes, the “package of care” must include modern, sophisticated imaging (CT, MRI, PET) both for treatment planning and follow-up, medical and intensive care support for chemotoxicity, PEG feeding, and complex salvage surgery for persistent cancer or for recurrence as well as dental, speech, swallowing and audiological rehabilitation. Salvage surgery requires high levels of surgical expertise including proficiency with free tissue transfer flaps.
Because chemoradiation is expensive, toxic25 and complex treatment and requires a “package of care” not available in many developing world centres, it has to be employed with great circumspection in a developing world setting.
Kumar et al reported a 14% mortality rate during and within 30 days of treatment in patients with advanced head and neck cancer treated with concomitant boost radiotherapy with concurrent weekly cisplatin at a tertiary hospital in India; the authors attributed the high mortality to poor support to deal with acute morbidity, poverty, malnutrition, illiteracy, and poor hygiene and concluded that “on present evidence in the setting of a developing country, CCRT with concurrent cisplatin cannot be recommended as primary therapy in advanced head and neck cancers without formal comparison with other treatment modalities” (Figure 7)26.

Figure 7: Locoregional Control (LRC) and Overall Survival (OS)26
Therefore, if chemoradiation is to be considered, patients must be carefully selected to predict favourable outcomes by considering factors such as age, general health, social support, immune (HIV) status, tuberculosis, and the “package of care” mentioned above should be available.
6. Altered fractionation
Altered fractionation schedules also improve locoregional control. Accelerated radiotherapy is perhaps better suited to a developing world setting than chemoradiation as it is cheaper and better tolerated. Overgaard et al reported that a 6-fractions per-week radiation schedule significantly improved locoregional control for squamous cell carcinoma of the larynx, pharynx and oral cavity when compared to conventional schedules of 5-treatments per-week. Despite increased acute morbidity, accelerated radiotherapy did not cause increased late morbidity and had the benefit of reducing overall treatment by 1 week27. Concomitant boost radiotherapy with a 2nd daily fraction to the gross tumor volume in the final 10 days of treatment also reduces overall treatment time, thus reducing the chance of repopulation and improves local control28.
However, unless a radiation therapy department already treats patients 6 days/week, both the above schedules require reorganization to accommodate the 2nd daily fraction.
7. Surgery
Surgery is often the only treatment available because of inadequate radiotherapy and chemoradiation facilities. Yet, surgeons in developing countries frequently lack head and neck surgical training, and modern surgical technology (bipolar cautery, laser, transoral robotic surgery, endoscopic surgery), frozen section, blood products, adequate operating time, good anesthesia and intensive care support are often lacking29. Surgeons in developing countries need to keep abreast of and adapt modern surgical principles and techniques to a lower technology type practice e.g. substitute transoral microsurgery for early laryngeal cancer with laryngofissure and other open partial laryngectomy procedures; ensure wide tumor resection margins in the absence of frozen section control and postoperative radiation therapy; liberally employ elective neck dissection in the absence of sophisticated imaging, and rely on a range of pedicled rather than microvascular free tissue transfer flaps to reconstruct surgical defects.
MANAGEMENT PROTOCOLS
Selecting appropriate treatment for head and neck cancer patients in developing countries is particularly challenging and involves complex, individualized decisions often without the benefit of special investigations such as FNAC, CT, MRI, PETCT and HPV status.
Unlike the situation in well-resourced health systems, it may not always be possible for treatment to be protocol-driven as most patients are dependent on state run services with poor health infrastructure and resources. For the same reasons, protocols designed for developed world settings are not always relevant; e.g. tuberculosis mimics metastases on PET scan, therefore limiting its utility as a staging tool in societies where tuberculosis is endemic (Figure 8).
Reliance therefore frequently is placed on clinical acumen, experience, intuition and institutional bias, often in the absence of scientific evidence to support clinical decisions. Investigations and treatment must be tailored to the individual patient taking into account resource constraints relating to e.g. CT, MRI, operating rooms, ICU, radiation facilities, and blood transfusions; treatment delays (often many months); likelihood of regular follow-up; access to drugs e.g. thyroid and calcium replacement; nutritional status; social support; poverty; comorbidities (often poorly treated or neglected) including HIV; cultural bias; and the availability of surgical expertise, radiation therapy and chemotherapy.

Figure 8: PET CT image of pulmonary TB:
Roy M, Ellis S. Radiological diagnosis and follow-up of pulmonary tuberculosis. Postgrad Med J 2010;86:663-74
Certain principles will now be highlighted that may be considered when designing management protocols in resource constrained settings.
History: Consider cultural and religious values of patients as these may affect management. Inquire about risk factors including betel nut, areca nut, reverse smoking, chewing tobacco and comorbidities e.g. tuberculosis.
Metastatic workup: When access to operating time and adjuvant radiotherapy is limited, one could argue in favor of employing CT (even if it is a limited and expensive resource) to rule out pulmonary metastases that are not evident on CXR before (inappropriately) committing scarce surgical resources to advanced T and N stage cancers that have metastases.
N0 neck: It is reasonable to have a low threshold to electively treat the N0 neck by means of a selective neck dissection in cases of unreliable follow-up, lack of specialized imaging (initially and for follow-up), and delayed adjuvant radiation. Surgeons should have a low threshold to convert a selective to a modified neck dissection when suspicious lymph nodes are encountered, especially in the absence of postoperative radiation capability.
N+ neck: Even though lymphadenopathy in patients from poorer communities may result from untreated dental, oral and pharyngeal infections, HIV, or TB, palpable nodes within the expected lymphatic drainage area of a cancer should be treated with modified or radical neck dissection to avoid undertreating a neck that harbors metastases.
Adjuvant irradiation: When the burden of disease outstrips a centre’s capacity to provide postoperative radiation to all deserving patients, an oncologist may have to make a call as to who are most likely to benefit. Even though studies do not rank accepted indications for postoperative radiation, Table 1 is an attempted ranking (“thumb-suck”) of indications for adjuvant radiation (not to be cited). E.g. having >2 cervical metastases is generally considered to be an indication for adjuvant radiation, even though the evidence to support this threshold is tenuous; hence centers that lack capacity to provide radiation to all deserving patients could argue that this threshold be adjusted upwards so that patients most likely to benefit e.g. with positive margins, extracapsular spread (ECS), and large tumor volumes are not deprived of adjuvant radiation30. A final (major) caveat relating to using histological criteria for adjuvant radiation is the inaccuracy of histopathological (under)reporting, especially of the presence of cervical micrometastases and perineural invasion (PNI).
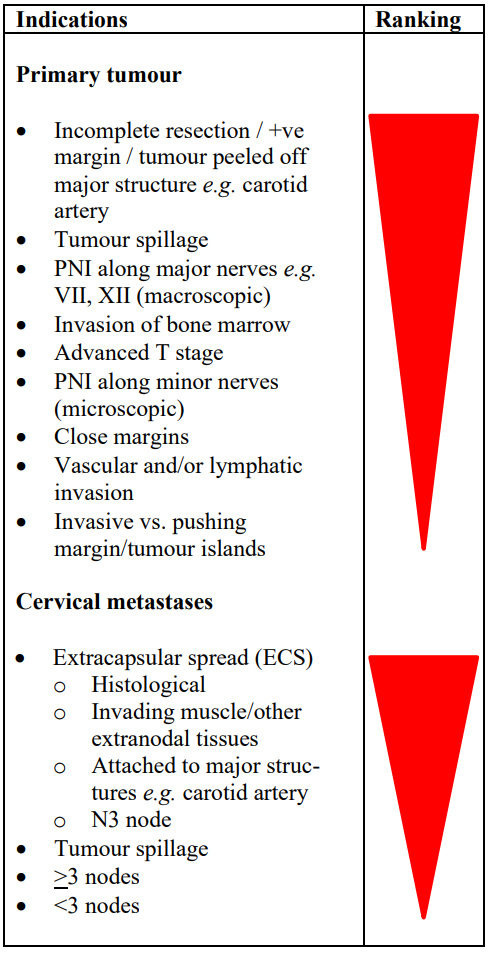
Table 1: Relative ranking of indications for postoperative irradiation (A thumb-suck, not to be cited)
Reconstruction: Although excellent functional results can be achieved with microvascular free tissue transfer flaps31, the surgery is time-consuming and requires specialized training. In the absence of microvascular free tissues transfer flaps, surgeons should become proficient at using a range of pedicled flaps e.g. pectoralis major, buccinator, temporalis, nasolabial, buccal fat pad, deltopectoral, Latissimus dorsi, paramedian forehead, trapezius, cervicofacial, supraclavicular and submental artery island flaps.
Oral cavity: When postoperative radiation is unavailable, surgery for T1 and T2 cancers should be prioritized, including cancers that are T4 due to only limited bony invasion that can be resected by marginal or segmental mandibulectomy or by partial maxillectomy. Cancers of the tongue and floor of mouth that are palpable (likely to be >4 mm thick) or are staged >T2 should undergo elective neck dissection due to the likelihood of there being occult cervical metastases. Preserving oral function is crucial; other than microvascular free transfer flaps, surgeons can employ pedicled flaps e.g. pectoralis major, buccinator, buccal fat pad, temporalis, nasolabial, and submental artery island and supraclavicular flaps. With inferior or total maxillectomy, one must separate the oral cavity from the nose; if prosthetic appliances are not available, this can be achieved with temporalis muscle flaps. Without the facility to reconstruct bone (e.g. free fibula flap), mandibular resection should not be extended beyond the midline to avoid the crippling and unsightly Andy Gump deformity (Fig 9).

Figure 9: Andy Gump deformity
Oropharynx: Management of cancers of the oropharynx has undergone a paradigm shift with the realization that HPV infection is both an etiologic and prognostic factor for a subset of oropharyngeal squamous cell carcinomas; the introduction of transoral robotic surgery (TORS) to resect oropharyngeal tumors as well as attempts to reduce the morbidity of chemoradiation by accepting smaller resection margins when combined with postoperative radiation. However, HPV testing, transoral robotic surgery, and chemoradiation are generally not available in developing world centers; neither is the ability to deal with adverse consequences of chemoradiation. Surgical resection and/or radiation are therefore the mainstay of treatment in developing countries. Pedicled flaps used to reconstruct soft palate, lateral pharyngeal wall or base of tongue include pectoralis major, buccinator, buccal fat pad, supraclavicular, submental artery island and temporalis flaps.
Larynx and Hypopharynx: In developed world centers early cancers are commonly excised with CO2 laser. Advanced cancers (dysfunctional larynx, cartilage invasion, tracheostomy for stridor) are treated with total laryngectomy. The remainder are offered chemoradiation with surgery reserved for salvage for persistent or recurrent cancer. CO2 laser is generally not available in developing world centers; chemoradiation is expensive and the package of care required to manage both acute and late consequences and complications of chemoradiation (dysphagia, PEG feeds, cancer surveillance with MRI and PET scans, complex salvage surgery, hypothyroidism, hypocalcaemia) is lacking. Therefore such centres have to rely on open approaches such as laryngofissure, vertical partial, supraglottic, supracricoid and neartotal laryngectomy for smaller cancers, and total laryngectomy for advanced cancers. When performing total laryngectomy, the surgeon should attempt to preserve both thyroid lobes and the parathyroids to minimize the risks of hypothyroidism and hypoparathyroidism, particularly when thyroid and calcium monitoring and replacement are difficult or impossible. With a dedicated speech therapy service, fistula (Figure 10) speech results can be achieved that match those of developed world centers even with poor, illiterate patients living long distances from treatment centers32. Voice prostheses are however expensive; hence the adoption of strategies such as using removable prostheses as indwelling prostheses to reduce expense32. Heat moisture exchange (HME) devices are used in developed world centers to humidify and warm inspired air; however, a homemade cloth stoma cover/bib (Figure 11) is equally effective at a fraction of the cost33.

Figure 10: Speaking valve

Figure 11: Cheap homemade Bib
Although esophageal speech does not cost anything, only 27% of patients in a Brazilian study mastered esophageal speech34. Another option is to use mucosal shunts; however, the surgery is technically difficult and can be used only in highly selected patients with good pulmonary function who can cope with aspiration34. Because there is a severe shortage of speech therapists in many developing countries28,34 an electrolarynx is a reasonable alternative to achieve post-laryngectomy speech.
Nasopharynx: Cancers of the nasopharynx occur mainly in the developing world (Figure 5). Chemoradiation is the mainstay of treatment35. However, the supportive care required for the extreme chemotoxicity is not always available in developing countries. Patients generally present with advanced disease; in an unpublished study conducted in Cape Town, South Africa, 50% of patients presented with Stage 4B disease of which 28% did not complete treatment for socioeconomic reasons (Dalvie et al: unpublished data). Hence it may be prudent to accept lower survival using radiation alone, than to try to improve survival with concurrent CTRT with its attendant morbidity. Intensity modulated radiotherapy (complex 3D conformal radiotherapy) has a documented survival benefit and can improve quality of life due to reduced xerostomia; however, this generally is not available in developing countries35.
Thyroid: Most thyroidectomies in developing countries are done by surgeons not specializing in endocrine surgery. Bilateral recurrent laryngeal nerve injury causing airway compromise, or hypoparathyroidism causing hypocalcaemia in situations where monitoring serum calcium and treating hypocalcaemia with calcium and Vitamin D are not possible may have fatal consequences. Regardless of surgical expertise, complication rates rise with the extent of resection. Subtotal thyroidectomy preserves the blood supply to the parathyroid glands and reduces the risk of hypocalcaemia. Thyroid lobectomy causes hypothyroidism in 25% of cases, but almost never causes significant hypoparathyroidism. Total thyroidectomy is however associated with both increased short- and long-term morbidity relating to recurrent laryngeal nerve paralysis and hypocalcaemia, particularly in an occasional thyroid surgeon’s hands. In the absence of convincing evidence that total thyroidectomy confers survival benefit in favourable, differentiated thyroid cancer36, 37 (especially when I131 therapy is not available), coupled with the morbidity and mortality of total thyroidectomy where calcium monitoring and replacement are suboptimal, the occasional thyroid surgeon practicing in a developing world center may be wise to perform thyroid lobectomy or subtotal thyroidectomy for such cases.
PREVENTION AND SCREENING
Given the cancer tsunami that the developing world is facing, and the late presentations of patients with advanced disease, prevention (education and anti-smoking campaigns) and screening would appear to be reasonable strategies to adopt. Yet a Cochrane review reported that visual screening for oral cancer did not have survival benefit, although there was some evidence that it might be effective in high-risk patients38, 39. Techniques using toluidine blue staining, brush biopsy or cytology, or fluorescence imaging as primary screening tool or as adjunct for screening have also not been shown to have any benefit38. Consequently, it appears that based on current evidence, there are more important interventions than investing scarce human and financial resources in screening that will improve head and neck cancer outcomes in a resource constrained setting. Our efforts should rather be directed towards educating patients to seek advice early when symptoms appear, educating doctors and health care workers to recognize potential malignancies and to refer patients early for appropriate management.
CONCLUDING REMARKS
There needs to be a global effort to educate and train oncologists and surgeons to manage head and neck cancer in developing countries through residency programs, clinical fellowships and outreach projects. These should focus on developing sustainable head and neck cancer programs, integrating them with existing local services and focusing on teaching and training. Open access to journals and textbooks should be encouraged. In addition, a multifaceted approach is required including lobbying international organizations, governments and aid organizations to support infrastructure development and research, and for industry to provide appropriate and affordable technology. In this way the developed world can make a substantial difference to the outcome of the enormous head and neck cancer burden in the developing world.
REFERENCES
- Farmer P, Frenk J, Knaul FM, et al. Expansion of cancer care and control in countries of low and middle income: a call to action. Lancet. 2010 Oct 2;376 (9747):1186-93
- Warnakulasuriya S. Global epidemiology of oral and oropharyngeal cancer. Oral Oncology. 2009;45:309-16
- GLOBOCAN 2008: International Agency for Research on Cancer (IARC), 2013 http://gco.iarc.fr/
- Yoshizaki T, Ito M, Murono S, et al. Current understanding and management of nasopharyngeal carcinoma. Auris Nasus Larynx 2012;39:137-44
- Maxwell JH, Kumar B, Feng F, et al. HPV positive, P16 positive, EBV negative nasopharyngeal cancer in White Americans. Head Neck 2010; 32(5): 562-7
- UNAIDS 2012 http://www.unaids.org/en/regionscountries/countries/southafrica/ (Accessed on 19 October 2013)
- R Newton, J Ziegler, C Ateenyi-Agaba, et al. The epidemiology of conjunctival squamous cell carcinoma in Uganda. Br J Cancer. 2002 July 29; 87(3): 301– 8
- Engsing FN, Gerstoft J, Kronborg G, et al. Head and neck cancer in HIV patients and their parents: a Danish cohort study. Clin Epidemiol. 2011; 3: 217–27
- Onyango JF, Macharia IM. Delays in diagnosis, referral and management of head and neck cancer presenting at Kenyatta National Hospital, Nairobi. East Afr Med J. 2006 Apr;83(4):85-91
- Da Lilly-Tariah OB, Somefun AO, Adeyemo WL. Current evidence on the burden of head and neck cancers in Nigeria. Head Neck Oncol. 2009 May 28;1:14 http://www.headandneckoncology.org/content/1/1/14 (Accessed on 19 October 2013)
- Abdel-Wahab M, Bourque J-M, Pynda Y, et al. Status of radiotherapy resources in Africa: an International Atomic Energy Agency analysis. The Lancet Oncology. April 2013;14(4): e168 - e175) http://www.thelancet.com/journals/lanonc/article/PIIS1470-2045%2812%2970532-6/fulltext#article_upsel
- Stephenson KA, Fagan JJ. Do Proton Pump Inhibitors Reduce the Incidence of Pharyngocutaneous Fistulae following Total Laryngectomy? Head Neck [Epub ahead of print]
- Jensen AR, Nellemann HM, Overgaard J. Tumor progression in waiting time for radiotherapy in head and neck cancer. Radiother Oncol. 2007 Jul;84(1):5- 10
- Sanfilippo NJ, Mitchell J, Grew D, DeLacure M. Toxicity of head-and-neck radiation therapy in human immunodeficiency virus-positive patients Int J Radiat Oncol Biol Phys. 2010 Aug 1;77(5):1375-9.
- Klein EA, Guiou M, Farwell DG, et al. Primary radiation therapy for head and neck cancer in the setting of human immunodeficiency virus. Int J Radiat Oncol Biol Phys. 2011 Jan 1;79(1):60- 4
- Housri N, Yarchoan R, Kaushal A. Radiotherapy for HIV patients: Are special precautions necessary? Cancer. 2010;116(2):273–83
- Global update on HIV treatment 2013: Results, impact and opportunities. WHO report in partnership with UNICEF and UNAIDS. June 2013 http://apps.who.int/iris/bitstream/10665/85326/1/9789241505734_eng.pdf (Accessed 19 October 2013)
- Okumu G, Makobore P, Kaggwa S, et al. Effect of emergency major abdominal surgery on CD4 cell count among HIV positive patients in a subSaharan Africa tertiary hospital - a prospective study. BMC Surg. 2013 Feb 26;13:4. doi: 10.1186/1471-2482-13-4
- Cacala SR, Mafana E, Thomson SR, Smith A. Prevalence of HIV status and CD4 counts in a surgical cohort: their relationship to clinical outcome. Ann R Coll Surg Engl. 2006 Jan;88(1):46-51
- Su J, Tsun A, Zhang L, et al. Preoperative risk factors influencing the incidence of postoperative sepsis in human immunodeficiency virus-infected patients: a retrospective cohort study. World J Surg. 2013 Apr;37(4): 774-9
- Guild GN, Moore TJ, Barnes W, Hermann C. CD4 count is associated with postoperative infection in patients with orthopaedic trauma who are HIV positive. Clin Orthop Relat Res. 2012 May; 470(5):1507-12
- Tatsuzaki H, Levin CV. Quantitative status of resources for radiation therapy in Asia and Pacific region. Radiother Oncol. 2001 Jul;60(1):81-9
- Zubizarreta EH, Poitevin A, Levin CV. Overview of radiotherapy resources in Latin America: a survey by the International Atomic Energy Agency (IAEA). Radiother Oncol. 2004 Oct;- 73(1):97-100
- Rønjom MF, Brink C, Bentzen SM, et al. Hypothyroidism after primary radiotherapy for head and neck squamous cell carcinoma: Normal tissue complication probability modelling with latent time correction. Radiother Oncol. 2013 Nov;109(2):317-22
- Machtay M, Moughan J, Trotti A, et al. Factors associated with severe late toxicity after concurrent chemoradiation for locally advanced head and neck cancer: an RTOG analysis. J Clin Oncol 2008;26:3582-9
- Kumar S, Pandey M, Lal P, et al. Concomitant boost radiotherapy with concurrent weekly cisplatin in advanced head and neck cancers: a phase II trial. Radiother Oncol. 2005 May;75 (2):186-92
- Overgaard J, Mohanti BK, Begum N, et al. Five versus six fractions of radiotherapy per week for squamous cell carcinoma of the head and neck (IAEA-ACC study): a randomised, multicentre trial. The Lancet Oncology 2012;11(6):553 - 60
- Fagan JJ, Jacobs M. Survey of ENT services in Africa: need for a comprehensive intervention. Global Health Action, Vol 2 (2009) http://journals.sfu.ca/coaction/index.ph p/gha/article/view/1932
- Fu KK, Pajak TF, Trotti A, et al. A Radiation Therapy Oncology Group (RTOG) phase III randomised study to compare hyperfractionation and two variants of accelerated fractionation to standard fractionation radiotherapy for head and neck squamous cell carcinomas: first report of RTOG 9003. Int J Radiat Oncol Biol Phys 2000;48(1):7- 16
- Brown JS, Shaw RJ, Bekiroglu F, Rogers SN. Systematic review of the current evidence in the use of postoperative radiotherapy for oral squamous cell carcinoma. Br J Oral Maxillofac Surg. 2012 Sep;50(6):481-9
- Van Zyl JH, Fagan JJ. Principles and technique of microvascular anastomosis for free tissue transfer flaps in head and neck reconstructive surgery in The Open Access Atlas of Otolaryngology, Head and Neck Surgery https://vula.uct.ac.za/access/content/group/ba5fb1bd-be95-48e5-81be586fbaeba29d/Principles%20and%20technique%20of%20microvascular%20anastomosis%20for%20free%20tissue%20transfer%20flaps%20in%20head%20and%20neck%20reconstructive%20surgery.pdf (Accessed on 20 October 2013)
- Fagan JJ, Lentin R, Quail G. International practice of laryngectomy rehabilitation interventions: a perspective from South Africa. Curr Opin Otolaryngol Head Neck Surg. 2013 Jun;21(3): 199-204
- Quail G, Fagan JJ, Raynham O, Krynauw MH, John LR, Carrara MH. The effect of cloth stoma covers on tracheal climate of laryngectomy patients. Head Neck. 2016 Apr;38 Suppl 1:E480-7
- Vartanian JG, Carrera-de-Angelis E, Kowalski LP. Practice of laryngectomy rehabilitation interventions: a perspective from South America Curr Opin Otolaryngol Head Neck Surg 2013, 21:212-7
- Bourhis J, Le Maitre A, Baujat B, et al. Individual patients’ data meta-analyses in head and neck cancer. Curr Opin Oncol 2007;19:188-94
- Lee J, Park JH, Lee CR, Chung WY, Park CS. Long-term outcomes of total thyroidectomy versus thyroid lobectomy for papillary thyroid microcarcinoma: comparative analysis after propensity score matching. Thyroid. 2013; 23(11):1408-15
- Nixon IJ, Ganly I, Patel SG, et al. Thyroid lobectomy for treatment of well differentiated intrathyroid malignancy. Surgery. 2012;151(4):571-9
- Brocklehurst P, Kujan O, Glenny AM, et al. Screening programmes for the early detection and prevention of oral cancer. Cochrane Database Syst Rev. 2010 Nov 10;(11):CD004150
- National Cancer Institute: PDQ® Oral Cancer Screening. Bethesda, MD: National Cancer Institute. Date last modified 09/26/2013. Available at: http://cancer.gov/cancertopics/pdq/scre ening/oral/HealthProfessional (Accessed 19 October 2013)
Chapter updated: September 2019
Author and Editor
Johan Fagan MBChB, FCS(ORL), MMed
Professor and Chairman
Division of Otolaryngology
University of Cape Town
Cape Town, South Africa
johannes.fagan@uct.ac.za
Authors
Clare Stannard MB.BS, FFRadOnc (SA)
Associate Professor Division of Radiation Oncology,
Groote Schuur Hospital,
University of Cape Town
Cape Town, South Africa
clare.stannard@uct.ac.za
Sameera Dalvie MBChB, FFRadOnc (SA)
Specialist Division of Radiation Oncology,
Groote Schuur Hospital,
University of Cape Town
Cape Town, South Africa
s.dalvie@uct.ac.za

The Open Access Atlas of Otolaryngology, Head & Neck Operative Surgery by Johan Fagan (Editor) johannes.fagan@uct.ac.za is licensed under a Creative Commons Attribution - Non-Commercial 3.0 Unported License



