4.17: Endoscopic Resection of Intraconal Tumors- Surgical Anatomy and Techniques
- Page ID
- 51276
OPEN ACCESS ATLAS OF OTOLARYNGOLOGY, HEAD & NECK OPERATIVE SURGERY
ENDOSCOPIC APPROACH TO ORBITAL CAVERNOUS HEMANGIOMA (OCH) AND INTRACONAL TUMORS: SURGICAL ANATOMY AND TECHNIQUES
Catherine Banks, Benjamin Bleier
The position of intraconal tumors relative to the optic nerve dictates the choice of surgical approach. Tumors with their epicenter medial to the optic nerve or “below a plane of resectability” (POR), which represents a plane subtended by the contralateral nostril and the long axis of the optic nerve, are amenable to an endoscopic approach. The feasibility and safety of this approach has been demonstrated in the literature. Tumors located lateral and superior to the POR are not candidates for an exclusively endoscopic resection.
The composition of the surgical team varies depending on the institution. However, a multidisciplinary team including an otolaryngologist, oculoplastic surgeon and in some cases, a neurosurgeon, is invaluable.
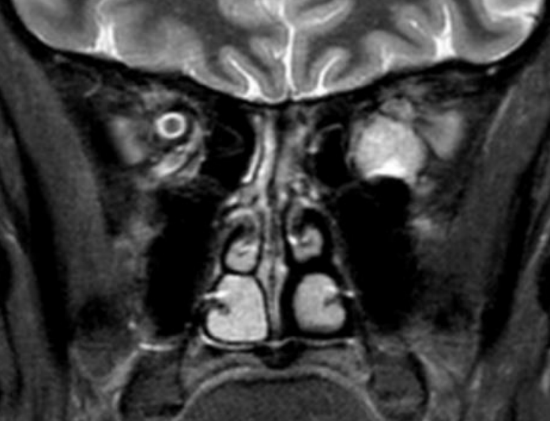
Figure 1: Coronal T2 weighted MRI scan demonstrating a left OCH
The endoscopic approach to intraconal tumors will be described in this chapter using orbital cavernous hemangioma (OCH) as an example, as is it the most common primary orbital tumor in adults, with a reported incidence of 5-15% of all orbital tumors (Figure 1).
Orbital cavernous hemangiomas (OCH)
The International Society for the Study of Vascular Anomalies classifies OCHs as slow-flow cavernous venous malformations. OCHs are more common in women and occur in the 4th and 5th decades of life1. The natural history remains elusive. Progesterone may play a role in the clinical course which may explain sudden growth of OCH seen during pregnancy and a reduction in size or stabilization in postmenopausal women; however, the exact role of progesterone is yet to be fully elucidated.
A significant number of OCHs present as asymptomatic lesions incidentally found on CT or MRI performed for unrelated reasons. Previous studies have shown that asymptomatic lesions often show no progression2,3. In many cases if an incidental OCH does not change over several years, it is unlikely to do so over more prolonged periods of followup.4
The majority are located between the optic nerve and the extraocular muscles and are therefore intraconal. It is well documented that OCH have a predilection for the intraconal space. The single most common anatomical location is lateral to the optic nerve, which may reflect the relationship between the optic nerve and the distribution of the ophthalmic vasculature. However, OCH can be found throughout the orbit, including the medial intraconal space, extraconal space, and within the optic canal.5 Rarely, they extend beyond the confines of the orbit into the pterygopalatine fossa,6 cavernous sinus,7 and intracranial space.8
Histologically OCHs appear as cavernous venous malformations and have exclusively venous vasculature. Despite suggestion of arterial flow on imaging studies, histologically there is no evidence of the elastic lamina associated with arterioles. The thicker walled vessels are thought to be the result of thrombosis and recanalization. If any arterial component is present, it is thought to be inconsequential both histologically and clinically.9 OCHs are characterized as lesions with mature cellular components and do not tend toward dysplasia or hypercellularity. Although they can incorporate surrounding vessels and nerves, they do not typically infiltrate into surrounding tissue.10 OCHs tend to be slow growing vascular lesions with a radiological growth rate of 10-15% per year.10 The growth of OCH is thought to be a cycle of stasis and thrombosis with endothelial cellular proliferation and recanalization into multiple clefts and vascular channels.
They have a firm fibrous capsule that makes them amendable to endoscopic resection. Surgical resection is indicated for symptomatic lesions whilst smaller asymptomatic lesions can be observed. The goal of surgery is definitive resection. However, given its benign nature, complete resection must be balanced against iatrogenic morbidity. Partial resection of intraconal OCH has been reported, but long term outcomes remain unknown.14
Surgical approach
The position of intraconal OCHs within the orbit relative to the optic nerve dictates the choice of approach. OCHs with an epicenter medial to the optic nerve or “below a plane of resectability” (POR), which represents a plane subtended by the contralateral nostril and the long axis of the optic nerve, are amenable to an endoscopic approach.11 The feasibility and safety of this approach has been consistently demonstrated.10,12-20 OCHs located lateral and superior to the POR are not candidates for an exclusively endoscopic resection.21
Surgical anatomy
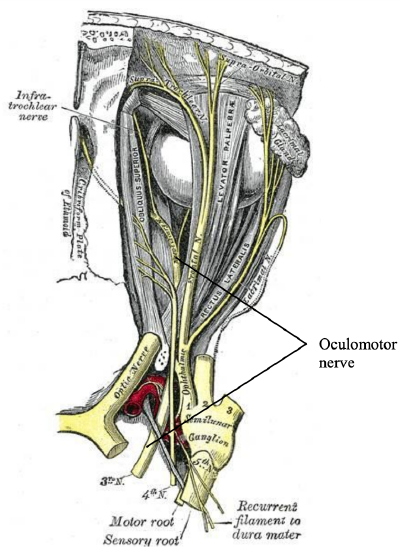
Figure 2: A branch of the oculomotor nerve penetrates the medial rectus one-third of the distance from the annulus of Zinn to its insertion onto the globe
Knowledge of the neurovascular anatomy within the orbit is crucial and directs safe placement of a ball probe on the medial rectus and the degree of retraction. A branch of the oculomotor nerve penetrates the medial rectus one-third of the distance from the annulus of Zinn to its insertion onto the globe (Figure 2); therefore, direct or prolonged traction here should be avoided.22
Operating room set-up
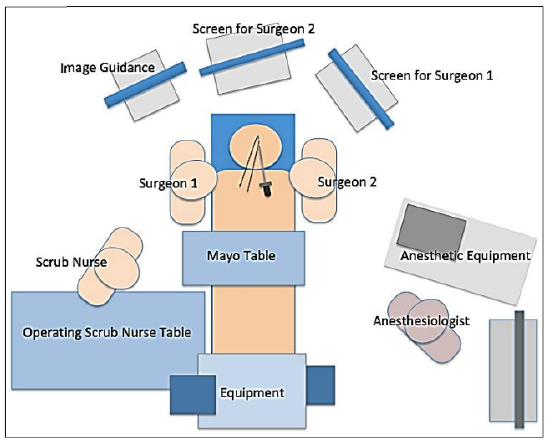
Figure 3: The operating room setup for a binarial four-handed 2 surgeon transseptal approach. The endoscope is held by surgeon 2 (in this case for a left-sided tumor; the surgeons can be reversed for right-sided tumors). Surgeon 1 performs the dissection and retraction
Figure 3 demonstrates a typical layout of the operating room. This allows for a binarial, four-handed transept approach that is commonly used for intraconal OCH resection. With a binarial approach, a posterior septectomy or septal window is required. This has minimal morbidity, allows maneuvering of endoscopes and other instruments, and may be integrated into elevation of a nasoseptal flap for medial wall reconstruction.20
Essential instrumentation (Figure 4)

Figure 4: (A) Penfield dissector number 4; (B) Curved olive tip suction; (C) Frazier suction cannula; (D) Lusk ball tip probe
- Rigid endoscopes:
- 0-degree
- Angulated 30, 45 and 70-degree
- Pledgets soaked in saline
- Suction cannulae
- Frazier size 9 and 10
- Curved olive tip suction
- Medial rectus retractor
- Penfield dissector Number 4
- Lusk ball tip probe
Surgical Steps
1. Anesthesia and positioning
The patient is placed supine in a head ring with the head of the bed raised 15-30 degrees. Total intravenous anesthesia is used. The patient is intubated with the tube positioned to the left. The room is set up as seen in Figure 3.
2. Preparation of nasal cavity
The nasal cavity is prepared with cottonoid pledgets soaked in 1:1000 adrenaline. This is then changed to saline soaked pledgets once the periorbita are exposed. The nasal cavity is infiltrated with 10 mL of lignocaine 1% with 1:100,000 adrenaline.
3. Create a working space and define orbital wall and orbital axis
This is a fundamental part of the surgery and requires the following
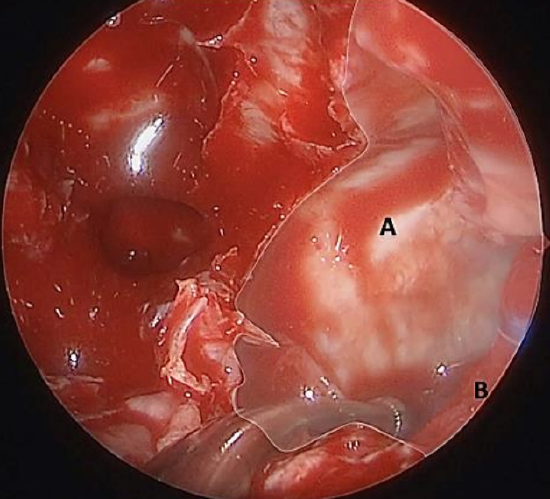
Figure 5: Placement of cottonoid to retract left posterior maxillary mucosa. (A) marks the maxillary sinus with the mucosa elevated to expose the bone of the posterior maxillary wall; (B) indicates the cottonoid placed between the mucosa and posterior maxillary wall to assist with the dissection and securing and protecting the mucosa
- Complete uncinectomy
- Wide maxillary antrostomy
- Sphenoethmoidectomy
- The middle turbinate may be resected to improve access and visibility
- Reflect the mucosa from medial to lateral off the posterior wall of the maxillary antrum
- Use the Lusk ball probe to assist with removal of the maxillary sinus mucosa and place a pledget to hold the mucosa out of the operating field (Figure 5)
4. Posterior septectomy & nasoseptal flap
- The need for a posterior septectomy vs. a septal window with reconstruction is based on the tumor location and extent of the dissection required
- Raise the nasoseptal flap on the contralateral side of the nose
- Tailor the size of the flap to the individual case
- Begin the incision inferiorly, starting at the anterior inferior choana and sweeping anteriorly and inferiorly towards the floor of the nose
- Carry this incision anteriorly along the floor of the nose, and tailor it to the anticipated defect of the exposed lamina papyracea
- Raise the anterior flap by blunt dissection with a Cottle elevator to the sphenoid ostium
- Make the superior incision with cold steel dissection
- Tuck the flap into the nasopharynx until completion of the resection
- The septectomy is then made posteriorly in a location that will permit unrestricted passage of instruments
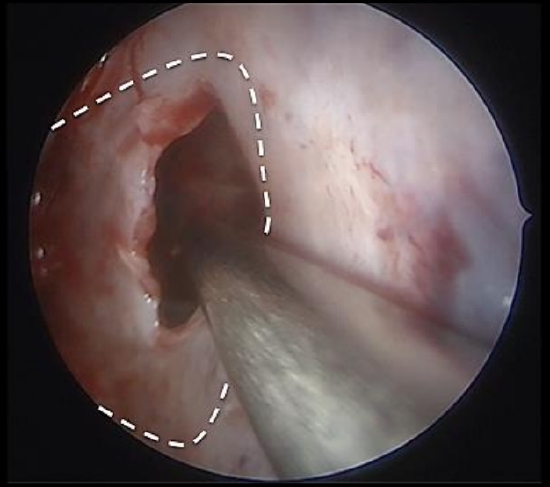
Figure 6: Nasal septal flap (outlined) and posterior septectomy viewed from right (contralateral) nose
5. Posterior maxillary wall and orbital process of palatine bone
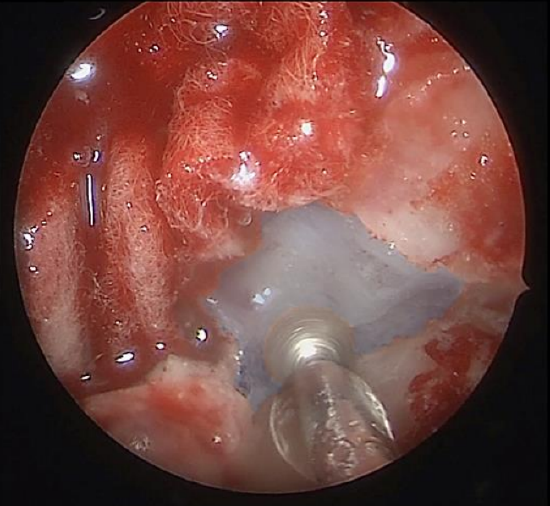
Figure 7: Posterior medial maxillary wall over the orbital process of the palatine bone is drilled with 15-degree diamond burr. (Blue - highlights the location of the orbital process of the palatine bone)
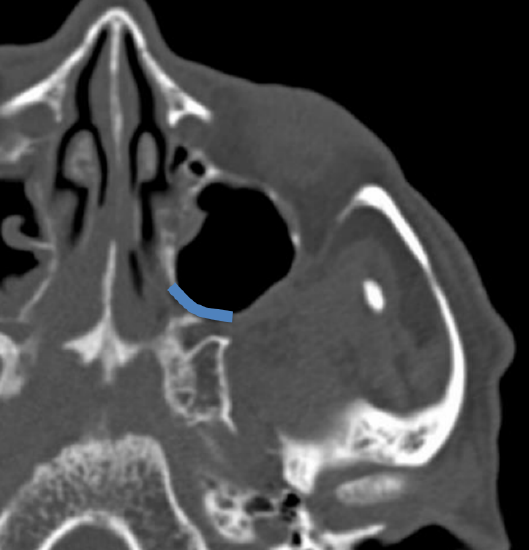
Figure 8: Axial CT scan demonstrating where bone is removed from the posterior medial maxillary wall and the orbital process of the palatine bone is thinned
- The ipsilateral posterior maxillary wall and the orbital process of the palatine bone is then thinned using a 15-degree diamond burr (Figures 7, 8)
- Removing the orbital process provides an additional 0.36+/-0.42cm3 of exposure in the inferolateral vector.23
6. Exposing periorbita (Figures 9, 10)
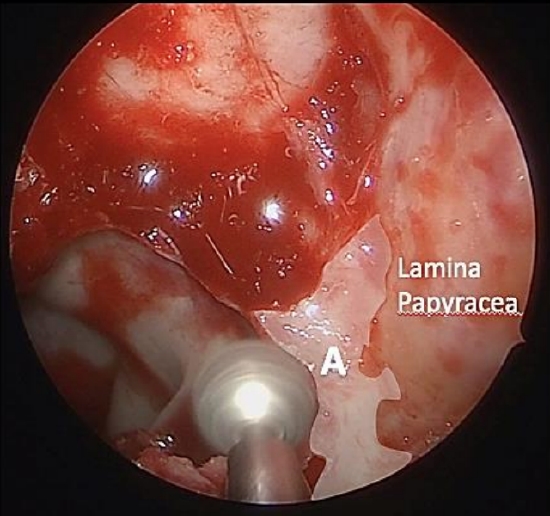
Figure 9: Lamina papyracea removal and drilling of the thick optic strut
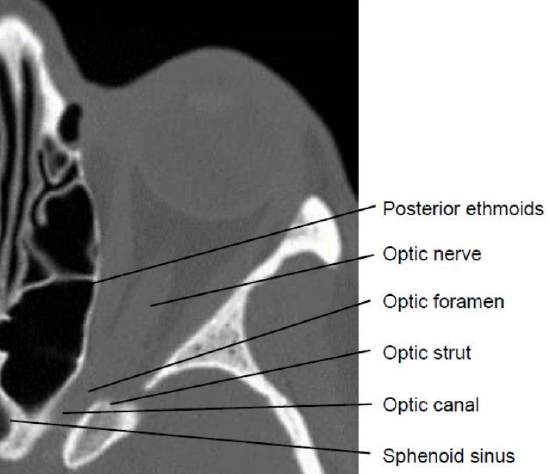
Figure 10: CT scan illustrating lamina papyracea and the thick optic strut
- Remove the lamina papyracea with blunt dissection to expose the underlying periorbita
- If necessary, the optic strut may be drilled to facilitate safe removal of the bone overlying the optic nerve (Figure 10)
7. Retraction of medial rectus
- Create a periorbital window to identify the extraconal fat and muscles
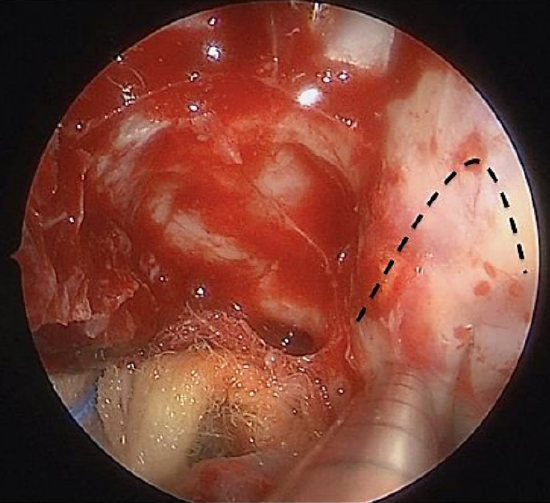
Figure 11: Incision line of the periorbita in a reverse hockey stick fashion. This can be performed with a sickle knife
- Incise the periorbita in a reverse hockey stick fashion using a sickle knife (Figure 11)
- Place the incision just anterior to the border of the tumor to prevent unnecessary fat prolapse into the anterior surgical field
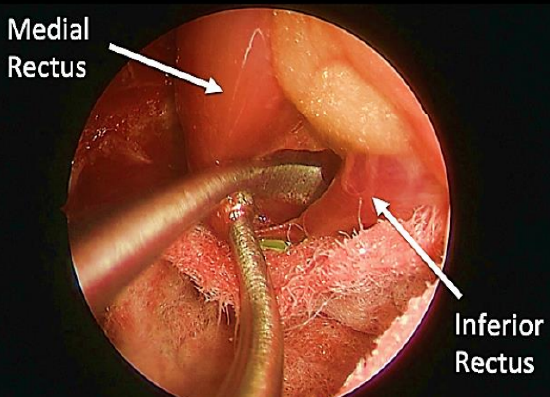
Figure 12: Medial rectus as a landmark of the medial orbit retracted to enter the intraconal space
- The intraconal dissection corridor is bordered by the medial rectus muscle above and the inferior rectus muscle below (Figure 12)
- Use a dissector e.g. Penfield or Lusk probe to bluntly dissect between the medial and inferior rectus muscles and to visualize the intraconal space
- The medial rectus serves as a landmark of the medial orbit and must be retracted to access intraconal OCHs
- Retract the medial rectus with care using a right-angled instrument under the inferior border of the muscle and gently and dynamically retract the muscle in a superomedial direction to prevent postoperative diplopia
8. Hemostasis
Judicious and precise use of bipolar cautery, saline soaked cottonoid pledgets, and warm water irrigation are relatively safe. Avoid monopolar cautery within the orbit due to the significant risk of thermal injury to important neural structures.
9. Orbital fat
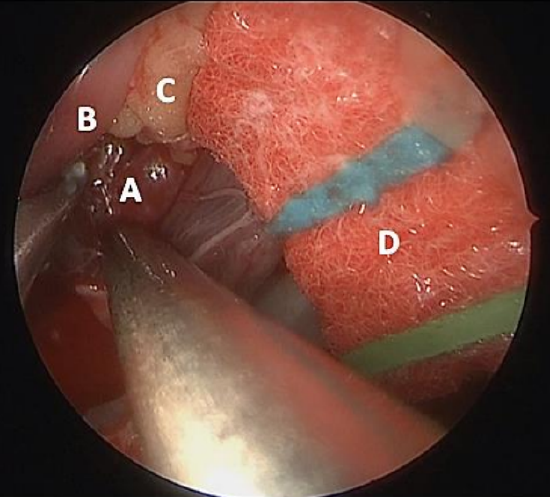
Figure 13: Cottonoid pledgets used to retract the intraconal fat. (A) Intraconal hemangioma; (B) Medial rectus; (C) Intraconal orbital fat; (D) Cottonoid pledget retracting orbital fat
Fat herniation can be troublesome and keeping the intraconal dissection corridor open is challenging. Preserving extraconal fat not only helps to preserve orbital volume, but also minimizes the risk of medial rectus scarring and entrapment. Proper placement of the periorbital incision as previously described and the use of cottonoid pledgets to separate and retract the orbital fat can assist with visualization and should avert the need to remove extraconal fat (Figure 13).
10. Resection techniques
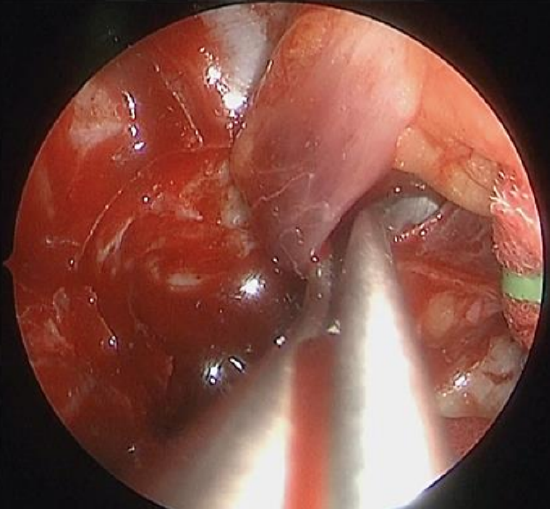
Figure 14: Blunt dissection is employed using the Lusk ball probe to retract the medial rectus and the Penfield number 4 dissector to resect the OCH
- The fibrous capsule permits dissection in an extracapsular plane with preservation of the capsule
- A process of gentle traction, use of cottonoids, and blunt dissection is used
- Sharp cutting instruments have also been used but do increase the risk of bleeding from inadvertent arteriolar injury
- Incomplete resection has been documented in cases where the lesion is adherent to the optic nerve, although long-term follow-up results are unknown 14
11. Reconstructing medial orbital wall
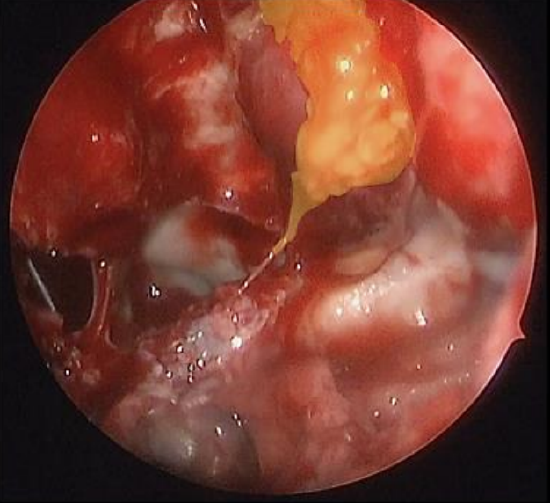
Figure 15: Extraconal orbital fat placed over the medial rectus muscle
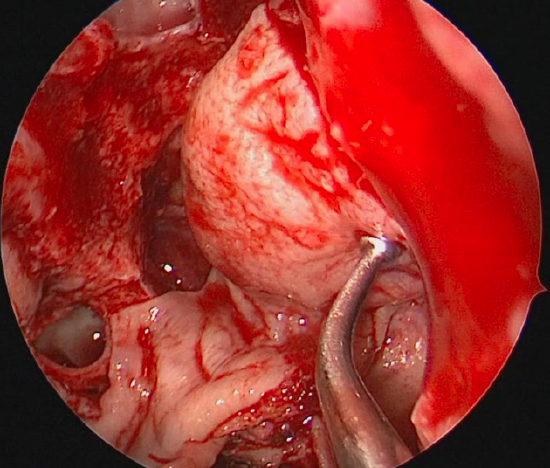
Figure 16: The pedicled nasoseptal flap draped along the medial orbital wall
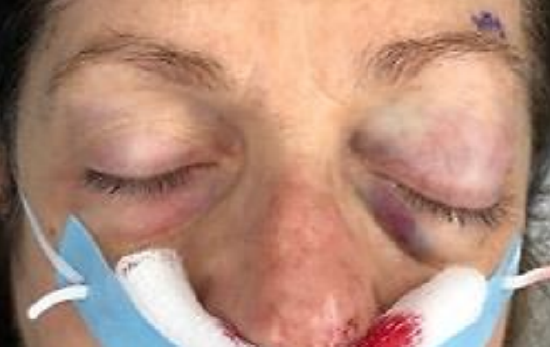
Figure 17: A left compartment syndrome with an IOP of 55 mmHg
- Placement of orbital fat over the exposed extraocular muscles should be considered to prevent scarring 12,26 (Figure 15)
- Reconstructing the medial orbital wall to preserve orbital volume should be strongly considered following removal of intraconal or large extraconal lesions
- There is no clear consensus on the best method of reconstruction
- A pedicled nasoseptal flap technique is preferred by the authors as it provides the opportunity for delayed contraction thereby reducing the risk of diplopia and enophthalmos 26 (Figure 16)
- Immediate rigid reconstruction does place the orbit at risk for compartment syndrome secondary to edema and postoperative bleeding (Figure 17)
12. Nasal packing
There is lack of consensus on the use of post-surgery nasal packing. Nasal packing is not recommended by some studies, citing possible exertion of pressure on the globe or optic nerve.12,21 Alternatively, absorbable hemostatic packing has been used 27 and the use of a polyvinyl acetate sponge placed in the nasal cavity to assist with adherence and positioning of a nasoseptal flap for 7 days has been reported. However, packing should not be placed in a position which could risk exerting direct pressure on the exposed orbit.
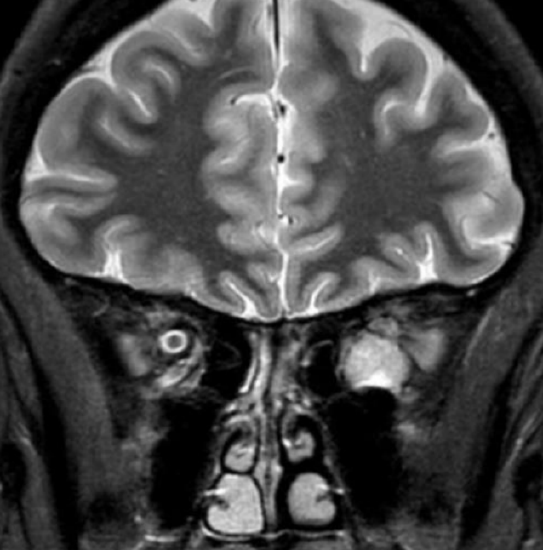
Figure 18a: A T2-weighted MRI coronal scan demonstrating a left orbital hemangioma
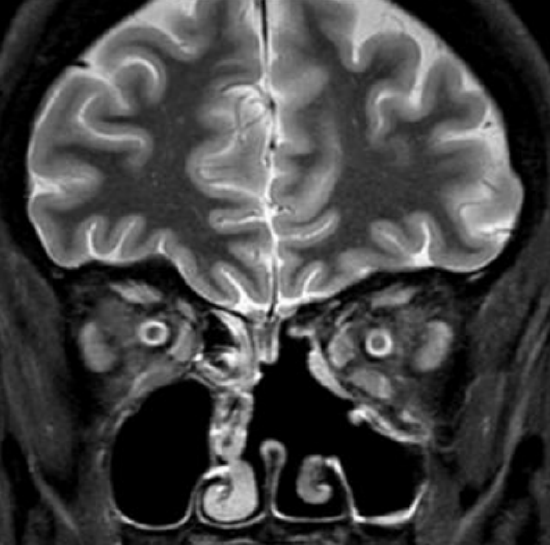
Figure 18b: A T2-weighted MRI coronal scan demonstrating postoperative situation with a left nasoseptal flap positioned along the medial orbital wall
References
- Calandriello L, Grimaldi G, Petrone G, et al. Cavernous venous malformation (cavernous hemangioma) of the orbit: Current concepts and a review of the literature. Surv Ophthalmol. 2017; 62 (4):393-403
- 2. Scheuerle AF, Steiner HH, Kolling G, Kunze S, Aschoff A. Treatment and long-term outcome of patients with orbital cavernomas. Am J Ophthalmol. 2004;138(2):237-44
- Harris GJ, Perez N. Surgical sectors of the orbit: using the lower fornix approach for large, medial intraconal tumors. Ophthalmic Plast Reconstr Surg. 2002;18(5):349-54
- McNab AA, Tan JS, Xie J, et al. The natural history of orbital cavernous hemangiomas. Ophthalmic Plast Reconstr Surg. 2015;31(2):89-93
- Chen Y, Tu Y, Chen B, et al. Endoscopic Transnasal Removal of Cavernous Hemangiomas of the Optic Canal. Am J Ophthalmol. 2017; 173:1-6
- Yoshimura K, Kubo S, Yoneda H, Hasegawa H, Tominaga S, Yoshimine T. Removal of a cavernous hemangioma in the orbital apex via the endoscopic transnasal approach: a case report. Minim Invasive Neurosurg. 2010;53(2):77-9
- Wiwatwongwana D, Rootman J. Management of optic neuropathy from an apical orbital-cavernous sinus hemangioma with radiotherapy. Orbit. 2008;27(3):219-21
- Lee KY, Fong KS, Loh HL, et al. Giant cavernous hemangioma mimicking a fifth nerve neurofibroma involving the orbit and brain. Br J Ophthalmol. 2008;92(3): 423-5
- Rootman DB, Heran MK, Rootman J, et al. Cavernous venous malformations of the orbit (so-called cavernous hemangioma): a comprehensive evaluation of their clinical, imaging and histologic nature. Br J Ophthalmol. 2014;98(7): 880-8
- Bleier BS, Castelnuovo P, Battaglia P, et al. Endoscopic endonasal orbital cavernous hemangioma resection: global experience in techniques and outcomes. Int Forum Allergy Rhinol. 2016;6(2):156-61
- Gregorio LL, Busaba NY, Miyake MM, et al. Expanding the limits of endoscopic intraorbital tumor resection using 3-dimensional reconstruction. Braz J Otorhinolaryn gol. 2017. https://doi.org/10.1016/j.
- Castelnuovo P, Dallan I, Locatelli D, et al. Endoscopic transnasal intraorbital surgery: our experience with 16 cases. Eur Arch Otorhinolaryngol. 2012;269 (8):1929-35
- Lenzi R, Bleier BS, Felisati G, Muscatello L. Purely endoscopic trans-nasal management of orbital intraconal cavernous hemangiomas: a systematic review of the literature. Eur Arch Otorhinolaryngol. 2016;273(9):2319- 22
- Chhabra N, Wu AW, Fay A, Metson R. Endoscopic resection of orbital hemangiomas. Int Forum Allergy Rhinol. 2014;4(3):251-5
- Stamm A, Nogueira JF. Orbital cavernous hemangioma: transnasal endoscopic management. Otolaryngol Head Neck Surg. 2009;141(6):794-5
- Tomazic PV, Stammberger H, Habermann W, et al. Intraoperative medialization of medial rectus muscle as a new endoscopic technique for approaching intraconal lesions. Am J Rhinol Allergy. 2011;25(5):363-7
- Lazar M, Rothkoff L, Drey JP. Treatment and long-term outcome of patients with orbital cavernomas. Am J Ophthalmol. 2005;139(4):753; author reply 753
- Karaki M, Kobayashi R, Mori N. Removal of an orbital apex hemangioma using an endoscopic transethmoidal approach: technical note. Neurosurgery. 2006;59 (1 Suppl 1):ONSE159-160; discussion ONSE 159-60
- Wu W, Selva D, Jiang F, et al. Endo scopic transethmoidal approach with or without medial rectus detachment for orbital apical cavernous hemangiomas. Am J Ophthalmol. 2013;156(3):593-9
- Murchison AP, Rosen MR, Evans JJ, Bilyk JR. Endoscopic approach to the orbital apex and periorbital skull base. Laryngoscope. 2011;121(3):463-7
- Paluzzi A, Gardner PA, FernandezMiranda JC, et al. "Round-the-Clock" Surgical Access to the Orbit. J Neurol Surg B Skull Base. 2015;76(1):12-24
- Bleier BS, Healy DY, Jr., Chhabra N, Freitag S. Compartmental endoscopic surgical anatomy of the medial intraconal orbital space. Int Forum Allergy Rhinol. 2014;4(7):587-91
- Mueller SK, Freitag SK, Bleier BS. Morphometric Analysis of the Orbital Process of the Palatine Bone and its Relationship to Endoscopic Orbital Apex Surgery. Ophthalmic Plast Reconstr Surg. 2018;34(3):254-7
- Lee JY, Ramakrishnan VR, Chiu AG, et al. Endoscopic endonasal surgical resection of tumors of the medial orbital apex and wall. Clin Neurol Neurosurg. 2012;114(1): 93-8
- Stamm AC, Vellutini E, Harvey RJ, et al. Endoscopic transnasal craniotomy and the resection of craniopharyngioma. Laryngoscope. 2008;118(7):1142- 8
- Healy DY, Jr., Lee NG, Freitag SK, Bleier BS. Endoscopic bimanual approach to an intraconal cavernous hemangioma of the orbital apex with vascularized flap reconstruction. Oph thalmic Plast Reconstr Surg. 2014;30 (4):e104-6
- Muscatello L, Seccia V, Caniglia M, et al. Transnasal endoscopic surgery for selected orbital cavernous hemangiomas: our preliminary experience. Head Neck. 2013;35(7):E218-20
Authors
Catherine Banks MBChB FRACS
Department of Otolaryngology
Head & Neck Surgery.
Rhinology Fellow
Massachusetts Eye and Ear Infirmary/
Harvard, Boston MA 02114 USA
catherinebanks@MEEI.HARVARD.edu
Benjamin Bleier MD FACS
Associate Professor of Otolaryngology
Massachusetts Eye & Ear Infirmary/
Harvard, Boston MA 02114 USA
Benjamin_Bleier@MEEI.Harvard.edu
Editor
Johan Fagan MBChB, FCS (ORL), MMed
Professor and Chairman
Division of Otolaryngology
University of Cape Town
Cape Town, South Africa
johannes.fagan@uct.ac.za


