3.12: Treatment of Epistaxis in Children
- Page ID
- 47876
OPEN ACCESS ATLAS OF OTOLARYNGOLOGY, HEAD & NECK OPERATIVE SURGERY
TREATMENT OF EPISTAXIS IN CHILDREN
Simone Hadjisymeou, Nico Jonas
Epistaxis (bleeding from the nose) occurs commonly in children. They usually seek medical attention when it becomes a recurrent problem.
It can be classified by its anatomic location into anterior and posterior epistaxis; anterior epistaxis is far more common.
Etiology
- Idiopathic
- Traumatic: nose picking
- Coagulopathy e.g. hemophilia and von Willebrand’s disease
- Juvenile nasopharyngeal angiofibroma (See chapter: Juvenile nasopharyngeal angiofibroma surgery)
Anatomy
The nose has a rich vascular supply with substantial contributions from arteries originating from both the internal (ICA) and the external (ECA) carotid arteries.
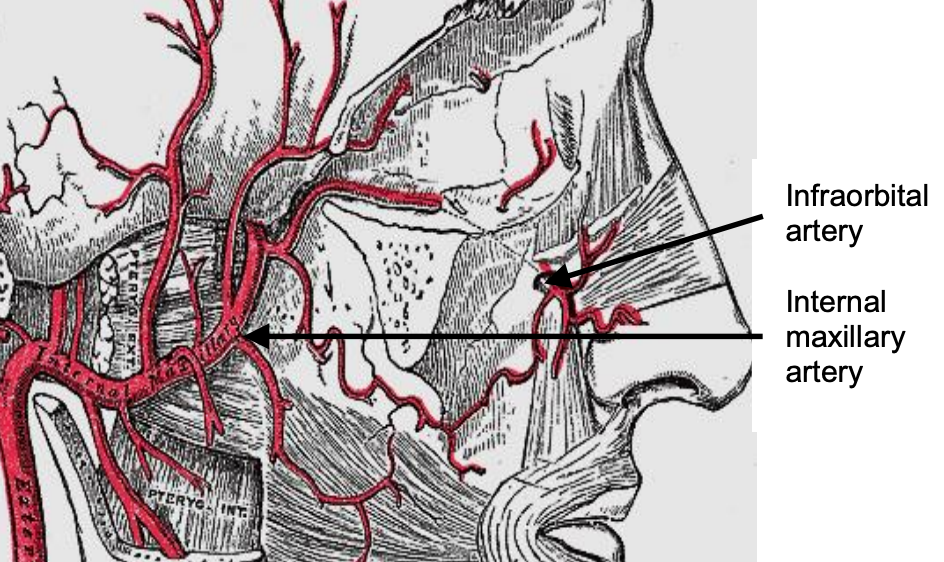
Figure 1: Internal maxillary artery entering pterygopalatine fossa through pterygomaxillary fissure (mandible removed)
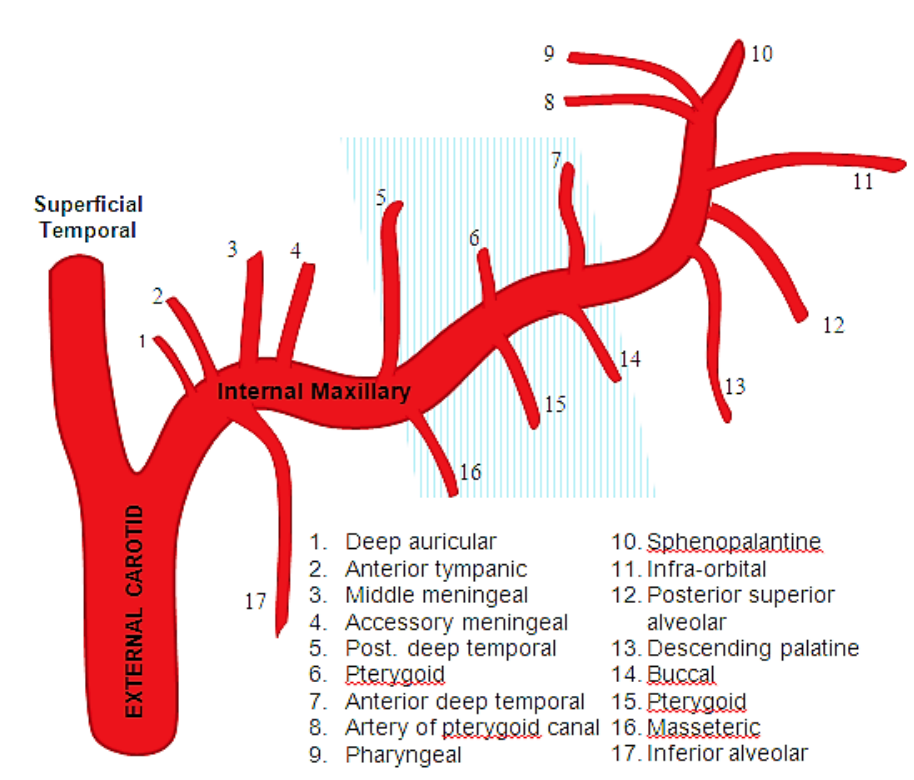
Figure 2: Branches of internal maxillary artery; blue shading denotes 2nd part of internal maxillary artery before it enters pterygopalatine fossa
The ECA system supplies blood to the nose via the facial and internal maxillary arteries. The superior labial artery is a terminal branch of the facial artery and contributes to the blood supply of the anterior nasal floor and anterior septum through its septal branch. The internal maxillary artery enters the pterygopalatine fossa where it divides into 6 branches: posterior superior alveolar, descending palatine, infraorbital, sphenopalatine, pterygoid canal, and pharyngeal (Figures 1 & 2). The descending palatine artery descends through the greater palatine canal and supplies the lateral nasal wall; a branch then returns to the nasal cavity via the incisive foramen to supply the anterior nasal septum. The sphenopalatine artery enters the nose near the posterior attachment of the middle turbinate to supply the lateral nasal wall; a branch also supplies the nasal septum.
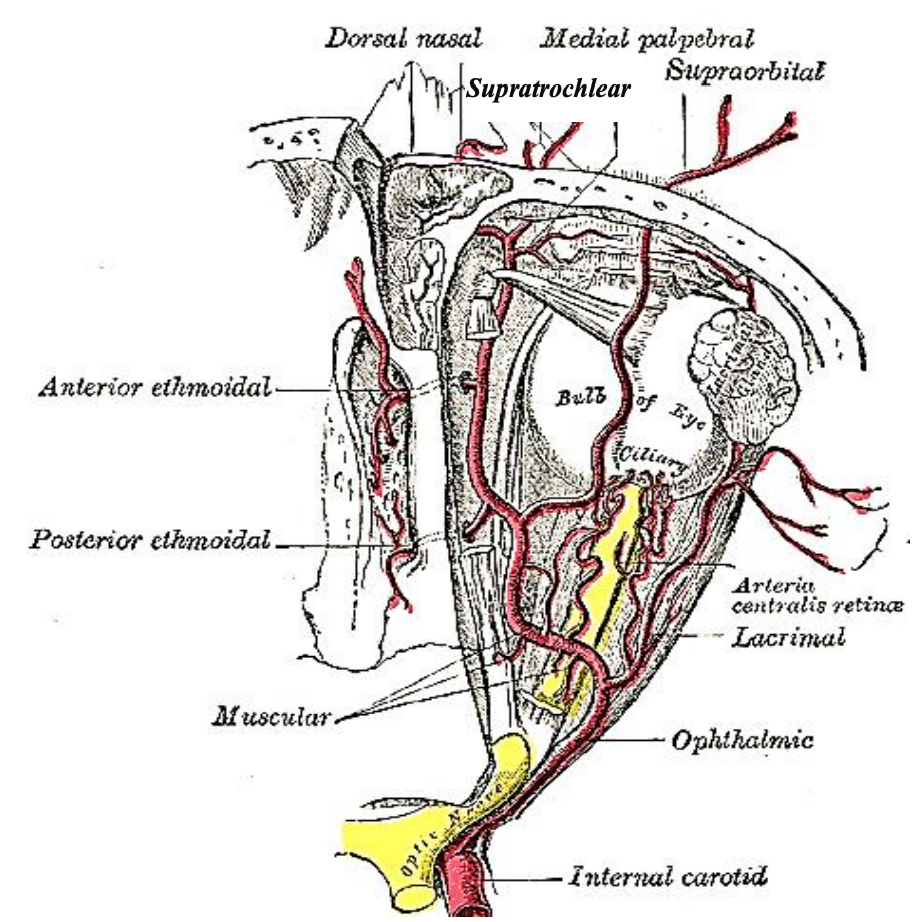
Figure 3: Ophthalmic artery gives rise to anterior and posterior ethmoidal arteries and the supratrochlear and supraorbital arteries
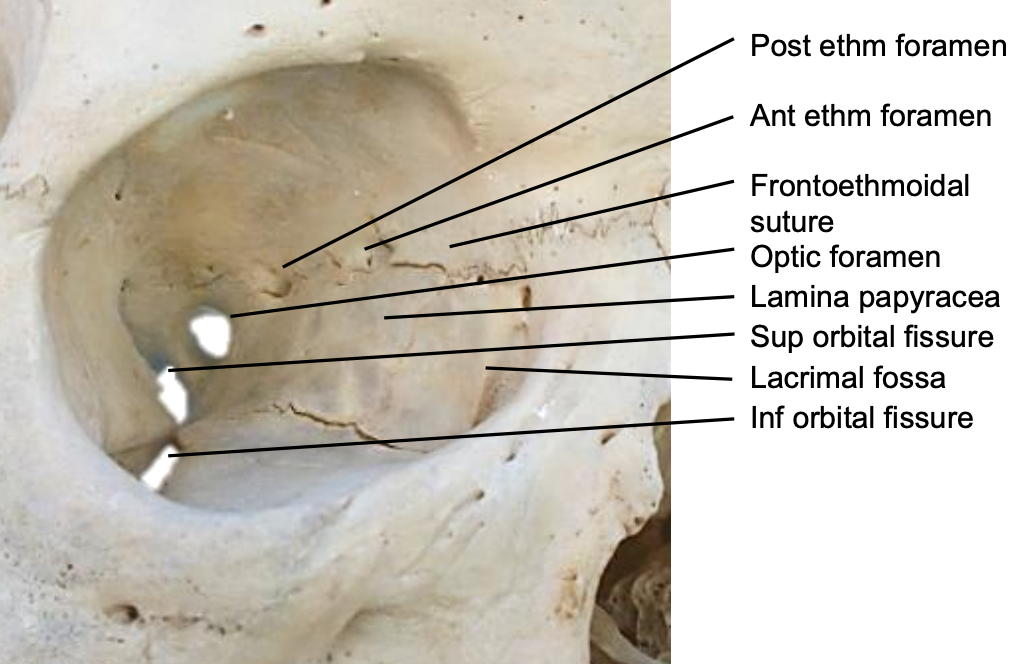
Figure 4: Right medial orbital wall illustrating the anterior and posterior ethmoidal foraminae
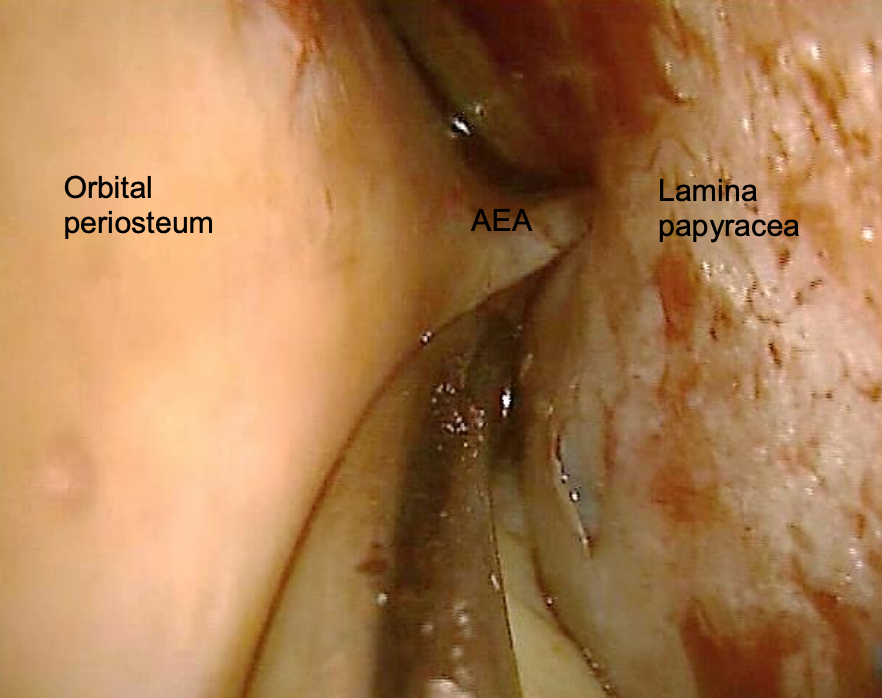
Figure 5: Anterior ethmoidal artery (AEA) exiting anterior ethmoidal foramen at level of frontoethmoidal suture line (right eye)
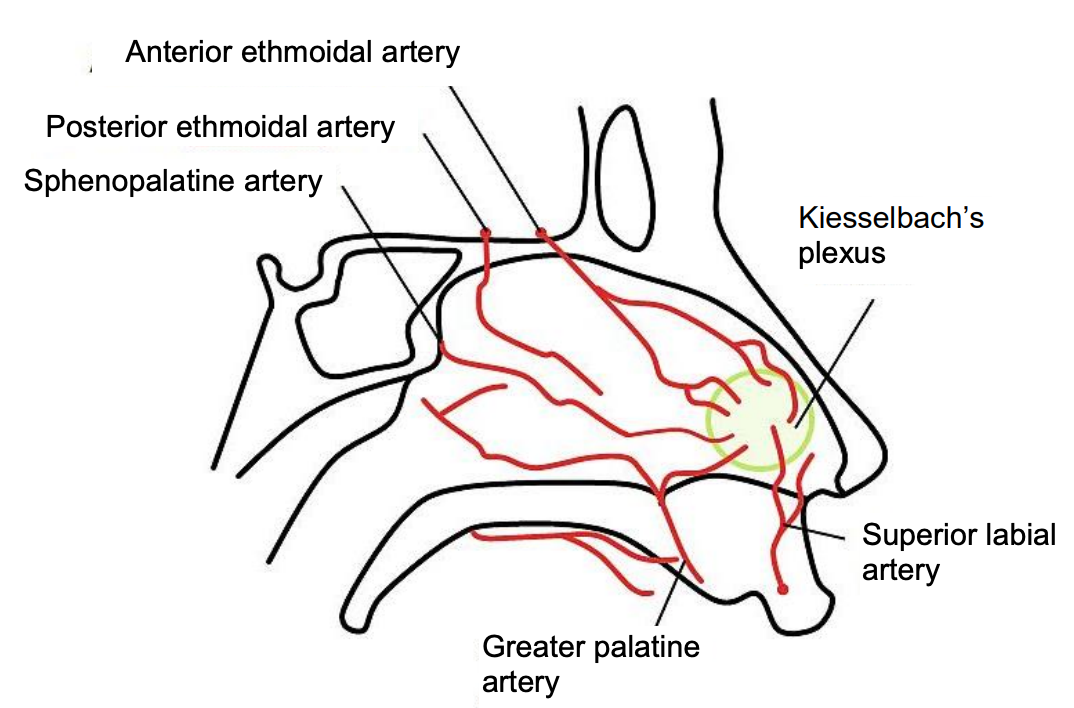
Figure 6: Arterial supply to nasal septum
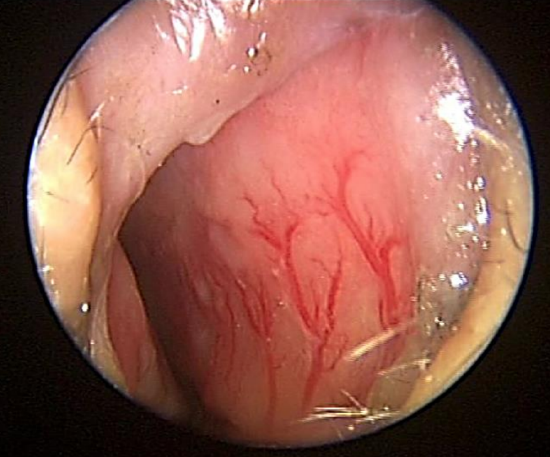
Figure 7: Prominent vessels located on the right anterior cartilaginous septum in Kiesselbach’s plexus/Little’s area
The ICA contributes through the ophthalmic artery which enters the bony orbit via the superior orbital fissure and divides into several branches (Figure 3). The posterior ethmoid artery exits the orbit through the posterior ethmoid foramen, located 2-9 mm anterior to the optic canal; the larger anterior ethmoid artery leaves the orbit through the anterior ethmoid foramen (Figures 4, 5). The anterior and posterior ethmoidal arteries cross the ethmoid roof to enter the anterior cranial fossa and then descend into the nasal cavity through the cribriform plate where they divide into lateral and septal branches to supply the lateral nasal wall and nasal septum (Figure 3). Kiesselbach’s plexus or Little’s area is located on the anterior cartilaginous nasal septum; it is an anastomotic network of vessels originating from both the ICA and the ECA (Figures 6, 7).
Assessment
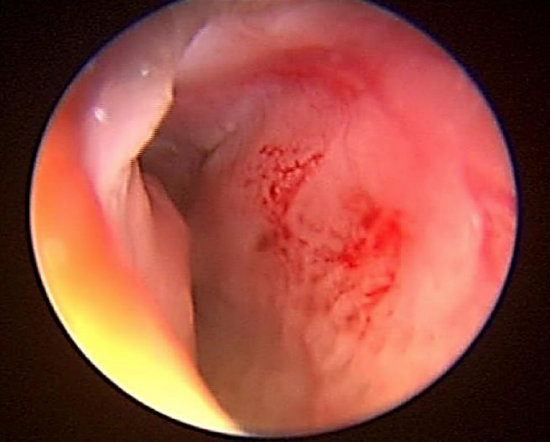
Figure 8: Ulceration of the right anterior nasal septum causing epistaxis
Anterior rhinoscopy usually reveals a bleeding point, prominent anterior septal vessels or ulceration (Figure 8). If a likely source of bleeding is not apparent, then nasendoscopy should be performed to exclude a posterior cause for the epistaxis.
First aid/Advice
While the nose is bleeding, sit the patient upright with the neck flexed and head forward. Pinch the anterior (soft) part of the nose between thumb and index finger for a few minutes to apply pressure to vessels in Little’s area. Apply an ice pack to the forehead, to the bridge of the nose, or place a block of ice in the mouth to promote vasoconstriction and hence reduce blood flow to the nose1, 2. These measures stop most acute nosebleeds.
Cautery (chemical or electrocautery)
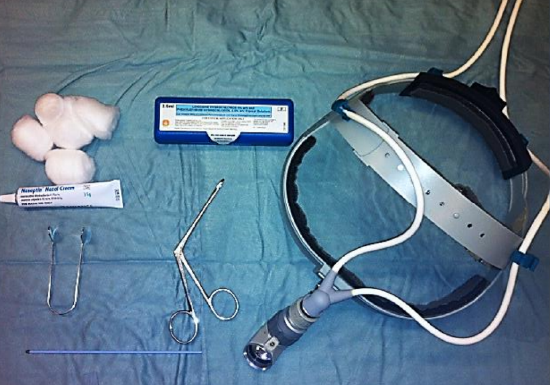
Figure 9: Headlight, local anesthetic / decongestant preparation, Thudicum nasal speculum, dressing forceps, cotton wool, silver nitrate cautery stick and antibiotic ointment
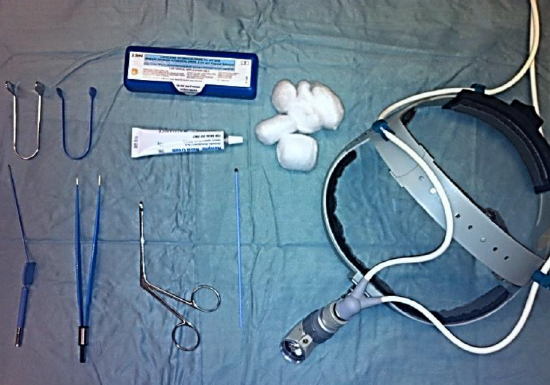
Figure 10: Headlight, local anesthetic / decongestant preparation, insulated and non-insulated nasal specula, insulated bipolar forceps, insulated monopolar needle, dressing forceps, cotton wool and antibiotic ointment
Anterior epistaxis originating from prominent vessels on the anterior septum can usually be controlled by applying antiseptic cream to the nasal vestibule, or by cauterization. The basic equipment required for nasal cautery with silver nitrate and for monopolar or bipolar electrocautery is outlined in Figures 9 & 10.
Chemical cautery

Figure 11: Silver nitrate sticks
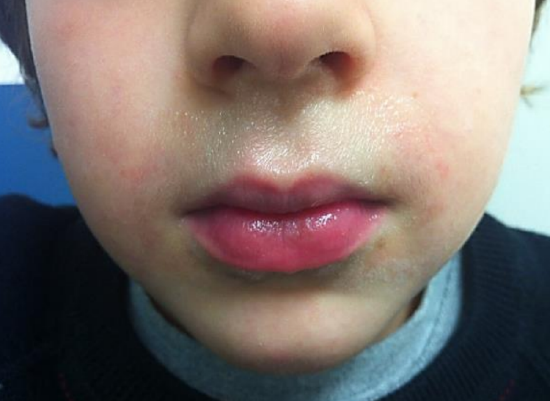
Figure 12: Paraffin jelly has been applied to the upper lip to avoid a chemical burn and staining of the skin by silver nitrate
A cotton wool ball is soaked in a decongestant and local anesthetic and is placed in the anterior nasal cavity for 10 minutes. Cautery is done with a silver nitrate stick (Figure 11). When silver nitrate comes into contact with a wet surface it forms nitric acid which causes a chemical burn. Paraffin jelly is applied to the upper lip to prevent a chemical burn and staining from silver nitrate mixed with nasal mucus spills onto the upper lip (Figure 12).
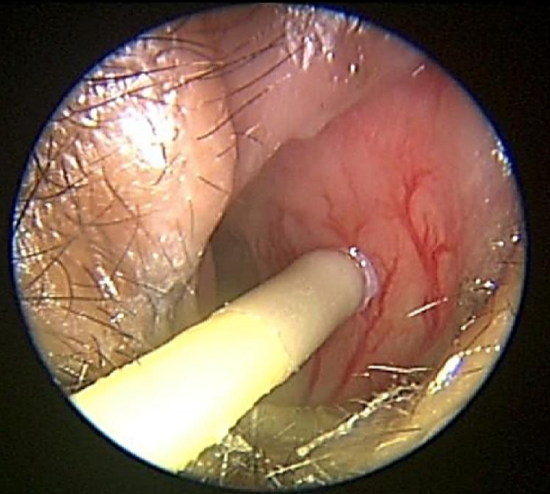
Figure 13: Cautery with silver nitrate to an anterior septal vessel
The patient is asked to blow and clear the nose. Initially cauterize the area surrounding the bleeding point to shrink vessels that supply it; then cauterize the bleeding point (Figure 13). Apply silver nitrate for 5-10 seconds to each location to avoid a deep burn which may cause necrosis and a septal perforation. The parent / caregiver is instructed to apply antibiotic cream to the cauterized area for a week following cauterization.
Electrocautery (Figure 14)
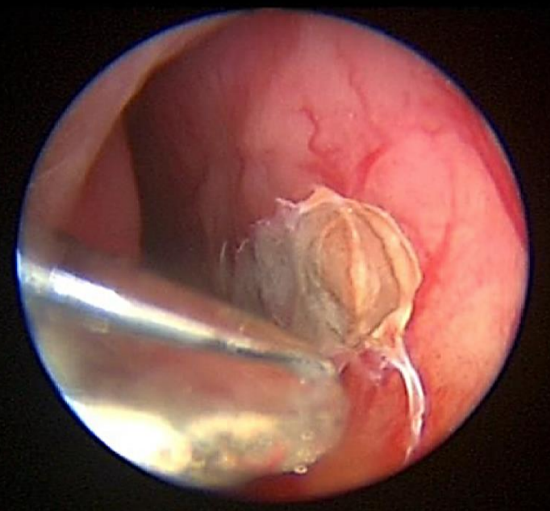
Figure 14: Cautery to anterior septal vessels using bipolar forceps
Bipolar or monopolar cautery may be used when chemical cautery fails to control anterior epistaxis on at least two occasions. It is generally done under general anesthesia in children.
Complications of cautery
These include nasal adhesions and septal perforation. Avoid cautery to opposing areas of the septum at the same sitting to reduce the chance of a septal perforation occurring. A chemical burn presenting as discoloration of the skin of the nasal vestibule and upper lip can persist for up to a week; hence the need to protect the skin by applying Vaseline to the upper lip.
Surgical intervention
This is considered if the former interventions fail to control anterior epistaxis, or with posterior epistaxis.
Examination of the nose under general anesthesia and electrocautery
- Prepare the nose with a topical decongestant and local anesthetic
- Examine the nose with a 4 mm Hopkin’s rod or if not available, with a headlight and Killian’s speculum
- Cauterize bleeding points with either an insulated monopolar needle or with bipolar forceps
- Occasionally cauterization cannot be performed or is unsuccessful and nasal packing must be inserted to control bleeding
Sphenopalatine artery ligation
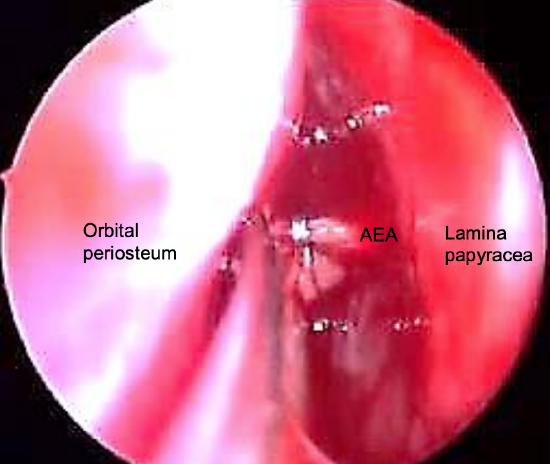
Figure 15: Liga clips being applied to the anterior ethmoidal artery (AEA)
If electrocautery fails to control the bleeding, the next step to consider is endoscopic sphenopalatine artery ligation. An incision is made in the middle meatal wall just anterior to the posterior attachment of the middle turbinate. A mucosal flap is carefully elevated to identify the ethmoidal crest. The sphenopalatine artery is seen to enter the nasal cavity just posterior to the crest. The artery is clipped or cauterized using sphenopalatine bipolar forceps (Figure 15).
Other arterial ligations
Ethmoidal, internal maxillary or external carotid artery ligation is very rarely required in children (Figure 15). Internal maxillary and external carotid artery ligation have the added disadvantage that it precludes subsequent embolization should it be required.
Embolisation
Bleeding from the ECA system may be controlled with embolisation, either as a primary modality or when surgery has failed to control bleeding. Angiography is performed to check for the presence of unsafe communications between the ICA and ECA systems. Selective embolization of the internal maxillary artery and sometimes the facial artery may be performed. The most common reason for failure is continued bleeding from the ethmoid arteries.
Postoperative care
Employ measures to promote healing of the cauterized areas e.g. saline nasal spray and antiseptic nasal ointment.
Patient education
The following measures are advised during the 1st few days following cauterization
- Saline nasal spray
- Antiseptic nasal ointment
- Avoid blowing the nose hard
- Avoid digital nasal manipulation/picking the nose
- Avoid aspirin and non-steroidal anti-inflammatories (NSAIDs)
References
- Porter MJ. A comparison between the effect of ice packs on the forehead and ice cubes in the mouth on nasal submucosal temperature. Rhinology. 1991; 29(1):11-5
- Scheibe M, Wüstenberg EG, Hüttenbrink KB, Zahnert T, Hummel T. Studies on the effects of ice collars on nasal blood volume using optical rhinometry. Am J Rhinol. 2006; 20(4): 394-6
Authors
Simone Hadjisymeou MBBCh, BSc, MRCS (ENT)
ENT Registrar
Guys and St Thomas’ Hospital
London, United Kingdom
simonehadji@gmail.com
Author and Pediatric Section Editor
Nico Jonas MBChB, FCORL, MMed
Pediatric ENT Consultant
Addenbrookes Hospital
Cambridge University Hospital NHS Foundation Trust
Cambridge, United Kingdom
nicojonas@gmail.com
Editor
Johan Fagan MBChB, FCS(ORL), MMed
Professor and Chairman
Division of Otolaryngology
University of Cape Town
Cape Town, South Africa
johannes.fagan@uct.ac.za


