1.34: Floor of Mouth Cancer Resection
- Page ID
- 17665
OPEN ACCESS ATLAS OF OTOLARYNGOLOGY, HEAD & NECK OPERATIVE SURGERY
RESECTION OF FLOOR OF MOUTH CANCER
Johan Fagan, Gerrit Viljoen
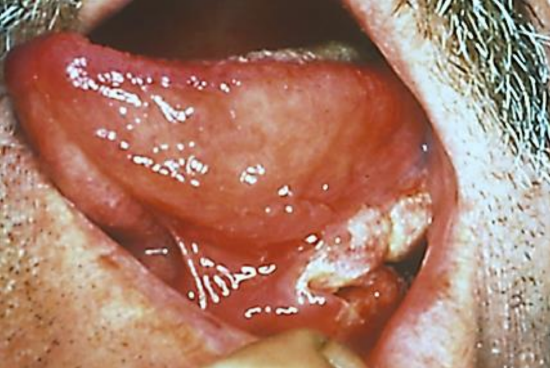
Figure 1: Cancer of lateral FOM
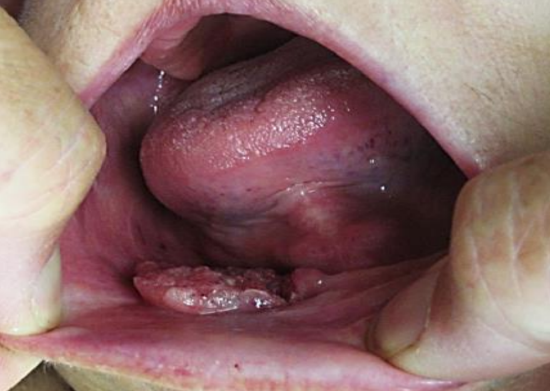
Figure 2: Cancer of anterior FOM
Cancers of the floor of the mouth (FOM) are generally treated with primary surgical resection (Figures 1 & 2). Adjuvant irradiation is indicated for advanced tumors, tumors with perineural invasion (PNI) or uncertain/close margins.

Figure 3: Andy Gump deformity
Resection of cancers of the FOM without taking cognisance of oral function may severely cripple the patient in terms of speech, mastication, oral transport and swallowing. Resecting the anterior arch of the mandible beyond the midline without reconstructing the bone with loss of the anterior attachments of the suprahyoid muscles (digastric, geniohyoid, mylohyoid, genioglossus) leads to an Andy Gump deformity with loss of oral competence, drooling, and a very poor cosmetic outcome (Figure 3).
Surgical Anatomy
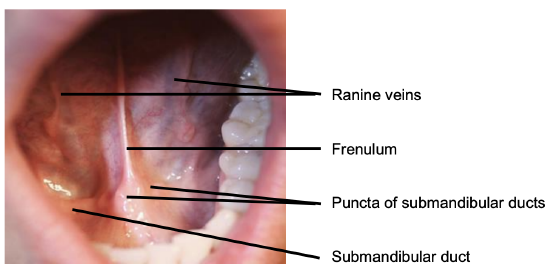
Figure 4: Anterior FOM
The FOM is a horseshoe-shaped area that is confined peripherally by the inner aspect (lingual surface) of the mandible. It extends posteriorly to where the anterior tonsillar pillar meets the tonsillolingual sulcus, and merges medially with under-surface of the oral tongue. It has a covering of delicate oral mucosa through which the thin walled sublingual/ranine veins are visible. The frenulum is a mucosal fold that extends along the midline between the openings of the submandibular ducts (Figure 4).
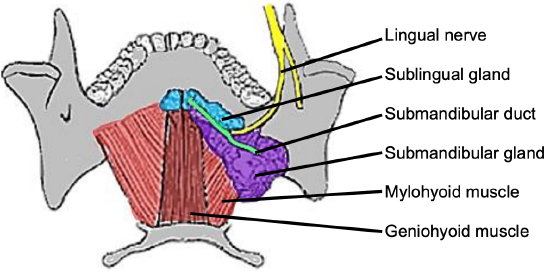
Figure 5: Superior view of FOM and submandibular gland and duct, lingual nerve and mylohyoid and geniohyoid muscles
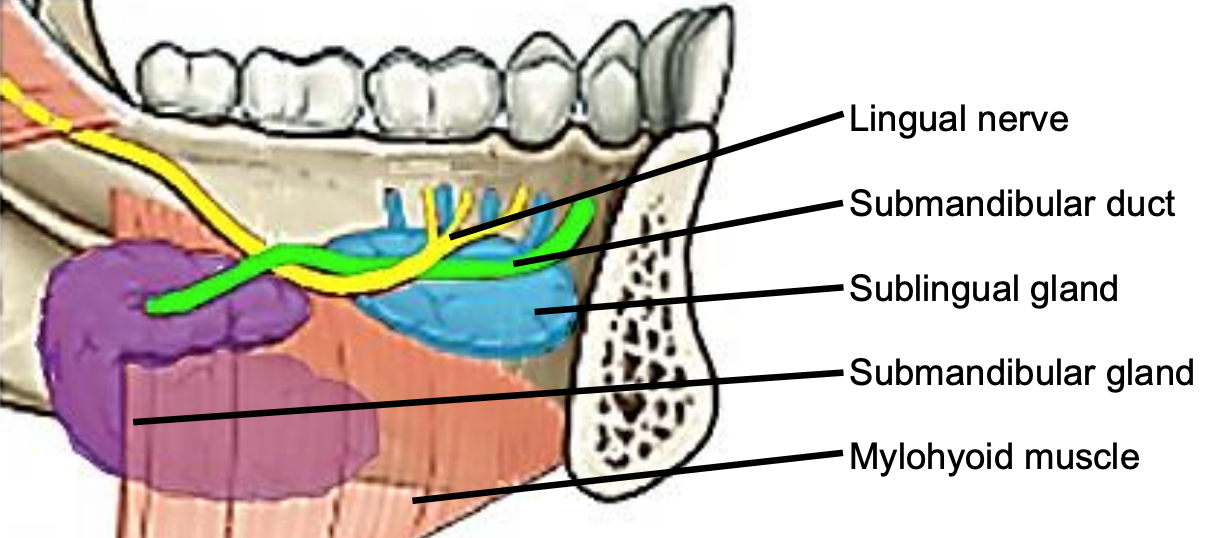
Figure 6: Intraoral view of left sublingual gland with ducts of Rivinus, submandibular gland and duct, lingual nerve and mylohyoid muscles
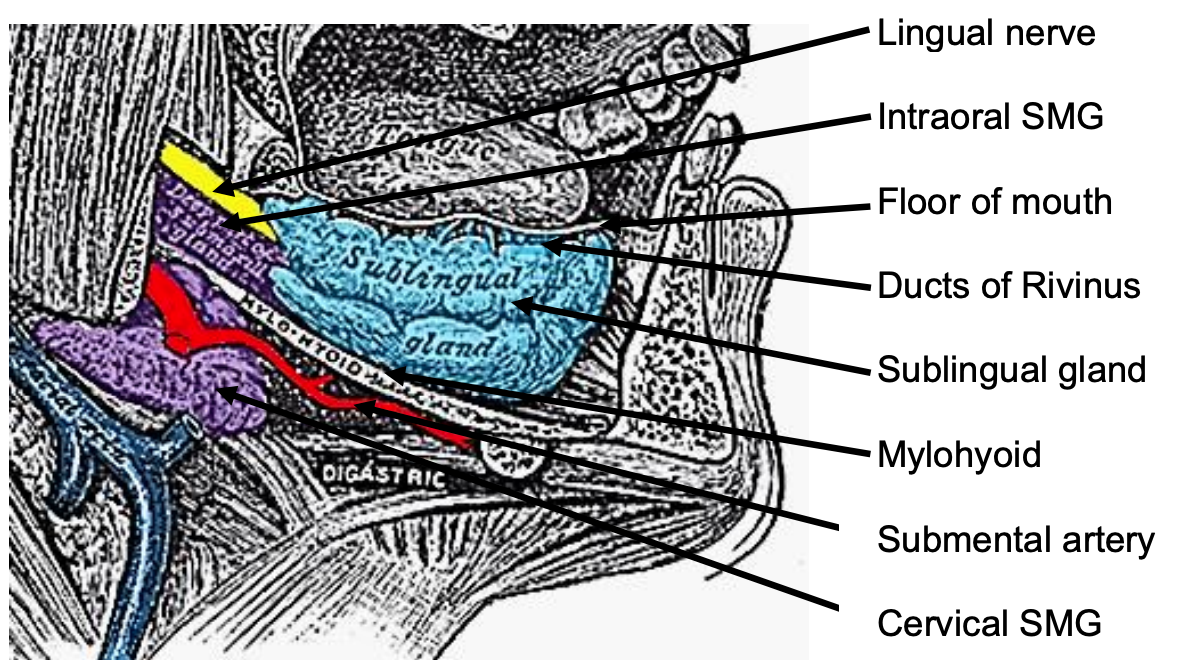
Figure 7: Sagittal view floor of mouth
The mylohoid muscle forms the diaphragm of the mouth and separates the FOM from the submental and submandibular triangles of the neck (Figures 5, 6, 7).
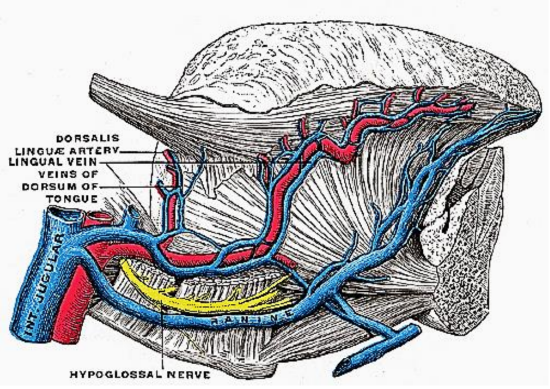
Figure 8: XIIn accompanied by ranine veins
The following structures are located between the mucosa and the mylohyoid muscle: paired geniohyoid muscles in the midline (Figure 5); sublingual salivary glands (Figures 5-7), submandibular ducts (Figures 5, 6), oral component of submandibular salivary glands (Figures 5-7), genioglossus muscle (Figure 8), and the lingual (Figures 5-7) and hypoglossal nerves (Figure 8).
The paired sublingual salivary glands are located beneath the mucosa of the anterior floor of mouth, anterior to the submandibular ducts and above the mylohyoid and geniohyoid muscles (Figures 5, 6, 7). The glands drain via 8-20 excretory ducts of Rivinus into the submandibular duct and also directly into the mouth on an elevated crest of mucous membrane called the plica fimbriata which is formed by the gland and is located to either side of the frenulum of the tongue (Figures 6, 7).
The submandibular duct is located immediately deep to the mucosa of the anterior and lateral FOM and opens into the oral cavity to either side of the frenulum (Figures 4, 5, 6).
The lingual nerve provides sensation to the floor of the mouth. It crosses deep to the submandibular duct in the lateral floor of mouth (Figures 5, 6, 7). In the anterior FOM it is located posterior to the duct (Figure 5).
Ranine veins are visible on the ventral surface of the tongue, and accompany the hypoglossal nerve (Figures 4, 8).
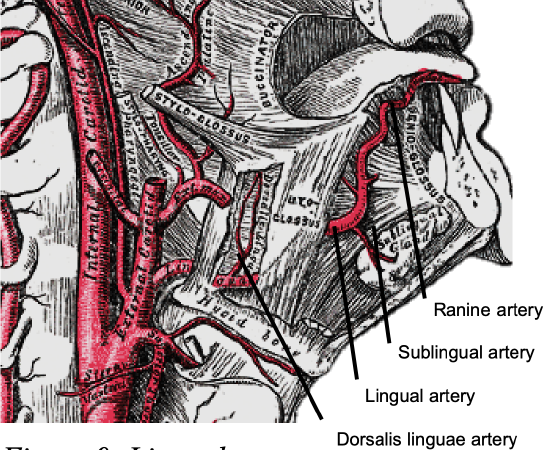
Figure 9: Lingual artery
Arterial supply to the tongue and FOM is derived from the lingual artery and its branches (ranine artery, dorsalis linguae, and sublingual arteries) (Figures 8, 9); and the mylohyoid and submental branches of the facial artery.
The lingual artery arises from the external carotid artery between the superior thyroid and facial arteries and courses obliquely forwards and medial to the greater cornu of the hyoid (Figures 8, 9).
It then loops downward and anteriorly and crosses medial to XIIn and the stylohyoid muscle. It then courses directly anteriorly below hyoglossus and finally ascends as the ranine artery (profunda linguae) submucosally on the undersurface of the tongue as far as its tip; it lies to either side of the genioglossus and is accompanied by the lingual nerve. Two or three small dorsales linguae arteries arise beneath the hyoglossus and ascend to the posterior part of the dorsum of the tongue and supply the mucous membrane of the posterior FOM, and oropharynx. The sublingual artery arises from the lingual artery at the anterior edge of the hyoglossus and runs forward between the genioglossus and mylohyoid and supplies the sublingual salivary gland and mucous membrane of the FOM and gingiva (Figures 8, 9). A branch of the sublingual artery pierces the mylohyoid muscle and anastomoses with the submental branch of the facial artery.
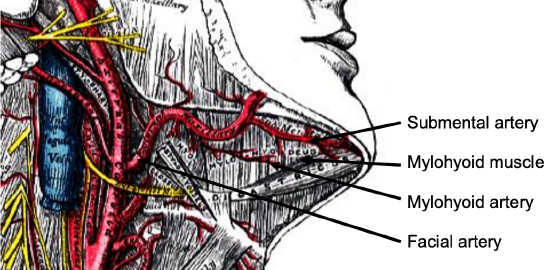
Figure 10: Facial artery emerges from behind posterior belly of digastric (removed), and gives rise to a few branches including submental artery
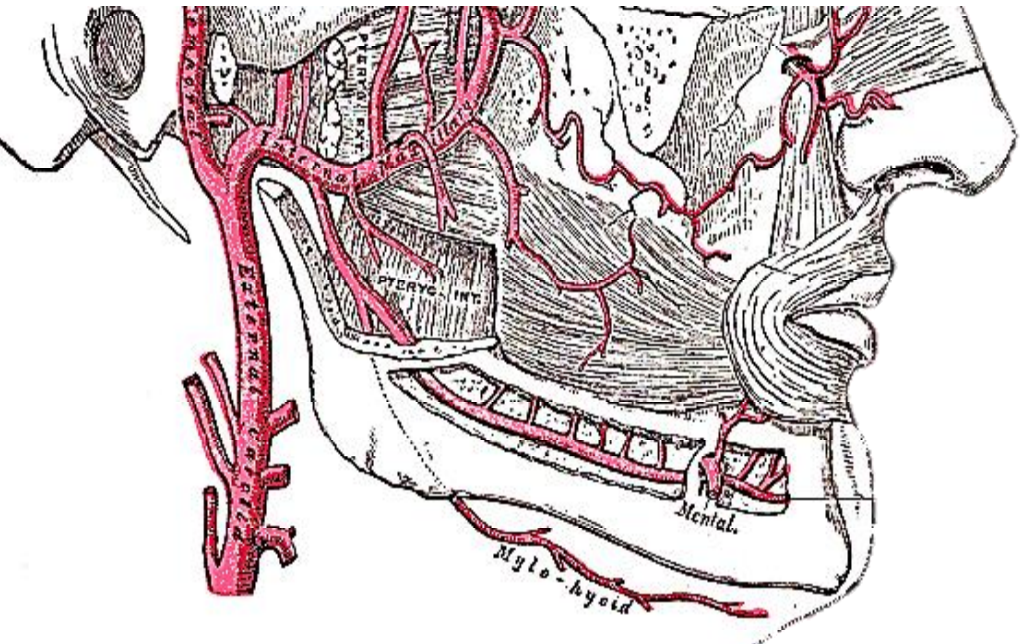
Figure 11: Mylohyoid artery is a branch of the inferior alveolar artery
The submental branch of the facial artery courses along the inferior, inner margin of the mandible (Figure 10). The mylohyoid artery and vein are encountered when the surgeon elevates the submandibular gland from the lateral surface of the mylohyoid (Figures 10 & 11). It branches from the inferior alveolar artery just before it enters the mandibular foramen, crosses the mylohyoid, and disappears anteriorly behind the digastric. It has connections with the submental artery, and via a defect in the mylohyoid with the sublingual artery in the floor of the mouth.
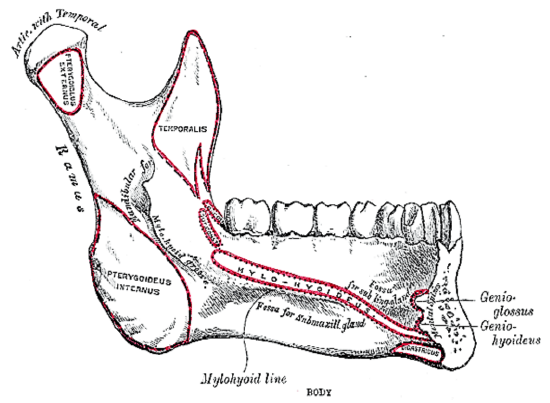
Figure 12a: Attachments of mylohyoid, geniohyoid, genioglossus and digastric muscles to inner aspect of mandible
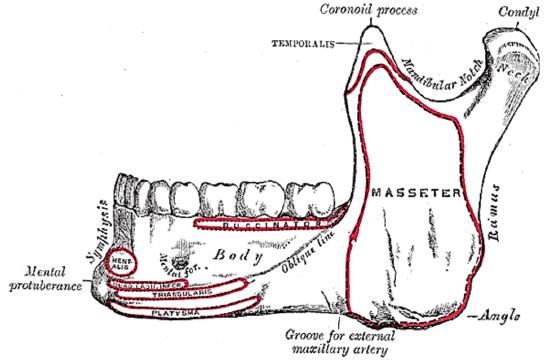
Figure 12b: Attachments of muscles to outer aspect of mandible and location of mental foramen
The mandible forms the peripheral border of the FOM and may be involved by FOM tumors or may have to be divided (mandibulotomy) or resected (alveolectomy/marginal mandibulectomy/segmental mandibulectomy/hemimandibulectomy). Important surgical features are the position of the mental foramina through which the mental nerves exit to innervate the lower lip, the mylohyoid line to which the mylohyoid muscle attaches, and when planning a marginal mandibulectomy, the height of the body of the mandible and the depth of the dental roots (Figures 12a, b).
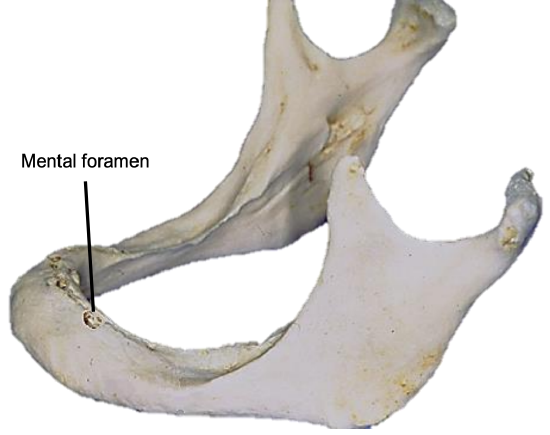
Figure 13: Position of mental foramen in a resorbed mandible
The mental foramen and inferior alveolar nerve may be very close to the superior surface of a resorbed mandible such as is seen in older, edentulous patients (Figure 13). A marginal mandibulectomy may also not be possible in such a resorbed mandible due to the lack of residual bone.
Surgical Objectives
- Clear resection margins
- Avoid postoperative orocervical fistulae
- Optimize cosmesis and function
- Maintain length and mobility of the tongue
- Avoid pooling of secretions and food in the reconstructed FOM
- Avoid obstruction of the submandibular ducts
- Avoid injury to lingual and hypoglossal nerves
- Maintain mandibular continuity and strength
- Restore dentition
The author advocates elective neck dissecttion (END) levels I-IV for squamous cell carcinomas that are >4 mm thick and/or >T2 stage. A useful rule of thumb is that a palpable tumor is likely to have a tumor thickness that warrants END. Tumors of the anterior FOM that approach the midline require bilateral END.
The remainder of this chapter will focus on the surgical treatment of the primary tumor.
Preoperative evaluation
- Are there synchronous primaries, or cervical or distant metastases?
- Is the tumor resectable? Other than tumors that extend posteriorly to involve the carotid sheath, very few FOM tumors are irresectable. The key consideration is surgical morbidity; this in turn is dependent on the range and reliability of reconstructive surgery that are available e.g. resection of the mandible beyond the midline can only be done if it is possible to reconstruct the bone with a free fibula flap to avoid an Andy Gump deformity (Figure 3).
- Is there clinical evidence of perineural invasion (PNI)? Examine the patient for neurological deficits of the hypoglossal, mental, inferior alveolar and lingual nerves. Widening of the alveolar canal on mandibular orthopantomography (Panorex) may be seen. MRI can demonstrate PNI. Should there be evidence of PNI then the affected nerve should be dissected proximally until a clear tumor margin is obtained on frozen section. In the case of the inferior alveolar nerve, this would require a hemi- or segmental mandibulectomy that encompasses the entire inferior alveolar canal.
- Status of dentition? Carious teeth may cause osteoradionecrosis. Carious teeth seen on Panorex may be removed at the time of the surgery
- Mandible: FOM cancers involving mandible require adequate oncological margins, may require osseous flap reconstruction, and have high local recurrence rates. Tumor may involve periosteum, invade only the inner cortex, or involve medullary bone. Tumors involving only the lingual alveolar mucosa adjacent to mandible but not invading bone, or eroding cortex only, require only a marginal mandibulectomy, whereas frank invasion of mandible, and involvement of the medulla require segmental resection.
Risk factors for tumor invasion of the mandible include size, site, and depth of invasion of the soft tissues; larger and deeper tumors are more likely to extend to, and eventually invade the mandible. The pattern of spread within bone is related to tumor extent, rather than anatomic factors providing potential routes and barriers to tumor spread1. Tumors enter the mandible at points of abutment (often junction of attached and reflected mucosa in both dentate and edentulous mandibles) rather than preferentially through the occlusal surface, neural foramina, or periodontal membranes. The presence or absence of teeth does not influence patterns of invasion, and the increased height of alveolar bone in dentate jaws does not delay invasive patterns of bone involvement.
There are two patterns of tumor invasion of the mandible;1 with an invasive pattern, fingers and islands of tumor advance independently into the cancellous spaces with little osteoclastic activity and no intervening connective tissue; with an erosive pattern, tumor advances over a broad front with a connective tissue layer and active osteoclasts separating tumor from bone. An erosive pattern can progress to an invasive pattern, depending on the extent and depth of invasion. An invasive pattern is associated with a significantly higher local recurrence rate and lower 5-year disease specific survival2.
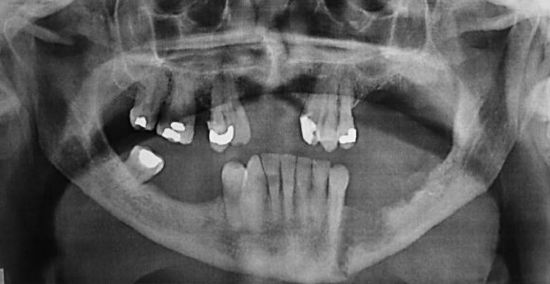
Figure 14: Panorex view of erosion and pathological fracture
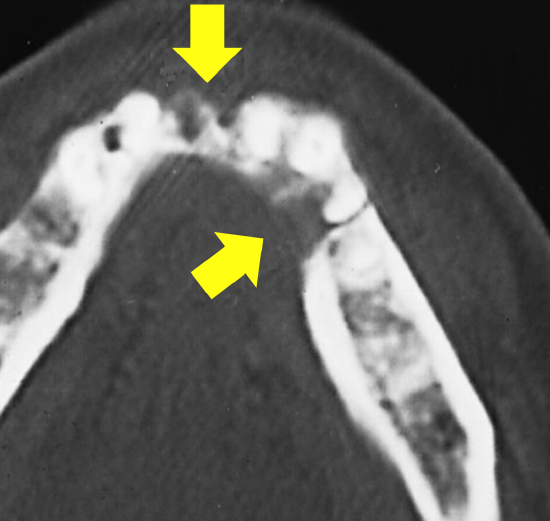
Figure 15: Cortical erosion visible on CT
Panoramic radiography (Panorex) is inexpensive and provides an excellent view of the body, ramus, and inferior alveolar canal (Figure 14). It has 80-92% sensitivity and 72-88% specificity for invasion of mandible.3,4 The overlying spine can obscure the symphyseal region, and it may be difficult to differentiate between periodontal disease and tumor erosion. CT scans (Figure 15) tend to underestimate the presence and extent of invasion. MRI and single photon emission CT (SPECT) are much more sensitive, but they tend to overestimate the extent of bone involvement.5
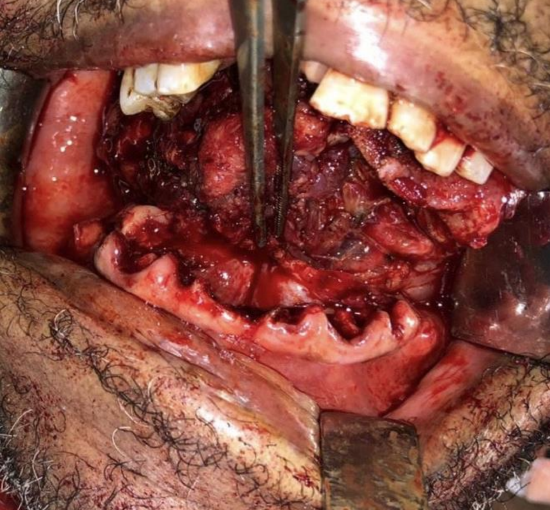
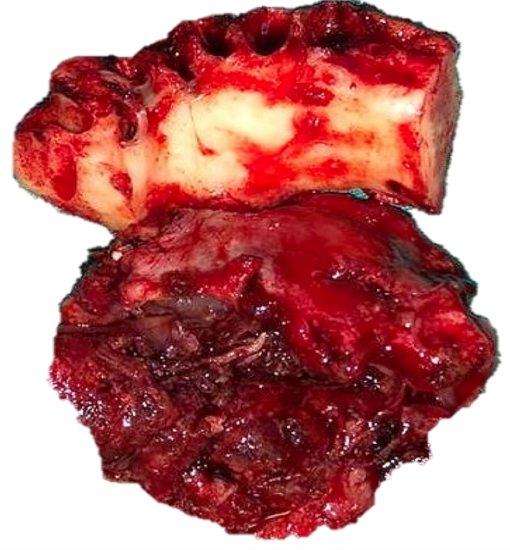
Figure 16a, b: Periosteal stripping reveals normal bone and suitability for marginal mandibulectomy
In the absence of CT scanning, a surgeon may elect to do periosteal stripping of tumor off the bone at the time of resection to inspect the bone for evidence of tumor infiltration and to decide how to proceed. Periosteal stripping is easily done using a periosteal elevator and is the most accurate method to determine bone erosion.6 It has a sensitivity of 96% and specificity of 65%.3 (Figures 16a,b).
Indications for bone resection include preoperative clinical or radiographic findings suggesting mandibular involvement; or if tumor is adherent to mandible and radiographic invasion is absent, intraoperative periosteal stripping can be done to for evidence of bone invasion as the extent of the invasion will dictate whether a marginal or segmental resection is indicated.
Marginal mandibulectomy is indicated in cancers encroaching on, adherent to, or superficially invading mandibular cortex; gingival carcinoma with superficial bone invasion at the level of the dental alveolus; and for cortical invasion, but only if >1 cm height of the lower contour of remaining mandible is retained. Therefore, it is important to determine the vertical height of the mandible clinically or by Panorex to predict whether a free bony composite flap would be required to bolster the remaining mandible.
Segmental mandibulectomy is indicated for medullary invasion; previous radiation therapy; to maintain oncological safety margin of soft tissue in more deeply invading tumors; and if inadequate bone would remain if marginal mandibulectomy is done.
Resection margins of at least 1-2 cm of uninvolved bone as measured from the macroscopic tumor or suspected bone involvement is required. This may require removal of 1 tooth on either side of the tumor in dentate patients. Some surgeons include the entire nerve-bearing segment when bone marrow invasion present.
6. What type of reconstruction is required?
7. Is a tracheostomy required? Patients with small tumors of the anterior FOM may not require temporary tracheostomy. However, whenever the laryngeal support of the mylohyoid, geniohyoid and genioglossus muscles is lost, and especially when a flap is used to reconstruct a FOM defect, the patient is at risk of airway obstruction and should have a temporary tracheostomy inserted.
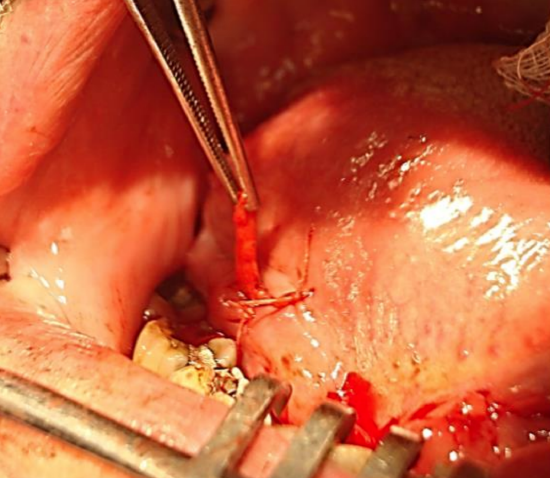
Figure 17: Submandibular duct translocated to right lateral floor of mouth and sutured to mucosa with a vicryl suture
8. Submandibular salivary glands: A tumor of the FOM may invade the submandibular duct and cause a hard, fibrotic submandibular salivary gland that can be confused with a lymph node metastasis. In a small T1 anterior FOM tumor that involves the duct in which an END is not planned the remaining duct may have to be translocated so as preserve salivary flow. This is simply done by tunneling over the duct with a hemostat just below the mucosa of the lateral FOM. The end of the transected duct is then grasped with the hemostat and passed posteriorly along the tunnel and delivered through a stab incision. It is secured to the mucosa with an absorbable suture (Figure 17).
Anesthesia
Nasal intubation is followed by tracheostomy. Perioperative antibiotics are administered for 24 hours.
Surgical access
Figure 18: Dental bite block is interposed between lateral teeth to keep mouth open
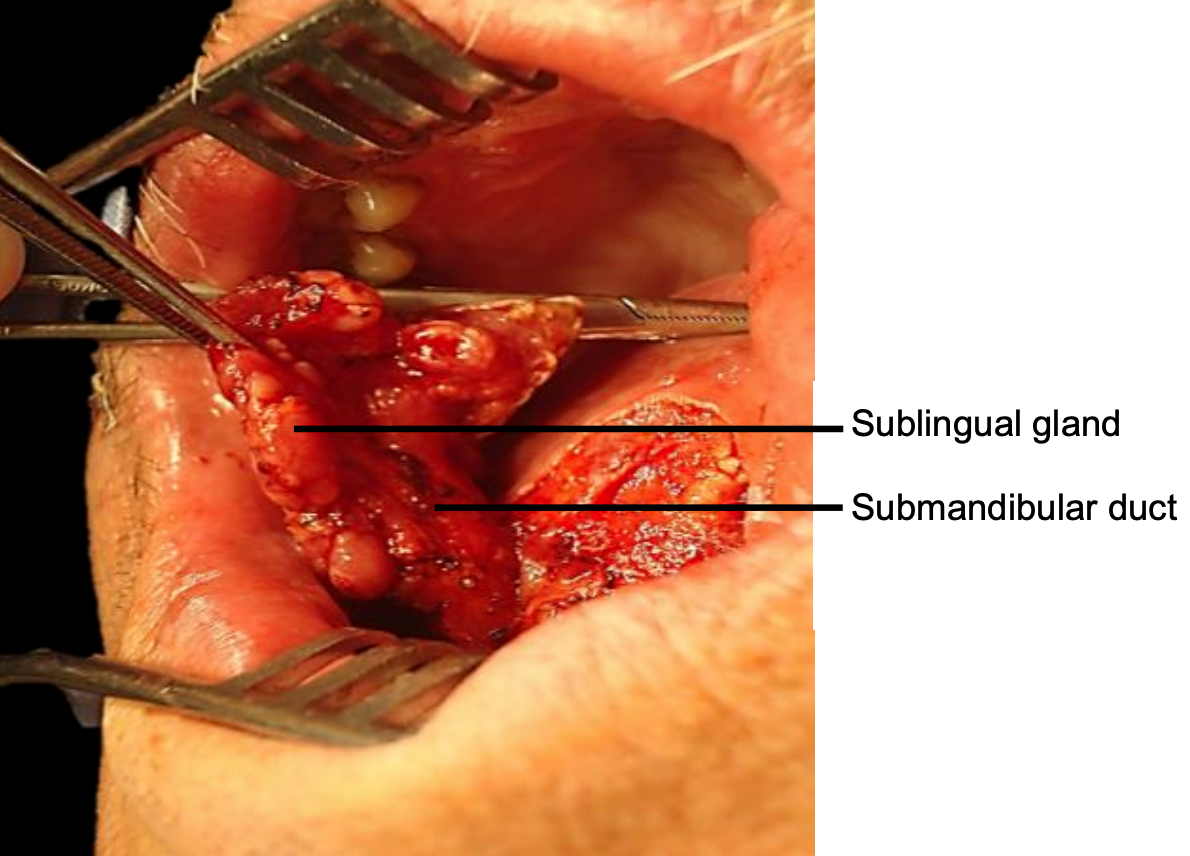
Figure 19: Self-retaining retractor in place; sublingual salivary gland and submandibular duct
Transoral: Smaller tumors (T1-T2) are usually easily excised though the open mouth in an edentulous patient. The mouth is kept widely open either with a dental bite bock (Figure 18) or with a selfretaining retractor taking care to protect the teeth (Figure 19).
However, resecting an anterior FOM tumor may be quite challenging in a patient with a full set of lower teeth as the teeth may obstruct access to the anterior FOM.
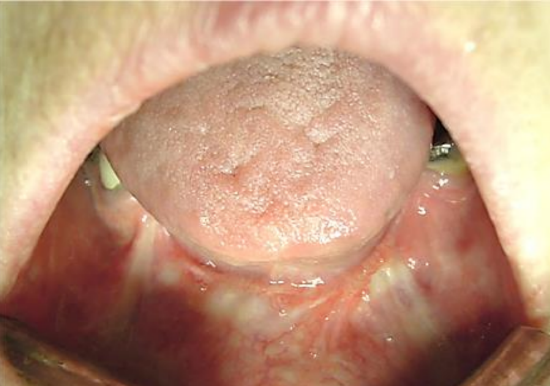
Figure 20: This marginal mandibulectomy defect was covered by advancing gingivolabial mucosa over the bone and suturing it to the edge of the FOM defect
In such cases lower teeth may need to be extracted and a marginal mandibulectomy (Figure 20) or a mandibulotomy may be required for access. When a FOM tumor abuts the anterior mandible then doing a marginal mandibulectomy in continuity with the tumor re-section greatly facilitates resection by freeing up the tumor.
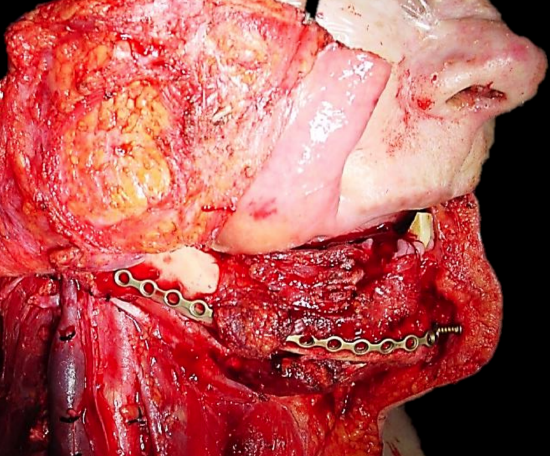
Figure 21: Midline lip-split incision and preplating of mandible before performing segmental mandibulectomy and resecting tumor
Midline lip-split: The lip is split in the midline after scoring/marking the vermillion border to ensure an accurate repair. The soft tissues are stripped off the front of the mandible (Figure 21).
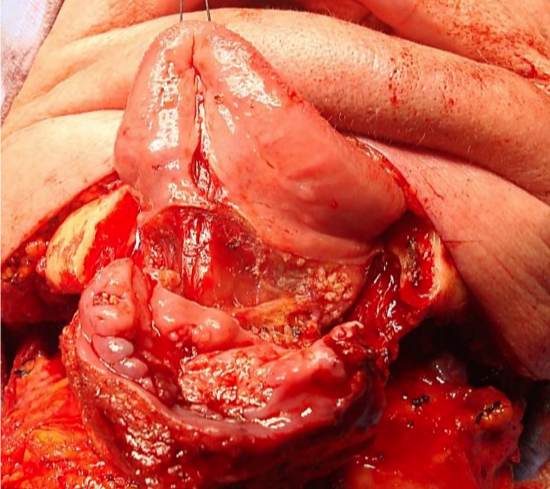
Figure 22: Visor approach with anterior segmental mandibulectomy for cancer of anterior FOM and gingiva
Visor flap: This is achieved by cutting along the gingivolabial and gingivobuccal sulci about 1 cm from the bone to permit placement of sutures when closing the wound, and then stripping the soft tissues from the outer aspect of the mandible. Take care not to transect the mental nerves if they can be saved. The skin flap is then retracted superiorly to expose the mandible (Figure 22).
Pull-through: This may be employed when the tumor stops some distance from the inner aspect of the mandible. Following bilateral neck dissections of Levels 1a and 1b, the mandibular attachments of the anterior bellies of digastric, mylohyoid, geniohyoids and genioglossus are divided with electrocautery working from inferiorly (Figure 12a). The mucosa of the anterior FOM is then divided 1 cm from the inner aspect of the mandible (to facilitate later repair). This permits the surgeon to deliver the anterior FOM and anterior tongue into the neck and then to proceed with the resection.
Tumor Resection
First complete Levels 1a and b of the neck dissection(s) before proceeding to the resection. This permits the surgeon to cut through the muscles in the FOM and tongue knowing the location of the XIIn, lingual nerve and lingual artery.
Mandibulectomy: If a marginal or segmental mandibulectomy is planned then it should be done before resecting the FOM tumor as it improves surgical exposure. If a segmental mandibulectomy is to be done, then preplate the mandible with a reconstruction plate to ensure alignment of the teeth and a good bony contour (Figure 20). Marginal mandibulectomy is done with small sharp osteotomes to avoid inadvertent fracture of the mandible, or with a powered oscillating or reciprocating saw. With marginal mandibulectomy the cut is made obliquely to preserve the height of the outer cortex for mandibular strength, but to remove the inner cortex that abuts the tumor. With segmental mandibulectomy the bone is cut at least 2 cm from visible tumor (Figure 22).
Tumor: Once the mandibulectomy has been completed, keep the bone attached to the tumor specimen and divide the gingival mucosa on the inside of bone cuts; this will release the tumor from bone and facilitate delivery of the tumor into the surgical field. Identify and preserve the submandibular duct(s), and the lingual and hypoglossal nerve(s) if possible. The sublingual gland may be encountered in anterior FOM cancer resections (Figure 19).
Use diathermy to resect the tumor with at least 1 cm margins. Cauterise the ranine vessels during the resection. Larger branches of the lingual artery are ligated as they are encountered. Obtain frozen section confirmation of clear tumor margins if available.
Repair
Following resection, the surgeon carefully assesses the defect to determine how best to restore form and function i.e. mandibular integrity and contour, and oral competence, mastication, oral transport, swallowing and speech.
Reconstruction pointers
- Mobility of the tongue is key to oral function
- It is imperative that reconstructive surgeons follow patients long-term to learn how best to optimize functional flap design
- Tongue length is more important than width
- Never suture the edge of the tongue to the gingiva; in such cases always maintain tongue mobility with a flap
- Avoid tethering the tip of the tongue
- Some defects are best left open to heal by secondary intention to retain mobility
- Simply shaping a flap to match the resected tissue may well restore form but may have poor functional results
- Too broad a flap in the FOM will cause pooling of saliva and adversely affect oral transport
- A marginal mandibulectomy may be done as part of the reconstructive procedure to enable the surgeon to suture the gingivobuccal or gingivolabial mucosa to the FOM mucosa or flap (Figure 20)
- Reduce the risk of orocervical fistula by approximating the mylohyoid to the digastric muscle in the neck, and ensuring that the suction drain is not placed in the upper neck
No repair: Small and/or superficial resections above the mylohyoid that do not communicate with the neck dissection may be left open to heal like a tonsillectomy wound. Resist the temptation to suture such defects as it may alter the shape of the tongue or fix the tongue to the anterior FOM.
Primary closure: Avoid tethering or distorting the tongue. Note the comment about marginal mandibulectomy above (Figure 20).
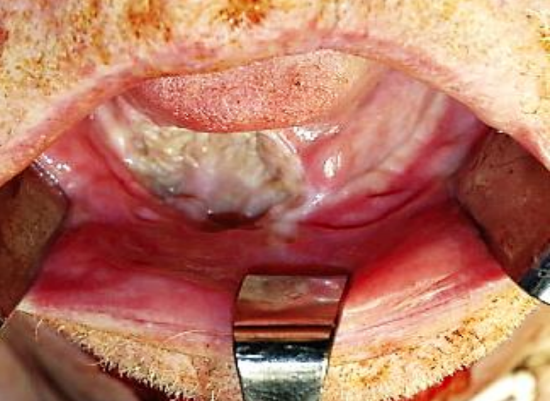
Figure 23: Healed split skin graft over marginal mandibulectomy defect7
Split skin graft: This may be used to cover a defect that could otherwise be left open but for concern about a through-and-through communication to the neck, or over a marginal mandibulectomy defect (Figure 23). The skin is sutured to the margins of the defect with absorbable sutures, the ends of which are left a few centimeters long. A bolster of antisepticimpregnated gauze is placed over the skin graft and tied down with the long sutures. The bolster is removed after about 5 days.
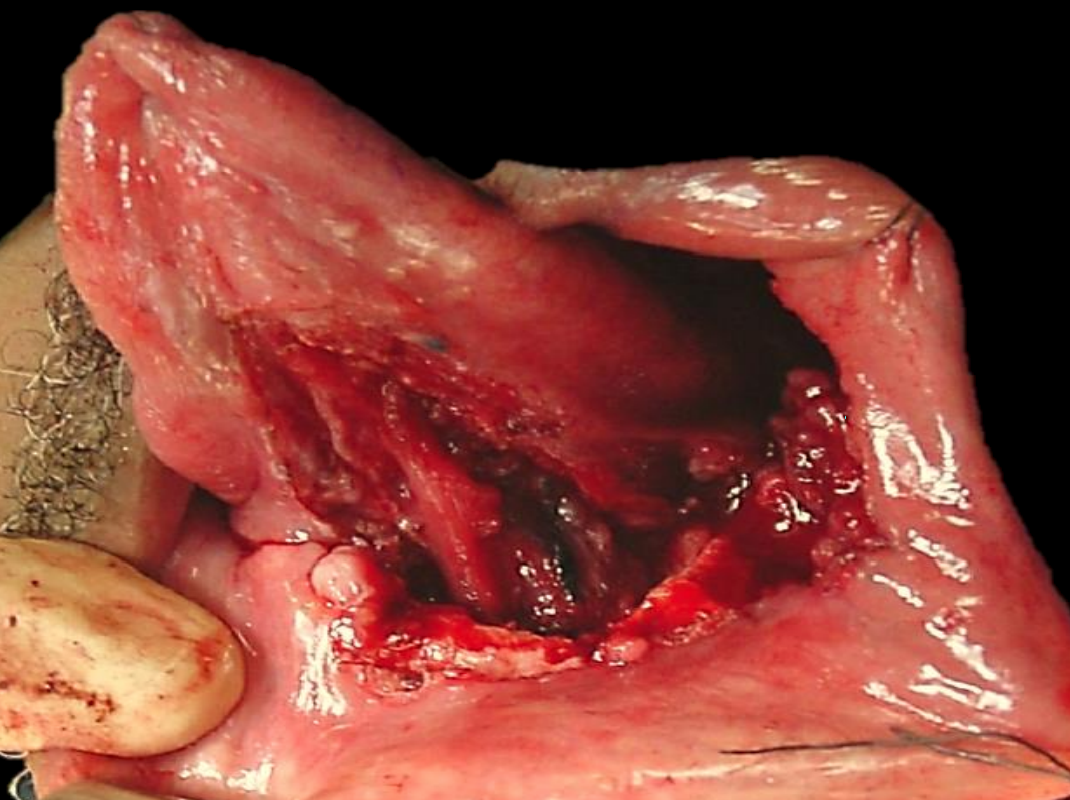
Figure 24a: FOM defect with marginal mandibulectomy
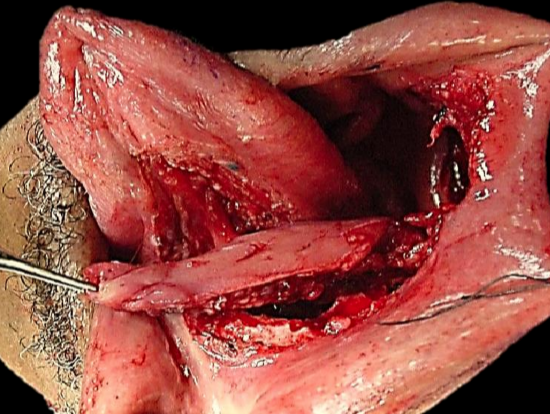
Figure 24b: Buccinator flap based on facial artery
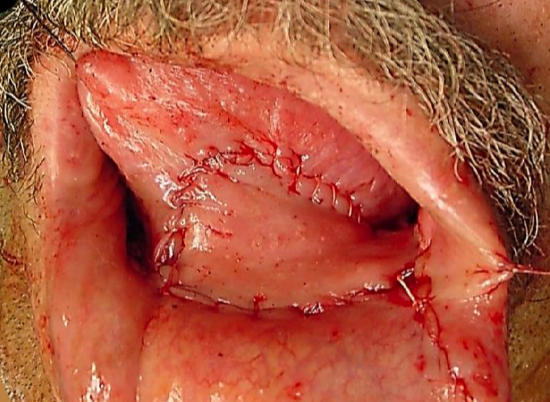
Figure 24c: Buccinator flap for FOM resection with marginal mandibulectomy
Buccinator myomucosal flap: (Figures 24a-c) This is an excellent flap for both anterior and lateral FOM defects as it has the same physical qualities as tissues of the FOM. The pedicle however crosses the mandible and is therefore best suited for edentulous patients, patients with missing teeth, or who have undergone marginal mandibulectomy. (See chapter: Buccinator myomucosal flap)
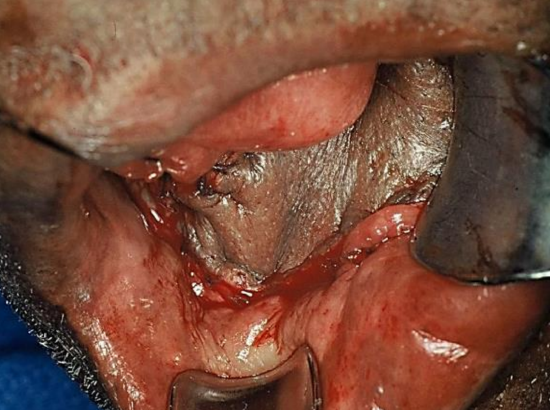
Figure 25: Nasolabial flap inset into lateral FOM
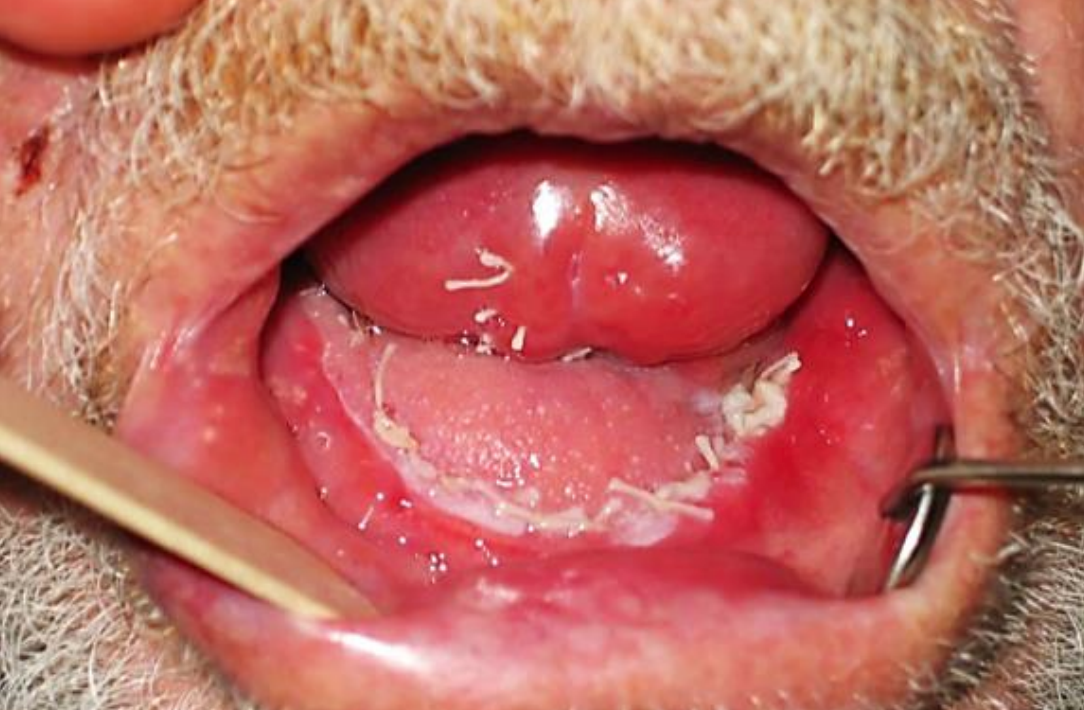
Figure 26: Nasolabial flap reconstruction of anterior FOM defect
Nasolabial flap: The nasolabial flap can be used both for lateral and anterior FOM defects (Figures 25, 26) (See chapter: Nasolabial flap for oral cavity reconstruction)
Submental artery island flap: Though this is an option, concerns exist clearing levels 1a and 1b of the neck (See chapter: Submental artery island flap)
Supraclavicular flap: This has excellent properties for the floor of mouth, being thin and pliable like a radial free forearm flap (See chapter: Supraclavicular flap)
Pectoralis major flap: This flap is only used if other flaps listed are not available. (See chapter: Pectoralis major flap)

Figure 27a: Marginal mandibulectomy
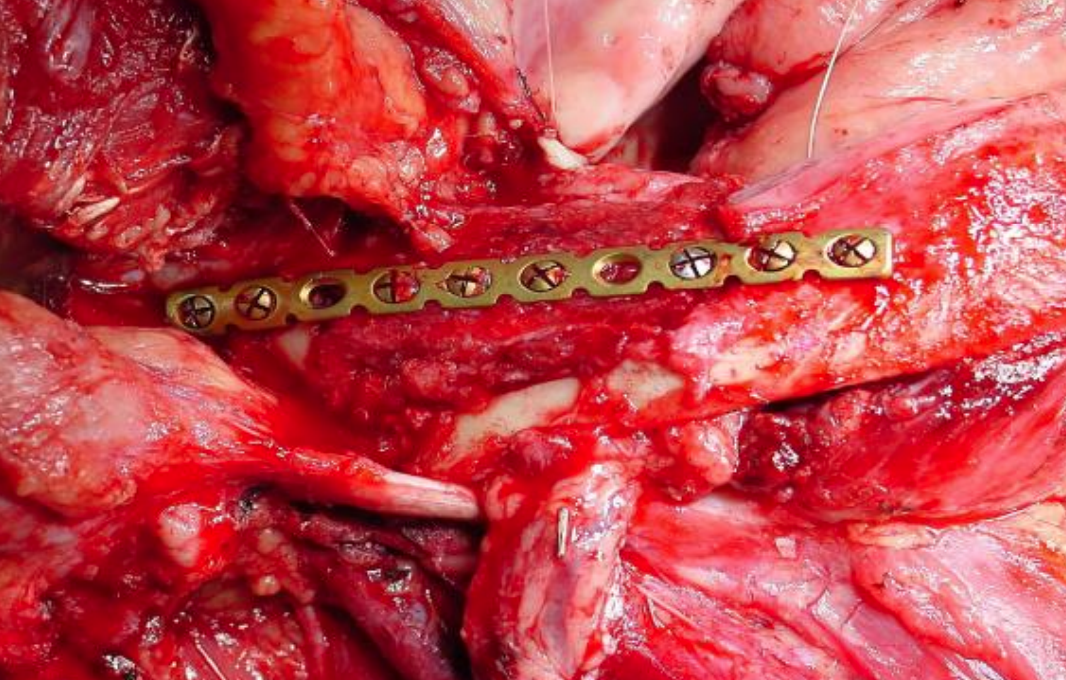
Figure 27b: Onlay radial osseocutaneous flap
Radial free forearm flap: This is a popular choice for FOM repair as the flap is thin and pliable. An osseocutaneous flap can be used as an onlay graft with marginal mandibulectomy defects (Figures 27a, b) (See chapter: Radial free forearm flap)
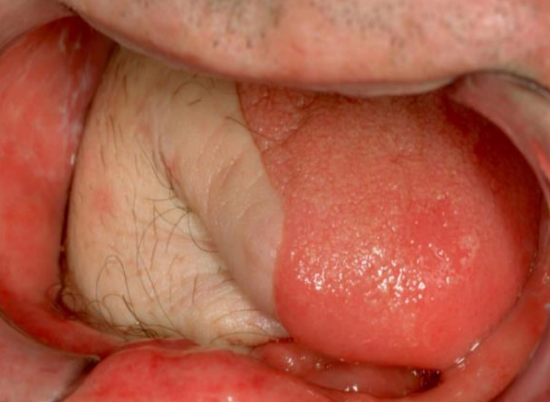
Figure 28: Anterolateral free thigh flap used for lateral FOM and tongue defect
Anterolateral free thigh flap: (Figure 28) Muscle harvested with the flap can be tailored according to the volume of the defect to be filled. However, it is less pliable than the radial free forearm flap and is only suitable for oral reconstruction in patients with thin thighs. (See chapter: Anterolateral free thigh flap)
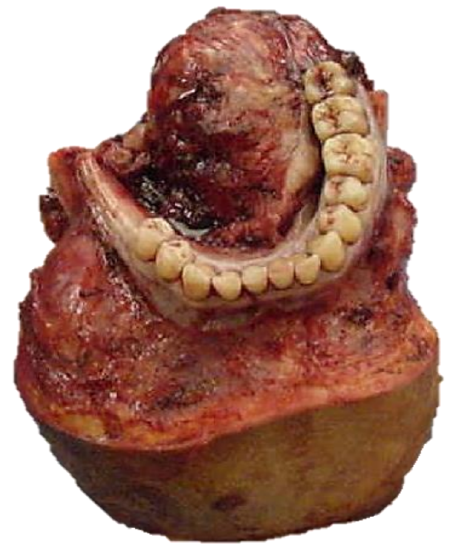
Figure 29a: Resected FOM minor salivary gland tumor
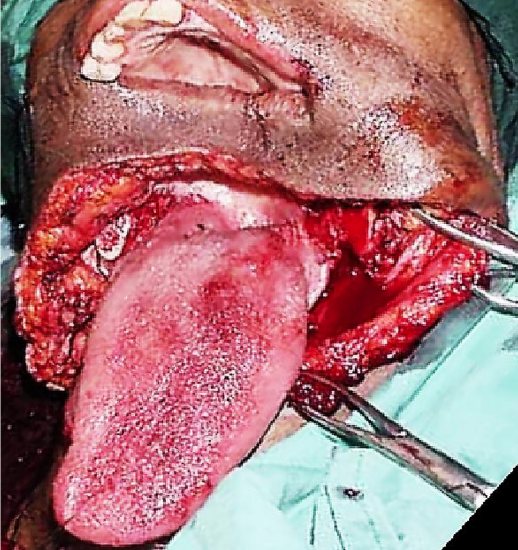
Figure 29b: Note absent mandibular body and FOM
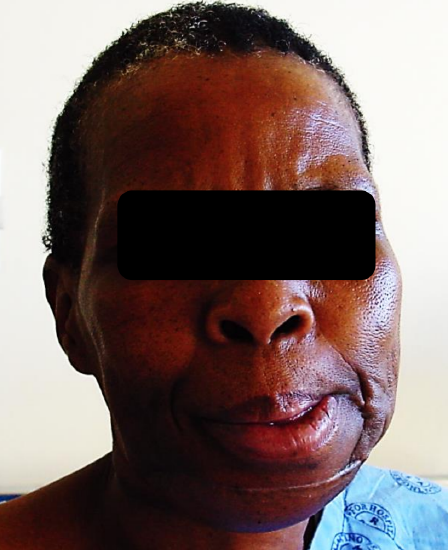
Figure 29c: Mandible reconstructed with free fibula flap
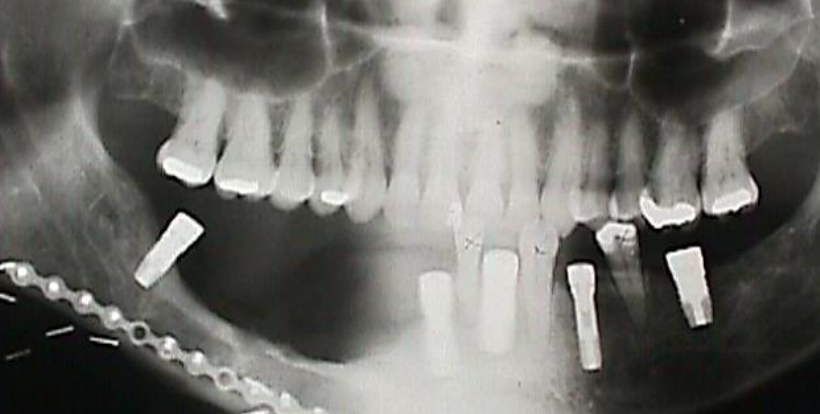
Figure 30a: Dental implants following free fibula flap for segmental mandibulectomy
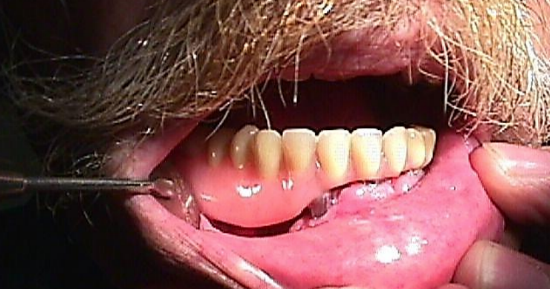
Figure 30b: Denture being slipped into place over implants
Free fibula flap: This is the workhorse of mandible reconstruction following segmental mandibulectomy (Figures 29 a-c). but can also be used as an onlay flap. It is also suitable for dental implants (Figures 30a, b). (See chapter: Free fibula flap)
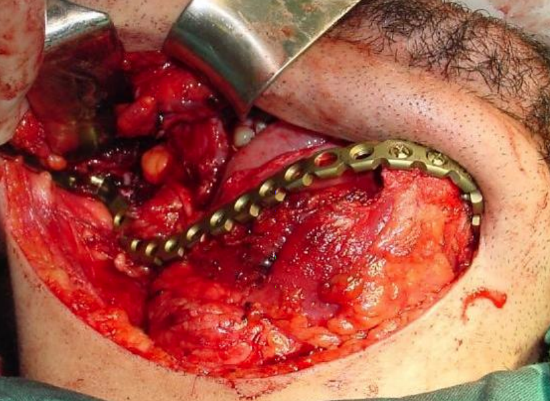
Figure 31: Titanium plate
Titanium reconstruction plate: Although it can be used to replace mandible, it is prone to extrusion both to the inside of the mouth and through skin (Figure 31).
Final comments
Resecting tumors of the FOM is challenging particularly in terms of optimizing oral function. The surgical team must master a wide array of reconstructive techniques to secure the best functional and cosmetic outcomes.
References
- Brown JS, Lowe D, Kalavrezos N, D’Souza J, Magennis P, Woolgar J. Patterns of invasion and routes of tumor entry into the mandible by oral squamous cell carcinoma. Head Neck. 2002;24(4):370-83
- Shaw RJ, Brown JS, Woolgar JA, Lowe D, Rogers SN, Vaughan ED. The influence of the pattern of mandibular invasion on recurrence and survival in oral squamous cell carcinoma. Head Neck J Sci Spec Head Neck. 2004; 26(10):861-9
- Rao LP, Das SR, Mathews A, Naik BR, Chacko E, Pandey M. Mandibular invasion in oral squamous cell carcinoma: investigation by clinical examination and orthopantomogram. Int J Oral Maxillofac Surg. 2004;33(5):454-7
- Acton CHC, Layt C, Gwynne R, Cooke R, Seaton D. Investigative modalities of mandibular invasion by squamous cell carcinoma. Laryngoscope. 2000;110(12):2050-5
- Brown JS, Browne RM. Factors influencing the patterns of invasion of the mandible by oral squamous cell carcinoma. Int J Oral Maxillofac Surg. 1995;24(6):417-26
- Genden EM, Rinaldo A, Jacobson A, et al. Management of mandibular invasion: When is a marginal mandibulectomy appropriate? Oral Oncol. 2005; 41(8):776-82
- Deleyiannis F, Dunklebarger J, Lee E, Gastman B, Lai S, Ferris R, Myers EN, Johnson J. Reconstruction of the marginal mandibulectomy defect: an update. Am J Otolaryngol Head Neck Med Surg. 2007; 28 (6): 363-6
AfHNS Clinical Practice Guidelines for Oral Cancers in Developing Countries and Limited Resource Settings
https://developingworldheadandneckca...e-oral-cavity/
Author
Gerrit Viljoen MBChB
Resident
Division of Otolaryngology
University of Cape Town
Cape Town, South Africa
dr.gerrit.viljoen@gmail.com
Author & Editor
Johan Fagan
MBChB, FCS(ORL), MMed
Professor and Chairman
Division of Otolaryngology
University of Cape Town
Cape Town, South Africa
johannes.fagan@uct.ac.za


