6.9.17: 17. Surgical Approaches - Oropharynx Cancer Resection- External Approaches
- Page ID
- 55468
OPEN ACCESS ATLAS OF OTOLARYNGOLOGY, HEAD & NECK OPERATIVE SURGERY
RESECTION OF CANCER OF THE OROPHARYNX
Johan Fagan, Eugene Myers
Management of cancer of the oropharynx has undergone a paradigm shift in recent years following the realization that HPV infection is both an etiologic and prognostic factor for a subset of oropharyngeal squamous cell carcinoma; the introduction of transoral robotic surgery (TORS) to resect oropharyngeal tumors; as well as attempts to reduce the morbidity of chemoradiation by accepting smaller resection margins when combined with postoperative irradiation.
This chapter describes transoral and transmandibular resection of cancer of the oropharynx; it does not address the debate of the relative merits of chemoradiation vs. transoral excision using electrocautery, CO2 laser or robotics vs. open resection. Readers are referred to other chapters in the Open Access Atlas of Otolaryngology, Head & Neck Operative Surgery for detailed descriptions of Transoral laser microsurgery (TLM), Transoral lateral oropharyngectomy/radical tonsillectomy, and Resection of cancers of the base of tongue.
Surgical anatomy
The oropharynx is comprised of the base of the tongue, tonsils, soft palate, and the lateral and posterior walls of the pharynx between the levels of the hard palate and hyoid bone.
Base of the tongue (BOT)
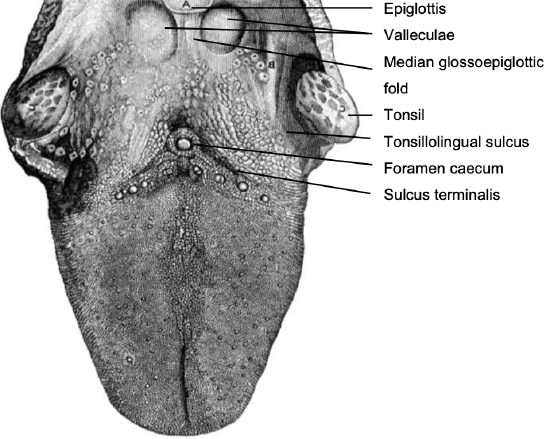
Figure 1: Topography of BOT
This comprises the posterior 1/3 of the tongue behind the foramen caecum and sulcus terminalis (Figure 1). The mucosa is rough, thick and fixed to the underlying muscle and contains lymphoid follicles (lingual tonsil); this makes it difficult to identify the edges of a BOT tumor; hence frozen section is especially useful to assess resection margins. Posterolaterally the tonsillolingual sulci separate the tongue from the tonsillar fossae. The valleculae separate the BOT from the lingual surface of the epiglottis and are separated in the midline by the median glossoepiglottic fold (Figure 1).
Soft palate
The soft palate has a complex muscular structure and innervation. It has key functions relating to speech and swallowing.
Resection and inadequate reconstruction of the palate results in loss of nasal separation which manifests clinically as nasal regurgitation of fluids and food, and hypernasal speech which can be quite disabling. The complex muscular anatomy of the soft palate and lateral pharyngeal wall is illustrated in Figure 4.
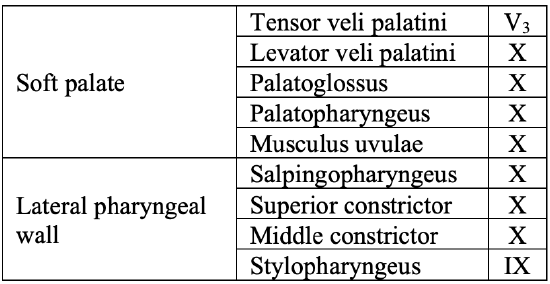
Table 1: Muscles of the soft palate and oropharynx and cranial nerves which innervate them
Muscles that contribute to the soft palate and lateral pharyngeal wall are summarized in Table 1.
Lateral and posterior pharyngeal walls
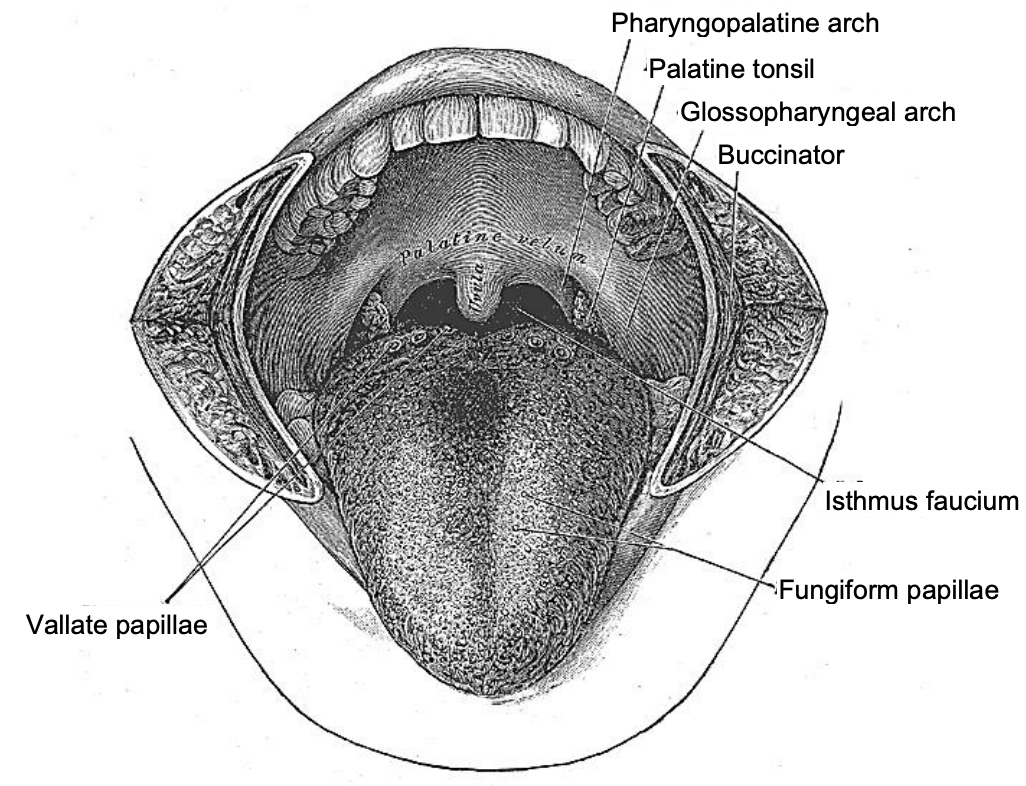
Figure 2: Anterior view of soft palate
Figure 3: Lateral view of soft palate and its attachment to the hard palate
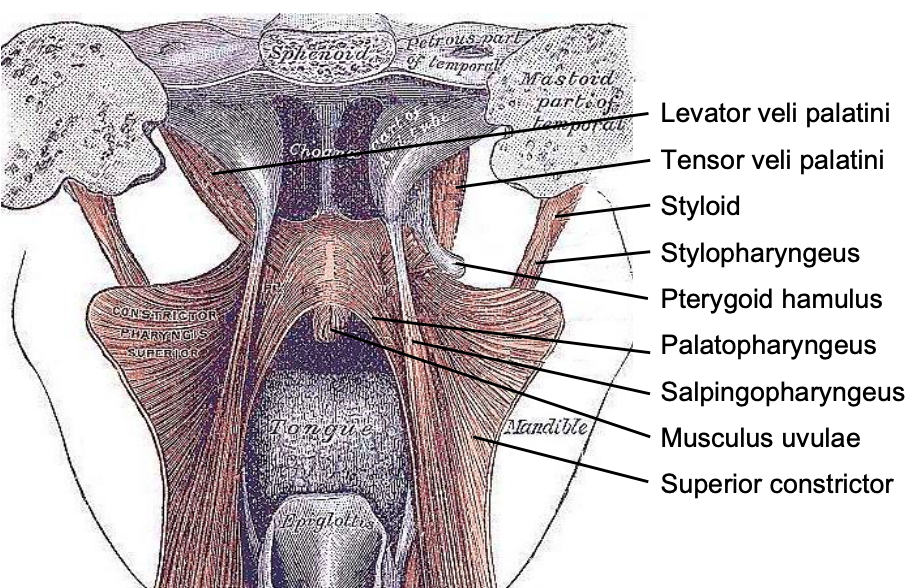
Figure 4: Posterior view of the soft palate and pharynx with the superior constrictor splayed open
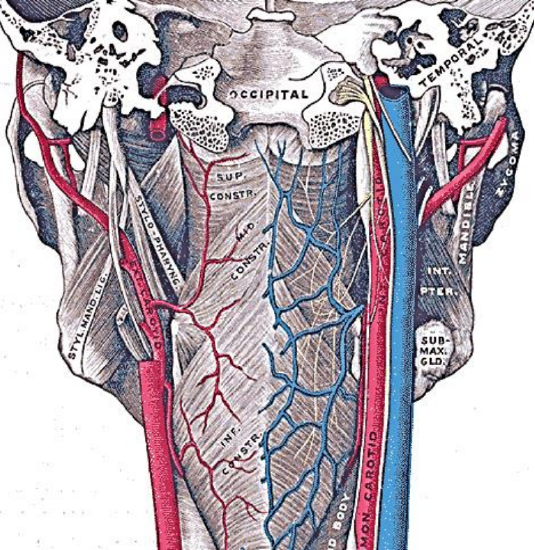
Figure 5: Posterior view of the pharynx illustrates the attachment of the superior constrictor to the pharyngobasilar fascia above and the median raphe to which the pharyngeal constrictors attach; note the close anatomical relationships of the internal and external carotid artery systems to the pharynx
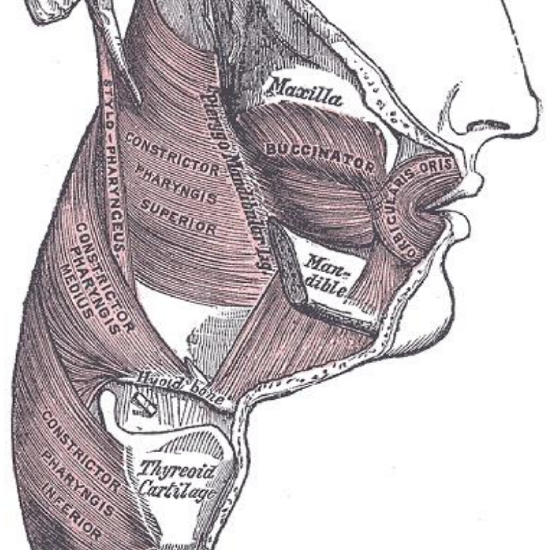
Figure 6: Note how buccinator and superior constrictor muscles attach to the pterygomandibular raphe
The pharynx has a number of layers i.e. mucosa, submucosa, muscle and serosa. The submucosa is represented by the pharyngobasilar fascia; it thins as it extends inferiorly from its attachment to the skull base and lines the inner aspect of the constrictor muscles. It fills the gaps between the skull base and the superior constrictor, the superior and middle constrictors, and the middle and inferior constrictors (Figure 5). Posteriorly it forms a median raphe to which the pharyngeal constrictors attach (Figure 5). The anterior faucial/tonsillar pillars are formed by the palatoglossus muscles with a thin mucosal covering and constitute the anterior limit of the oropharynx, behind which it is encircled by the superior and middle constrictor muscles (Figures 2, 4, 5, 6). The palatopharyngeus muscles constitute the posterior faucial/tonsillar pillars. The tonsils are situated between the anterior and posterior pillars, with the pharyngeal constrictor muscle lateral to the tonsil. The buccopharyngeal fascia constitutes the serosal layer; it is the outermost coat of the pharynx and is a thin layer of connective tissue that forms the anterior wall of the prevertebral space.
Nerves
The only notable nerves encountered at surgery are the lingual, inferior alveolar and mental nerves, and possibly the XIIn if resection includes the lateral tongue.
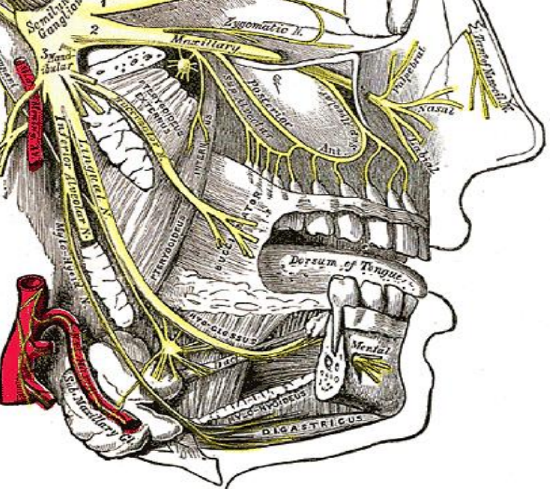
Figure 7: V3, lingual, inferior alveolar and mental nerves
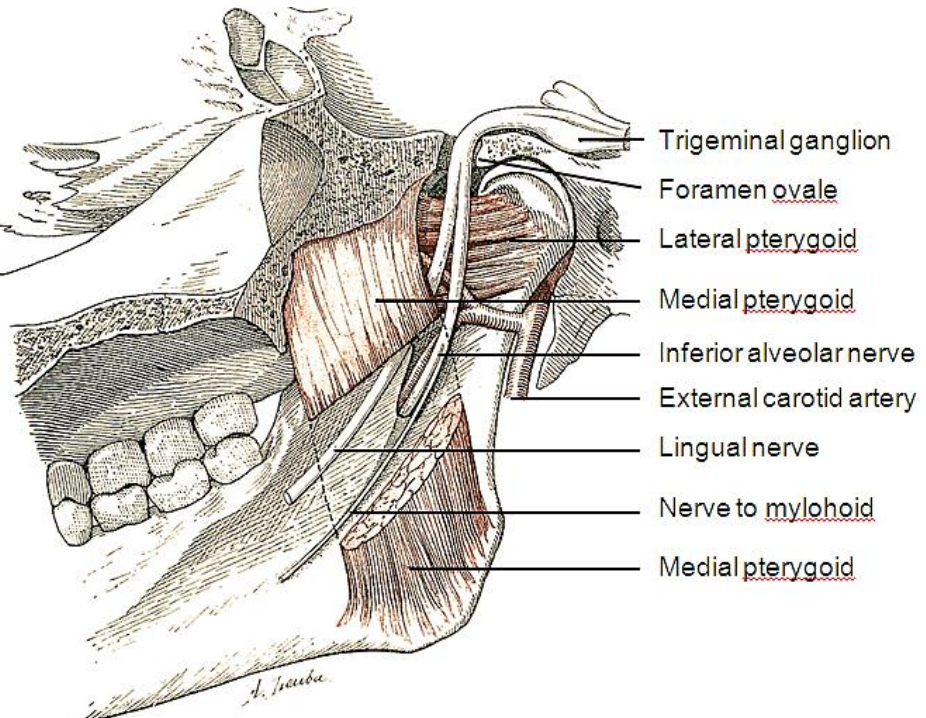
Figure 8: V3, lingual, inferior alveolar and mental nerves
The lingual nerve is a branch of V3 and provides sensory innervation to the floor of the mouth (FOM). The nerve crosses deep to the submandibular duct in the lateral FOM; in the anterior FOM it is located posterior to the duct (Figures 7, 8). It is worthwhile attempting to preserve the nerve when resecting smaller tumors in the tonsillar region.
The inferior alveolar nerve and its accompanying vessels enter the mandibular canal at the lingula and innervate the inferior alveolus and teeth (Figures 7, 8). The nerve is transected whenever a segmental mandibulectomy is done. The mental branch of the nerve exits the mandibular canal at the mental foramen to innervate the skin of the lower lip and should be preserved when doing a mandibulotomy by placing the osteotomy anterior to the mental foramen (Figure 7).
Arteries
Knowledge of vascular anatomy enables the surgeon to anticipate where arteries will be encountered and helps to minimize blood loss. Depending on the surgical procedure, vessels that may be encountered include the inferior labial, facial, lingual, inferior alveolar, mental, palatal and tonsillar arteries and the pterygoid venous plexus.
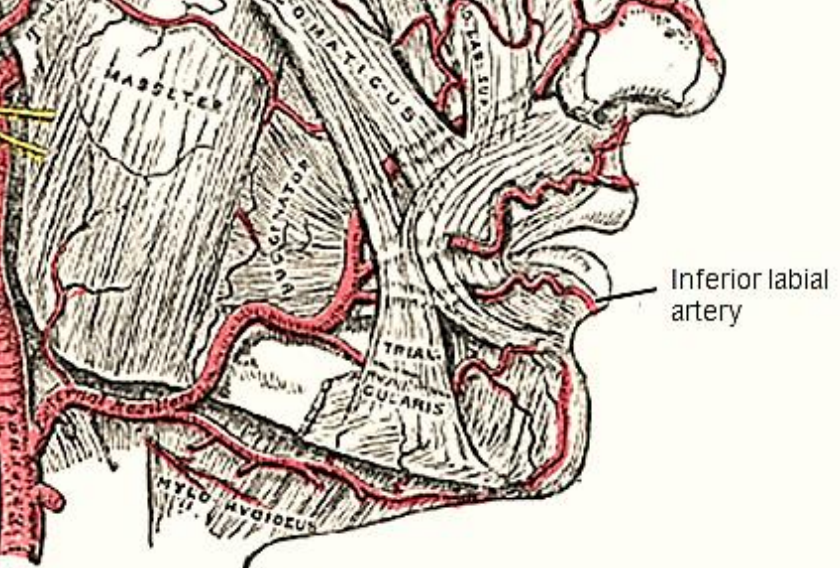
Figure 9: Inferior labial artery
The inferior labial artery is transected when the lower lip is divided in the midline. A branch of the facial artery, it is located superficially, just deep to the surface of the lip, and is controlled with bipolar cautery (Figure 9).
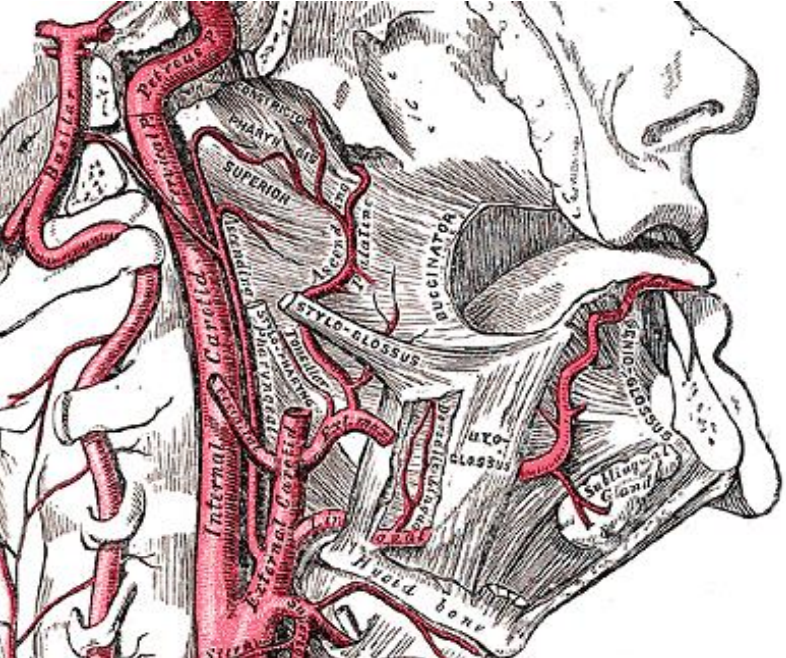
Figure 10: The ascending pharyngeal and ascending palatine and tonsillar branches of the facial (external maxillary) artery lie on the pharyngeal constrictor deep to the medial and lateral pterygoid muscles
The facial artery is generally not a problem as it is reflected laterally in the cervicofacial flap that is raised when exposing the lateral surface of the mandible (Figure 9). It is commonly used as a donor vessel for free microvascular transfer flap reconstruction. The tonsillar and ascending palatine arteries are branches of the facial artery (Figure 10). The lingual artery is only encountered when resection includes the posterolateral tongue (Figure 10).
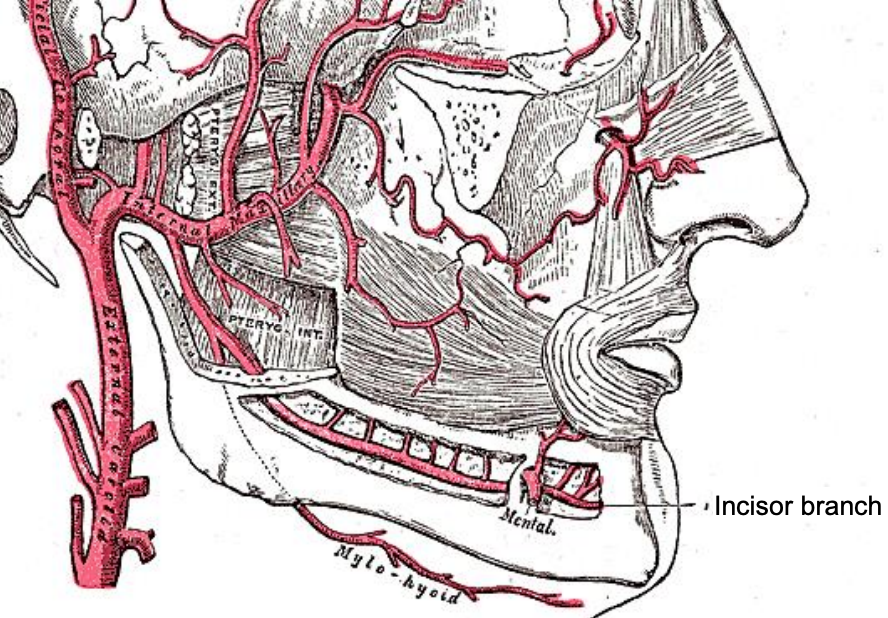
Figure 11: Buccinator, inferior alveolar and mylohyoid arteries
The inferior alveolar artery is a branch of the internal maxillary artery, and courses between the medial pterygoid muscle and mandible where it enters the mandibular canal at the lingula accompanied by the inferior alveolar nerve and vein (Figures 5, 8, 11).
The mental artery accompanies the mental nerve and exits the mandible at the mental foramen (Figure 11). If a mandibulotomy is to be done for access just anterior to the mental foramen, avoid dissecting right up to the foramen so as not to cause bleeding requiring cautery, as this might injure the nerve. The arterial supply to the soft palate is derived mainly from the ascending pharyngeal artery, which anastomoses with the greater palatine artery at the junction of the hard and soft palate; additional supply is derived from the ascending palatine and tonsillar arteries (Figure 10). Despite transecting the soft palate with diathermy, one may encounter troublesome bleeding which is controlled with diathermy.
Muscles
The muscles of the soft palate, the pharyngeal constrictors and the palatopharyngeus and palatoglossus are generally not individually dissected and identified but are simply resected with the primary tumor using electrocautery and scissors. Muscles that are encountered depending on the surgical approach and that are individually identified include orbicularis oris, masseter, temporalis, mylohyoid, and the medial and lateral pterygoids.
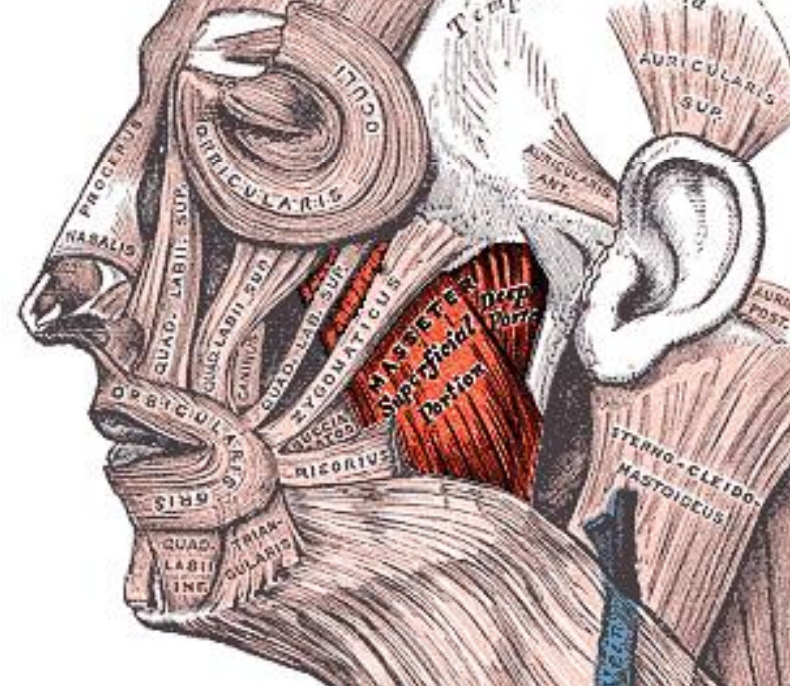
Figure 12: Orbicularis oris and masseter
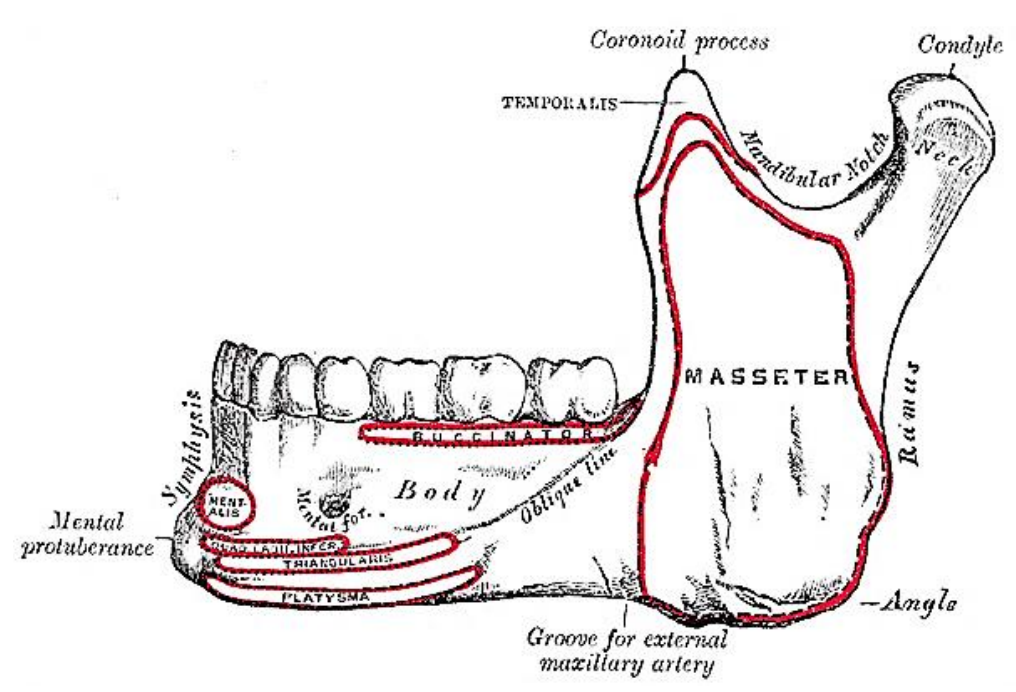
Figure 13: Muscle attachments to outer aspect of mandible and location of mental foramen
The orbicularis oris is transected with a lip-split incision and has to be carefully reapproximated at conclusion of surgery (Figure 12). Dissecting along the lateral surface of the mandible, one next encounters the masseter muscle (Figure 12). It is a thick quadrilateral muscle with superficial and deep portions, the fibers of which converge at the insertion to the lateral aspect of the vertical ramus of the mandible (Figure 13). Both the superficial and deep portions arise from the zygoma. Laterally the muscle is covered by the parotid gland. It is innervated by V3.
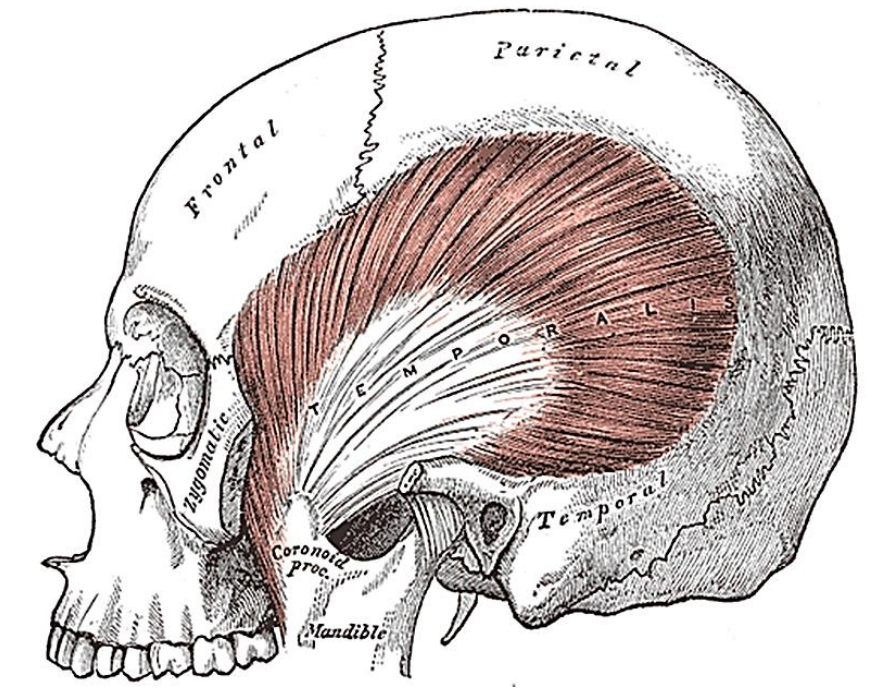
Figure 14: Temporalis muscle inserts into coronoid and anteromedial surface of vertical ramus of mandible
The next muscle one encounters as one dissects along the lateral surface towards the coronoid process of the mandible is the temporalis muscle (Figures 13, 14).
The temporalis muscle arises from the temporal fossa and temporalis fascia and passes behind the zygomatic arch to insert onto the coronoid process and anteromedial surface of the vertical ramus (Figures 13, 14). It is innervated by V3. It receives its blood supply from the deep temporal arteries; preserving these arteries is crucial if a temporalis muscle flap is to be used to reconstruct a surgical defect.
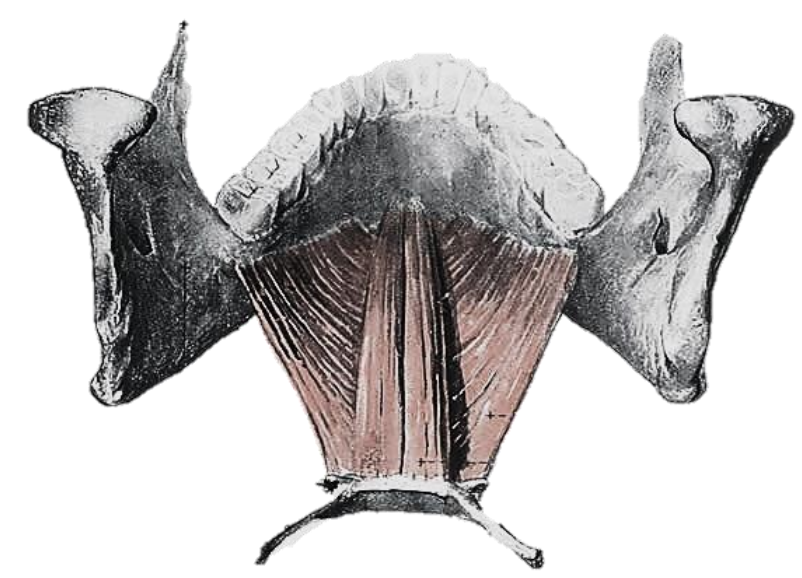
Figure 15: Geniohyoid and mylohyoid
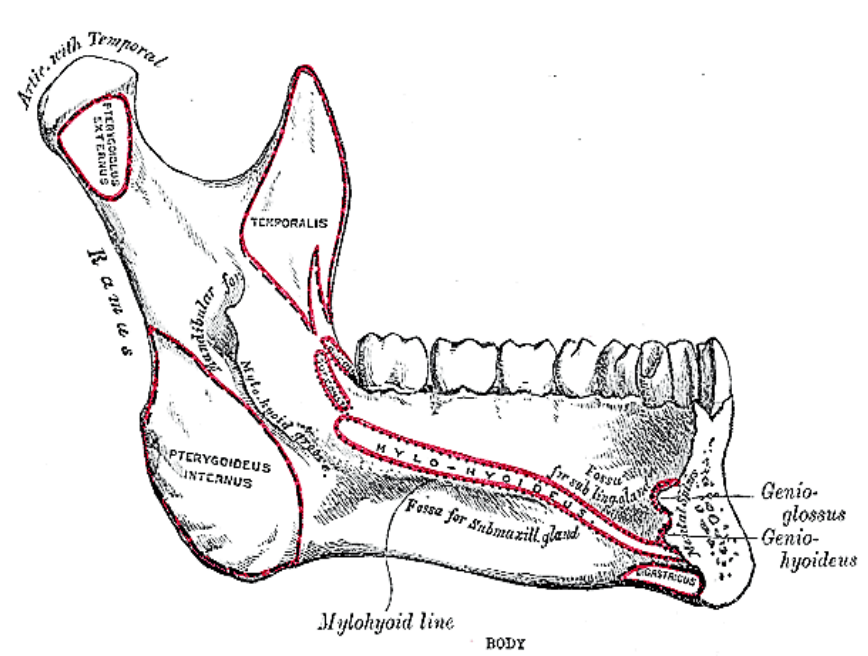
Figure 16: Attachments of mylohyoid, geniohyoid, genioglossus and digastric muscles to inner aspect of mandible
The mylohyoid muscle is the “diaphragm” of the mouth, and is transected along the medial margin of the mandible when doing a mandibular swing approach to the oropharynx (Figures 15, 16).
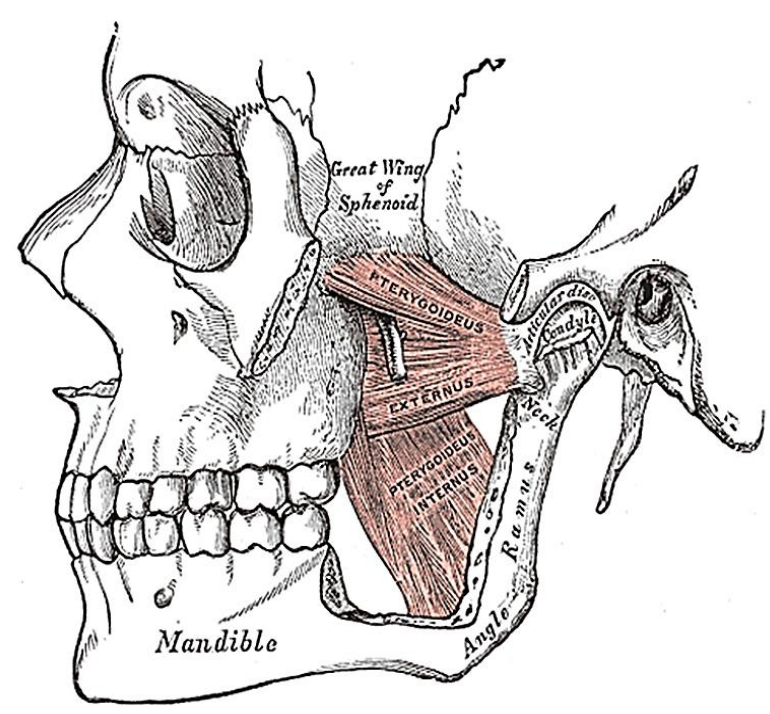
Figure 17: Medial and lateral pterygoids
The next muscle encountered along the medial aspect of the mandible is the medial pterygoid (Figures 8, 17). It has two heads (Figure 17): the larger deep head arises from just above the medial surface of the lateral pterygoid plate; the smaller superficial head arises from the maxillary tuberosity and the pyramidal process of the palatine bone (Figure 17). The muscle passes inferolaterally to insert onto the inferomedial surface of the ramus and angle of the mandible (Figures 16, 17). It is innervated by V3.
The final muscle that may be encountered is the lateral pterygoid; its superior head originates from the infratemporal surface and crest of the greater wing of the sphenoid, and the inferior head from the lateral surface of the lateral pterygoid plate (Figure 17). It inserts onto the neck of mandibular condyle and the articular disc and capsule of the temporomandibular joint (Figures 16, 17). It is innervated by V3.
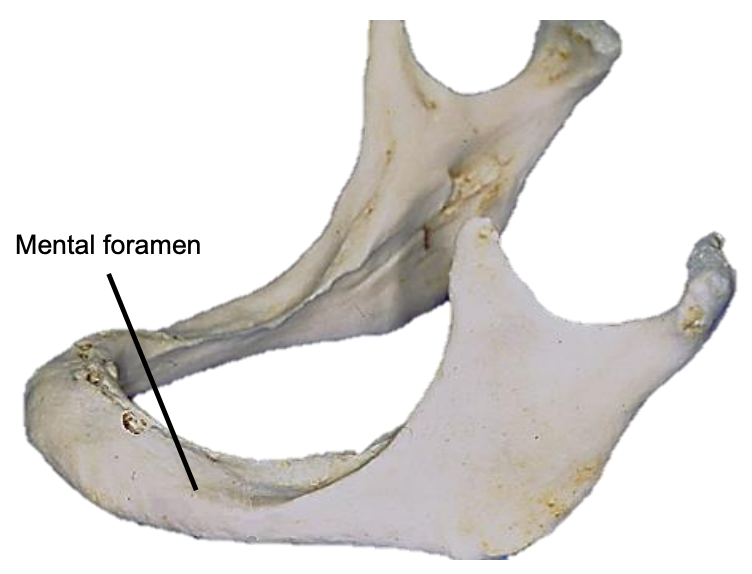
Figure 18: Position of mental foramen in a resorbed mandible
The mandible may need to be divided (mandibulotomy) to gain access to the oropharynx, or the angle and vertical ramus of the mandible may be included in the tumor resection (marginal, segmental or hemimandibulectomy). Important surgical features are the position of the mental foramen through which the mental nerve exits to innervate the lower lip, the mylohyoid line to which the mylohyoid muscle attaches, and when planning a marginal mandibulectomy, the height of the body of the mandible and the depth of the dental roots (Figures 13, 16). The mental foramen and inferior alveolar nerve may be very close to the superior surface of a resorbed mandible such as is seen in older, edentulous patients (Figure 18). A marginal mandibulectomy may not be possible in such a resorbed mandible due to the lack of residual bone.
Surgical Objectives
- Adequate tumor resection margins; this requires a surgical approach that permits good exposure
- Surgical morbidity relating to palatal function (nasal regurgitation and speech), swallowing, mandible, lingual nerve function and cosmesis should be kept to a minimum; prevent pharyngocutaneous fistulae
- Control occult cervical metastasis; the neck is electively treated when there is significant risk of occult cervical metastases; the author generally does elective neck dissection (END) levels I-III for squamous cell carcinoma; and bilateral END with cancers of the soft palate and/or base of tongue
The remainder of this chapter focuses on surgical treatment of the primary tumor.
Preoperative evaluation
- Are there synchronous primaries, cervical or distant metastases? CXR or CT chest, and panendoscopy
- Is the tumor resectable? Since local control and ultimately cure depend upon complete resection of cancer with clear margins, there is little point in doing an incomplete tumor resection. It may be difficult to assess the extent of the primary due to pain, tenderness or trismus. If in doubt, a patient requires imaging such as CT/MRI, or examination under anesthesia. Examples of irresectable tumors include posterior extension to involve the carotid sheath and parapharyngeal space, superior extension to the infratemporal fossa and skull base, perineural extension to the trigeminal ganglion, and irresectable node metastases. A key consideration is surgical morbidity; this in turn is dependent on the range and reliability of reconstructive surgery and prosthodontic support services
- Is there clinical evidence of perineural invasion (PNI) of major nerves? Examine the patient for neurological deficits of the hypoglossal, mental, inferior alveolar, lingual, and trigeminal nerves and of the sympathetic trunk (Horner’s syndrome). Widening of the mandibular canal on mandibular orthopantomography (Panorex) may be seen. MRI may demonstrate PNI. Should there be evidence of PNI then the affected nerve should be dissected proximally until a clear tumor margin is obtained on frozen section. In the case of the inferior alveolar nerve, this would require a hemi- or segmental mandibulectomy that encompasses the entire inferior alveolar canal Mental foramen
- Is a tracheostomy required? Most patients with resections requiring any form of reconstruction with a flap are at risk of airway obstruction and should have a temporary tracheostomy. Oral or transnasal intubation may be difficult or impossible due to tumor and/or trismus; a tracheostomy is then done under local anesthesia and the patient is intubated through the stoma
- What type of surgical approach is required? Select surgical approaches that ensure adequate exposure; transoral access is often inadequate
- Does the mandible have to be resected? Tumor may involve the medial pterygoid muscle at its insertion onto the mandible (clinically manifested by trismus), extend onto periosteum, invade only the inner cortex, involve medullary bone, or manifest PNI of the inferior alveolar nerve. Panorex only shows gross involvement of bone (Figure 19). Figure 19: Panorex shows mandibular destruction by tumor Invasion of cortical bone is better assessed with CT; MRI is superior for assessing medullary invasion. In the absence of CT scans the surgeon may elect to strip tumor off bone at the time of resection taking care not to seed tumor, inspect the bone for evidence of periosteal and bony infiltration, and then decide how to proceed. If only the periosteum is involved, then a marginal mandibulectomy (removal of cortical bone) may suffice. Should marginal mandibulectomy be considered, then the vertical height of the mandible should be assessed clinically or by Panorex so as to predict whether a free bony composite flap would be required to strengthen the remaining mandible. Once the cortex is invaded, then either marginal or segmental mandibulectomy is required. Medullary invasion requires segmental or hemimandibulectomy that includes at least a 2 cm length of mandible on either side of the tumor
- Status of dentition? Carious teeth are removed at the time of the surgery to reduce the risk of osteoradionecrosis
- What type of reconstruction is required? This can often only determined once the resection has been done and the defect can be assessed
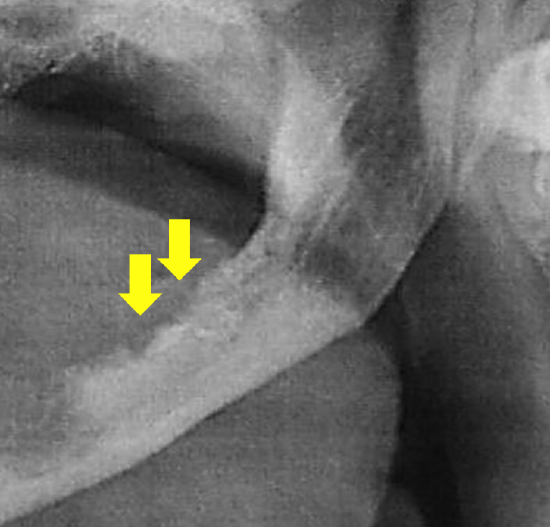
Figure 19: Panorex shows mandibular destruction by tumor
Key surgical points
- Preserve function
- Attempt to preserve the lingual and hypoglossal nerves
- Reconstruct soft palate defects
- Reconstruct mandibular defects
- Do not compromise margins to preserve function
- Employ frozen section (if available) to ensure clear resection margins
- Have a low threshold for performing a tracheostomy
Anesthesia
The surgeon always stands by the patient’s side during induction of anesthesia as it may be difficult or impossible to intubate a patient who has a bulky tumor, or who has trismus. Should the anesthetist be unable to intubate the larynx, the surgeon may be able to intubate through a laryngoscope or do an emergency tracheostomy or cricothyroidotomy; it is prudent to inject the tracheostomy or cricothyroidotomy site with local anesthetic (with adrenaline) prior to induction. Nasal intobation facilitates resection of pharyngeal tumors and is converted to a tracheostomy during the course of the operation.
Perioperative antibiotics are prescribed for 24 hours.
Surgical access
Good surgical access is essential in order to attain adequate resection margins, to control bleeding, and for reconstruction. A combination of surgical approaches can be used and will now be discussed. Level 1 of the neck should first be dissected before proceeding to address the primary tumor if neck dissection is indicated.
Transoral resection with electrocautery (Bovie)
Transoral access varies considerably. Edentulous patients are generally better suited to transoral resection. A useful way to predict whether transoral resection is likely to be possible is to pull on the anterior tongue with a cotton swab during preoperative clinical evaluation and to then see how accessible the tumor is. Note that tumors become more visible and accessible as the resection proceeds, especially once the thick BOT lining has been incised around tumor.

Figure 20: Dental bite block is interposed between lateral teeth to keep mouth open
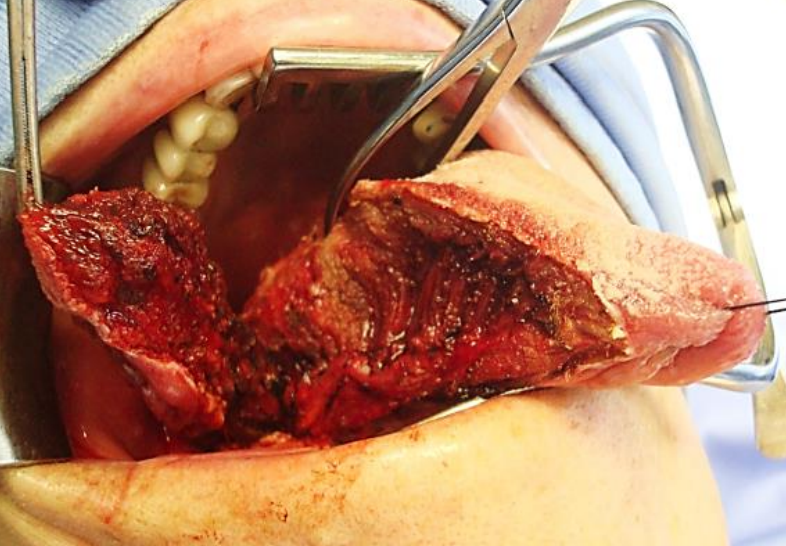
Figure 21: Self-retaining retractor; lateral oral and BOT tumor being resected

Figure 22: Dingman retractor
The mouth is kept wide open either with a dental bite block (Figure 20), or with a self-retaining retractor, taking care to protect the teeth from injury (Figure 21), or with a tonsil gag or Dingman retractor (Figure 22).
Apply traction to the tongue and the tumor with silk traction sutures or with tissue forceps (Figure 21). Resect the tumor with at least a 1 cm margin of normal tissue using electrocautery (Bovie). As the resection proceeds, insert new silk traction sutures or reapply the tissue forceps to facilitate delivery of the tumor.
Mandibulotomy with mandibular swing
This affords excellent access to the oropharynx. It does however leave a facial scar; it may cause deformity of the lower lip; there is a risk of complications relating to the mandibulotomy and dental malocclusion; and the lingual nerve is at risk of injury.
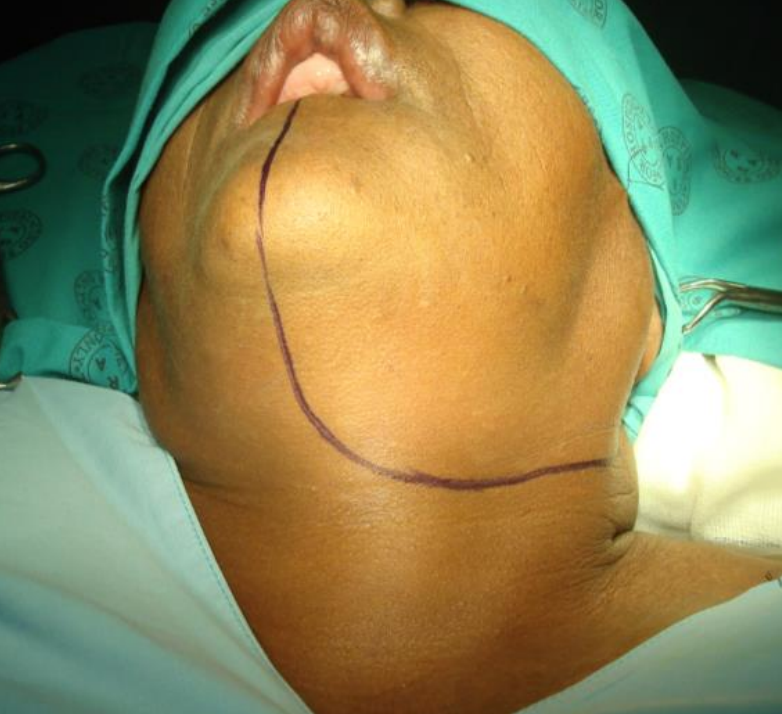
Figure 23: Skin incisions for Level 1 neck dissection and mandibulotomy approach
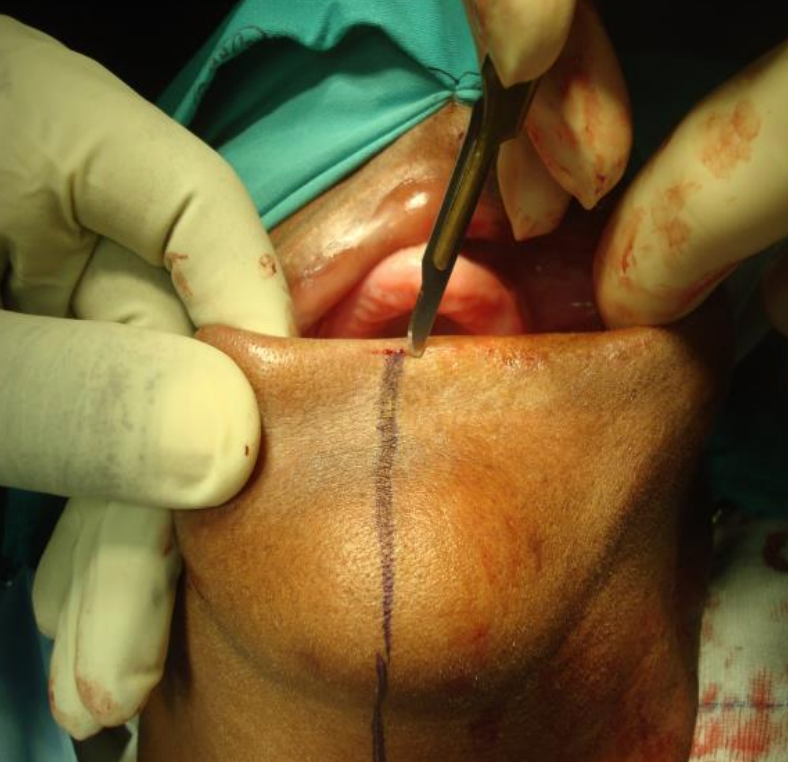
Figure 24: Scoring the vermillion border
Having completed the Level 1 neck dissection, the vermillion border is scored or marked so as to ensure an accurate repair (Figures 23, 24).
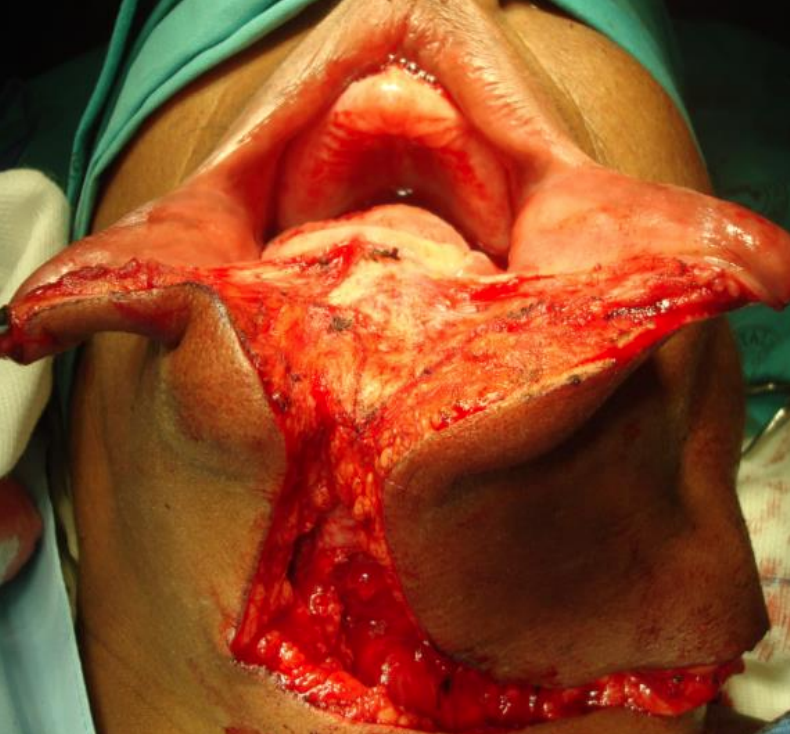
Figure 25: The lower lip is divided vertically in the midline onto bone after Level 1 neck dissection has been completed
The lower lip is vertically divided in the midline (Figure 25). Bleeding from the inferior labial artery is controlled with bipolar cautery. At this point, some surgeons prefer to do a median mandibulotomy; it is however the authors’ preference to do a paramedian mandibulotomy.
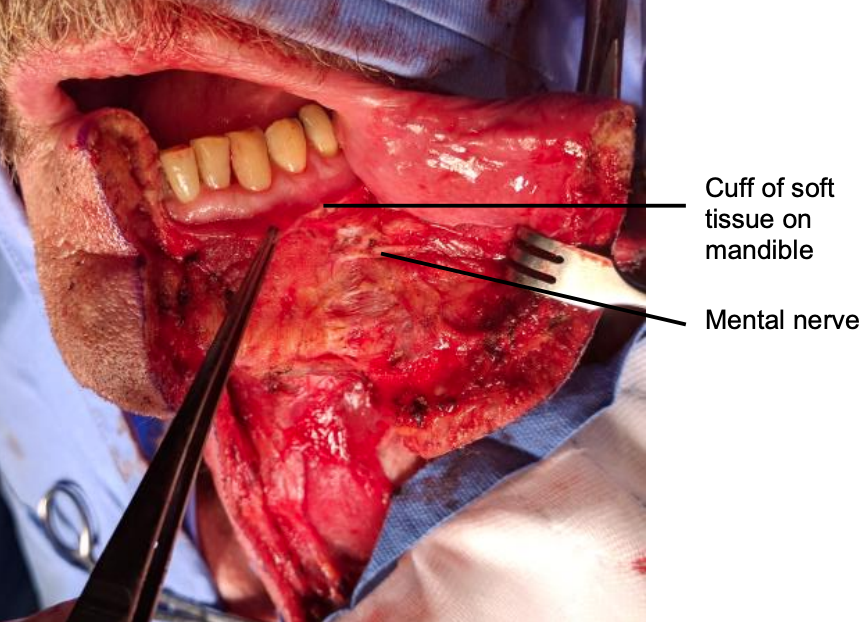
Figure 26: Mental nerve and cuff of soft tissue
Incise the gingivolabial and gingivobuccal mucosae >0.5 cm from the bone to leave a cuff of soft tissue on the bone to facilitate subsequent soft tissue closure (Figure 26).
Strip soft tissue off the mandible with monopolar cautery or with a periosteal elevator stopping just short of the mental foramen so as not to injure the mental nerve or mental artery where it exits the foramen (Figure 26).

Figure 27: Gigli saw
A paramedian mandibulotomy is done just anterior to the mental foramen. Compared to median mandibulotomy, the advantages are that it is closer to tumor, it does not disturb the genioglossus, geniohyoid, and digastric muscles, there is less disruption of the FOM, and should segmental mandibulectomy be required, then only the lateral mandible is sacrificed. The mandible is divided just anterior to the mental foramen with a Gigli or a powered saw (Figure 27).
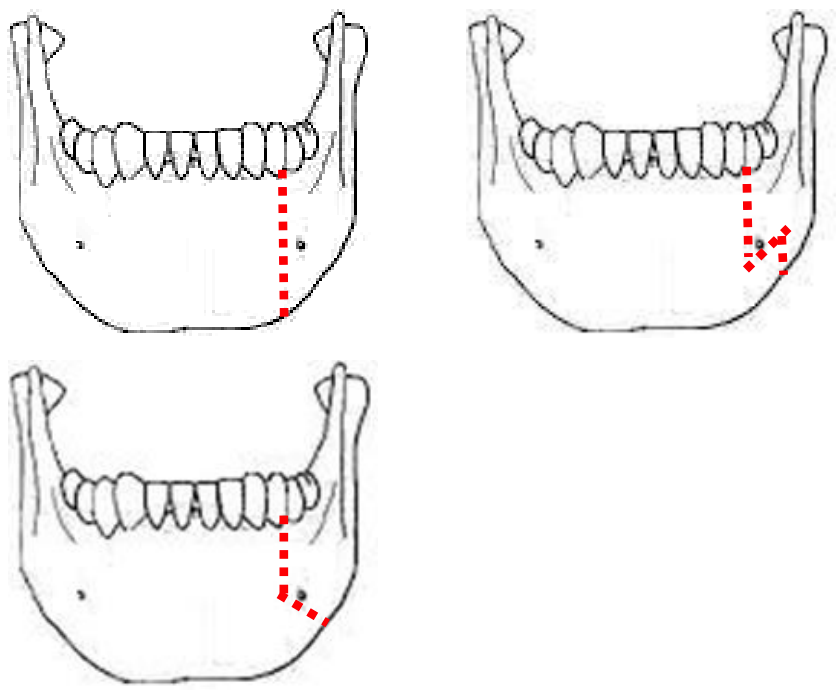
Figure 28: Examples of osteotomies
The osteotomy may be made vertically or alternatively in a step- or V-shaped fashion to achieve a more stable repair (Figure 28). It is advisable to extract a tooth and make the osteotomy through the dental socket to avoid devitalizing adjacent teeth.
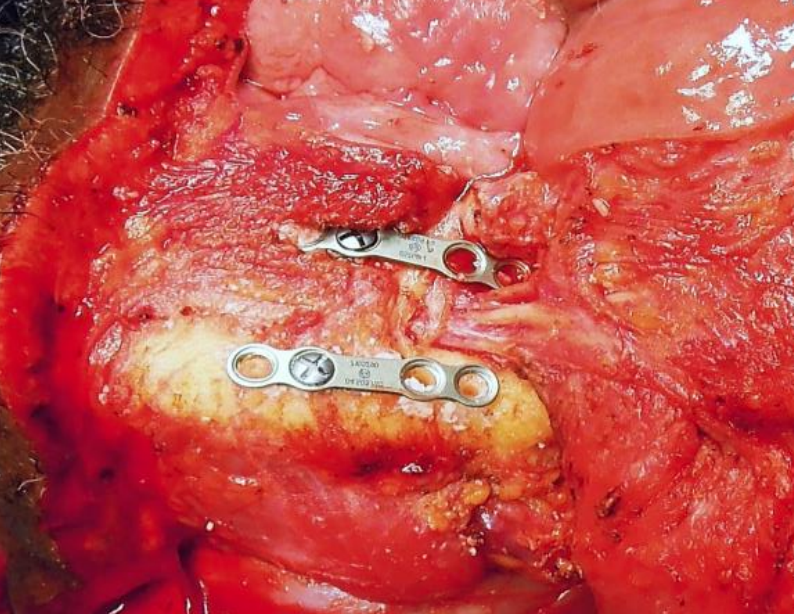
Figure 29: Preplating with 4-holed miniplates above and below mental nerve
In dentate patients the mandible is preplated with titanium miniplates contoured to the mandible to ensure perfect dental alignment. Two 4-holed, non-compression, 2 mm mandibular plates, one placed along the inferior border of the mandible and the other placed more superiorly, are used (Figure 29). Once the plates have been contoured and the holes drilled, they are removed, and the bone cut is made.
The surgeon then distracts the cut ends of the mandible to gain access to the oral cavity and proceeds to divide the FOM mucosa and mylohyoid muscle about 1 cm from, and parallel to, the mandible, so as to leave soft tissue on the mandible for FOM repair at conclusion of the surgery. Continue the incision posteriorly along the FOM until the tumor comes into view; the lingual artery (medial to the hyoglossus muscle) and the XIIn course medial to the FOM incision and are not at risk of injury at this stage of the dissection.
The tumor is resected using electrocautery. Posteriorly the lingual nerve extends from the skull base, passes behind medial pterygoid and crosses the incision line from lateral to medial to course along the lateral FOM; preserve the nerve if possible.
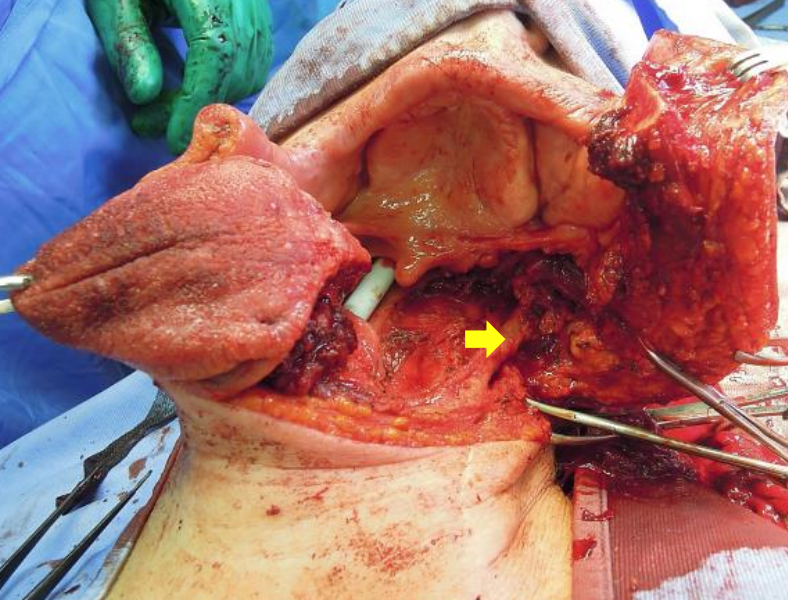
Figure 30: Note the excellent access afforded by mandibulotomy approach (Arrow points to internal carotid artery)
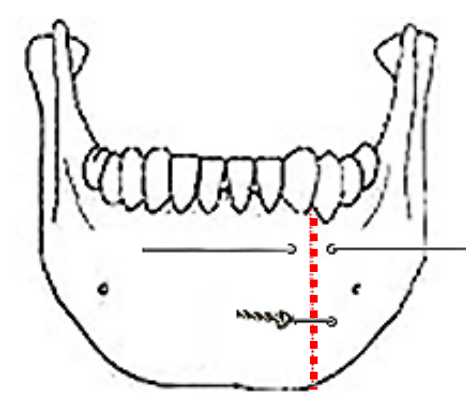
Figure 31: Mandible wired together with stainless steel wire
At the conclusion of surgery, the FOM is closed with a running vicryl suture, and the osteotomy is plated; when plating sets are not available, opposing holes are drilled on either side of the osteotomy and the mandible is wired together with stainless steel wires (Figure 31).
Marginal and segmental mandibulectomy
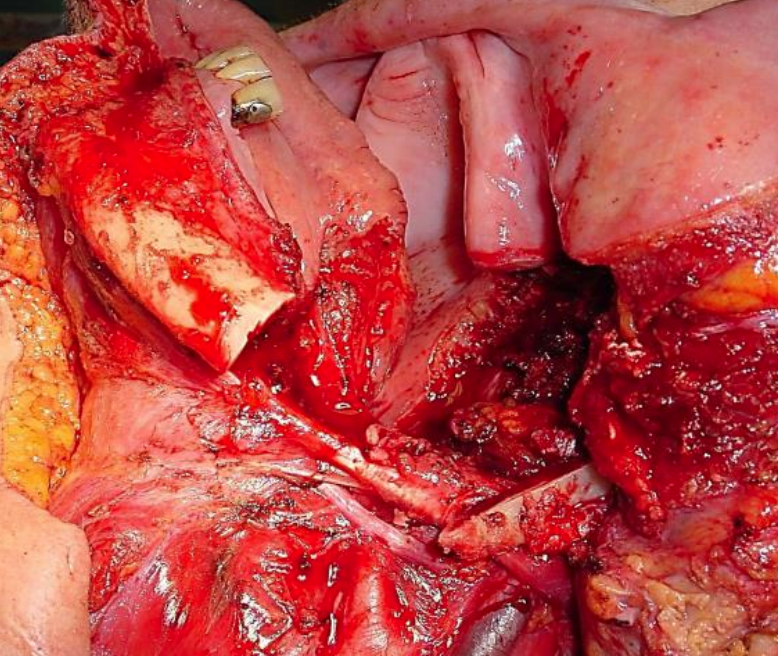
Figure 33: Segmental mandibulectomy preserving only posterior rim of vertical ramus (note preplating holes)
Marginal mandibulectomy is done for access, for securing adequate oncologic resection margins, or to facilitate mucosal repair by suturing gingivolabial to gingivobuccal mucosa (Figure 32). Segmental mandibulectomy is done to secure adequate oncologic margins (Figure 33).
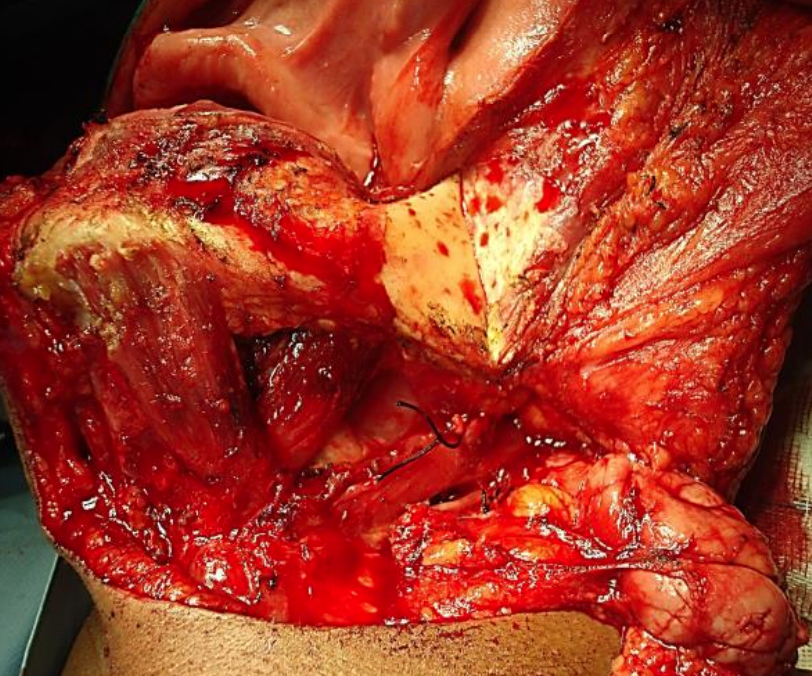
Figure 34: Stripping masseter off bone
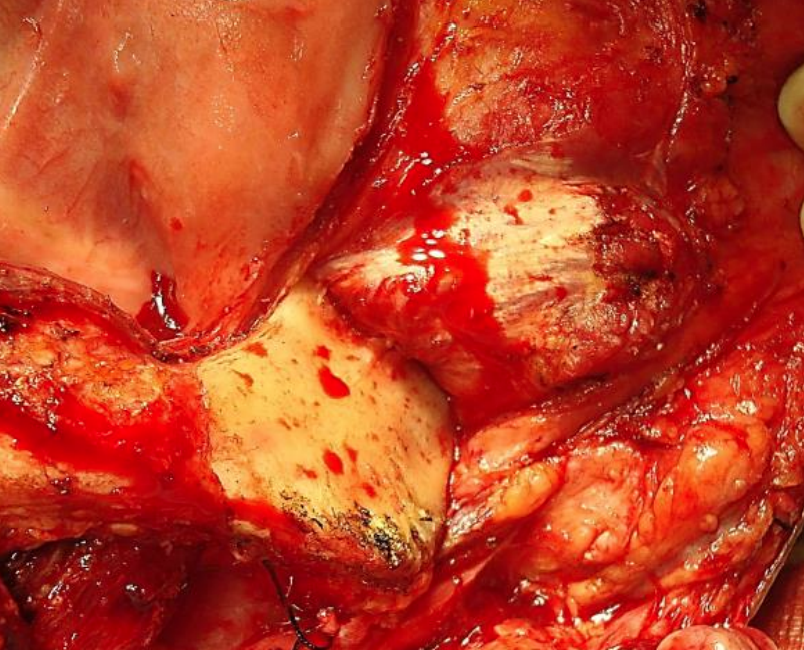
Figure 35: Exposing vertical ramus of mandible up to coronoid process, mandibular notch and neck of condyle
The initial surgical steps are as described for paramedian mandibulotomy (up to Figure 26). Transect the mental nerve and artery with cautery and continue to strip soft tissue off the lateral aspect of the mandible up to the masseter muscle; then strip the masseter off its mandibular insertion using electrocautery (Figures 34, 35).
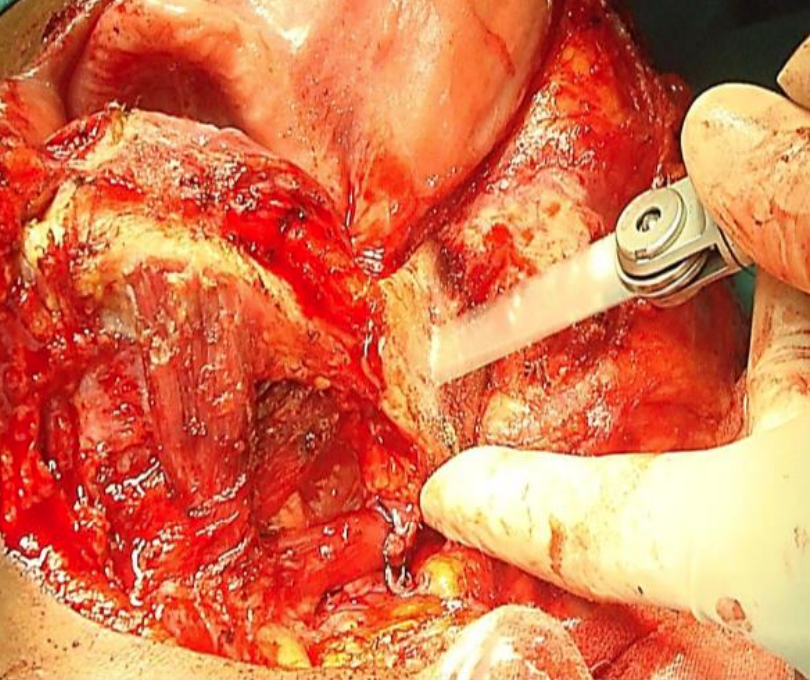
Figure 36: Osteotomy with oscillating saw
Inspect the tumor, and decide as to whether marginal, segmental or hemimandibulectomy is required to obtain clear margins and then plan the osteotomies. Use an oscillating/reciprocating saw or a Gigli saw for the osteotomies (Figure 36). Beware of the close proximity of the external carotid artery and its branches to the vertical ramus when making the osteotomies (Figures 5, 11).
With segmental mandibulectomy in dentate patients one should preplate the mandible with a reconstruction plate to ensure accurate repair and perfect dental occlusion.
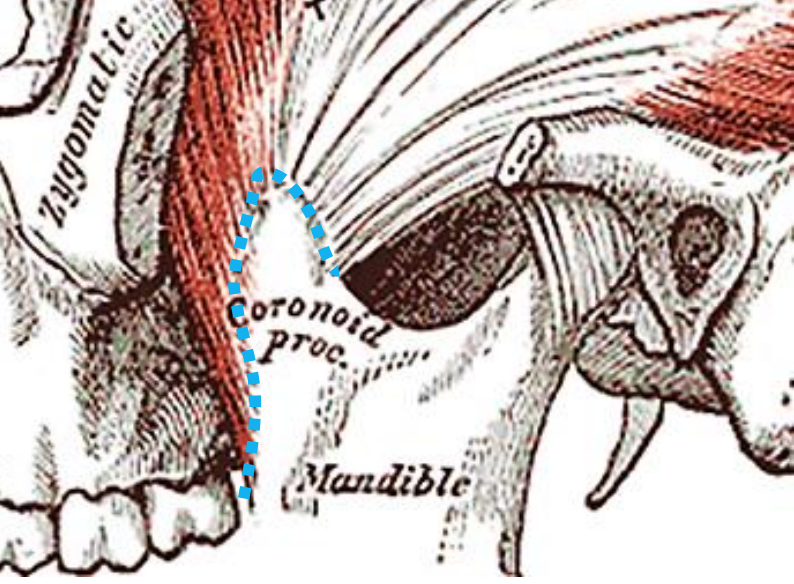
Figure 37: Broken blue line indicates where insertion of temporalis muscle is freed with cautery
To resect the coronoid process, strip the masseter off the entire length of the vertical ramus, identify the mandibular notch, and while pulling caudad on the mandible, free the insertion of the temporalis muscle from the coronoid with cautery, keeping close to bone throughout to avoid blood vessels situated medially; this step is simpler if the distal osteotomy has already been done (Figure 37).
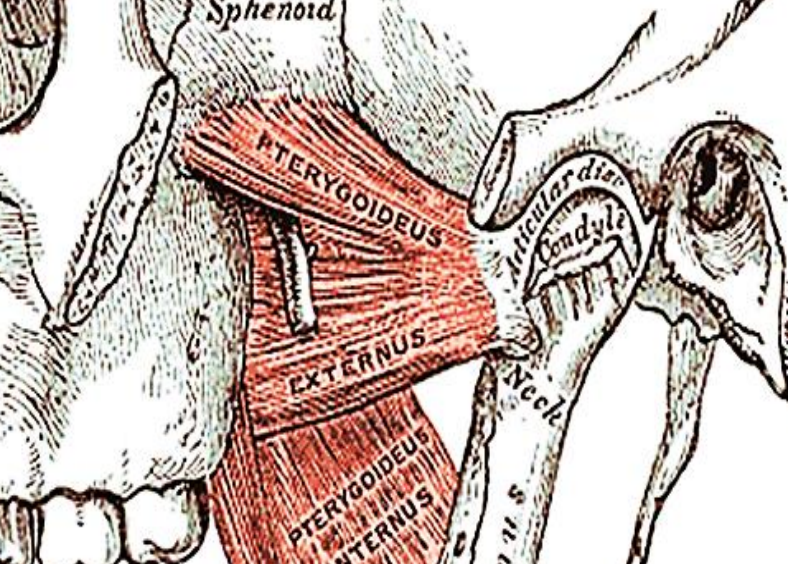
Figure 38: To resect the condyle, transect the lateral pterygoid at its insertion onto the condylar neck and the joint capsule
It is generally not necessary to resect the condyle; rather carefully transect the condylar neck bearing in mind the proximity of the internal carotid artery. To resect the condyle, transect the lateral pterygoid at its insertion onto the condylar neck and the joint capsule (Figure 38).
Tumor resection
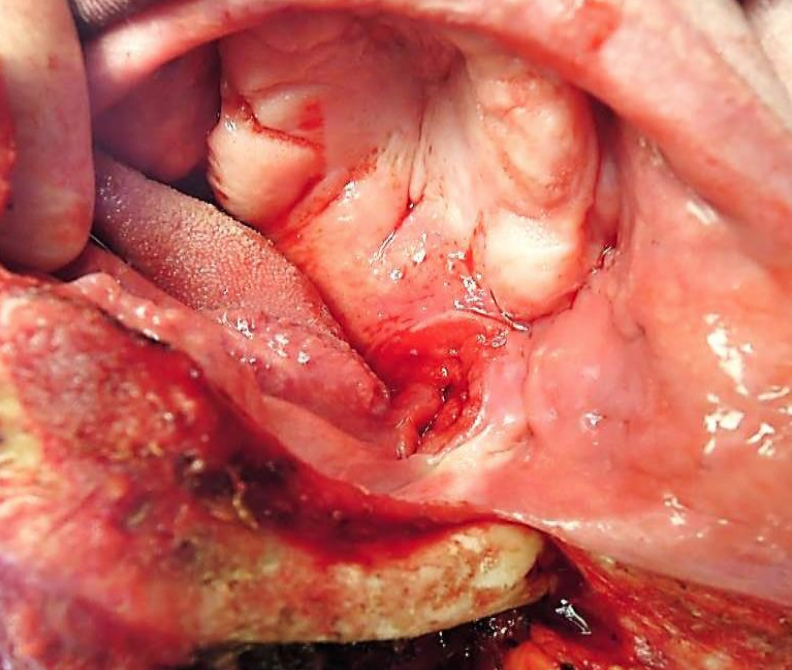
Figure 39a: Cancer of the tonsil fossa, soft palate & BOT fully exposed
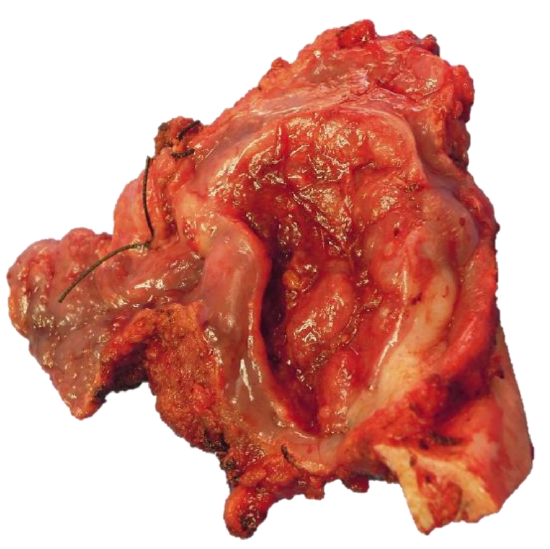
Figure 39b: Marginal mandibulectomy with cancer attached
With adequate exposure, resecting the tumor is generally quite straightforward using electrocautery (Figures 39 a, b). The surgeon must remain aware of the locations of the internal and external carotid arteries; palpating the styloid process is useful as the internal carotid artery lies medial to the bone.
Orientate the tumor for the pathologist with a silk suture (Figure 39b); obtain frozen section of the margins if available.
Repair / Reconstruction
Obtain meticulous hemostasis using ties, monopolar and bipolar cautery before closing the defect.
The lip is carefully repaired in layers to approximate the muscles as well as mucosa and skin.
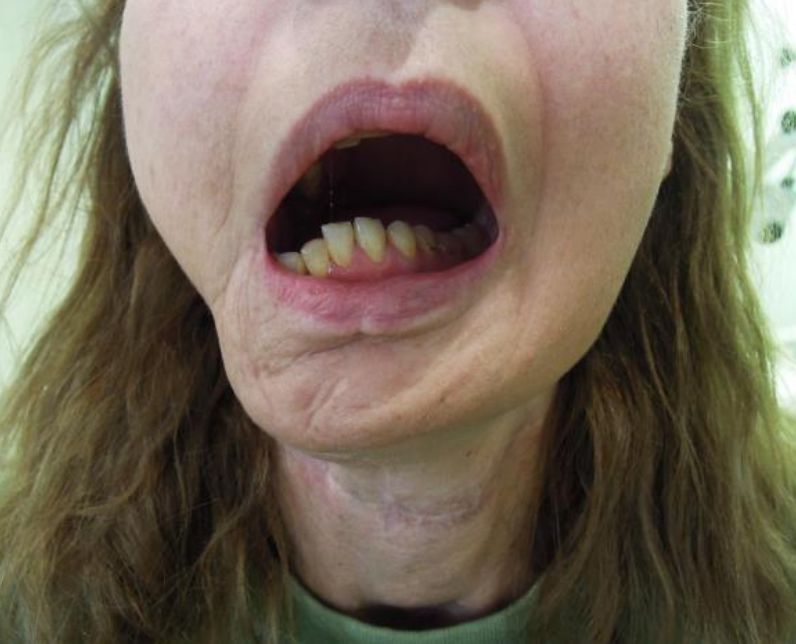
Figure 40: Missing lateral mandibular segment causes mandible to “swing” towards the side of the resection
The pharynx is a complex functional unit. Whether and how to repair or reconstruct oropharyngeal defects requires that a surgeon critically follows his/her patients and learns from successes and failures. Poor repairs and reconstructions cause hypernasal speech and nasal regurgitation. Failure to reconstruct a lateral mandibular segmental defect (allowing mandible to “swing”) may have significant cosmetic and functional consequences (Figure 40).
Healing by secondary intention is not utilized frequently enough with BOT resections, but is of course commonly employed with tonsillectomy, TLM and TORS.
Primary closure of BOT defects yields the best swallowing results, although inadequate bulk impairs speech if the BOT cannot approximate the soft palate. Primary closure of palatal defects may cause hypernasal speech and nasal regurgitation.
Pedicled and free tissue transfer flaps may be used to reconstruct oropharyngeal defects. Simply shaping a flap to match the resected tissue may well restore form yet have a poor functional result. Therefore, one must carefully assess the defect to determine how best to optimize function.
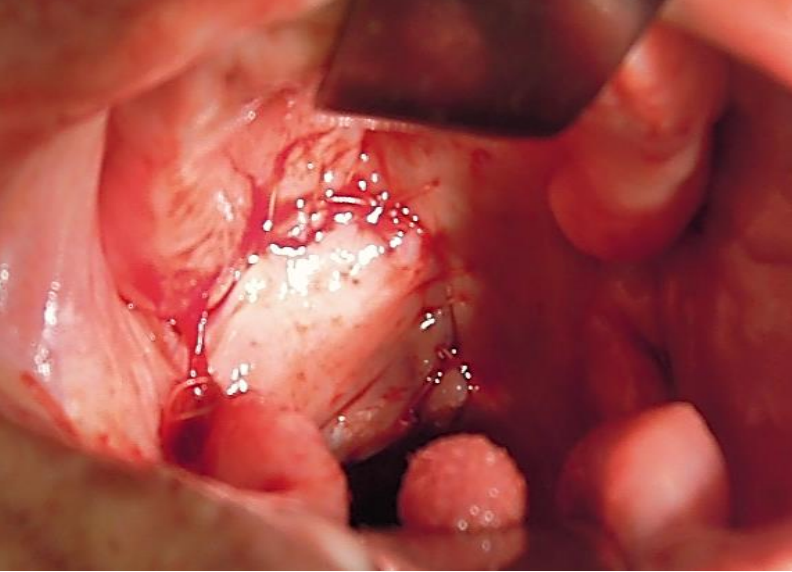
Figure 41: Buccinator myomucosal flap used to restore soft palate and tonsil fossa defects
Buccinator myomucosal flap: (See chapter: Buccinator myomucosal flap) A posteriorly-based buccinator flap can be used to repair defects of the soft palate, lateral pharyngeal wall and BOT. It is an excellent choice as it has the same physical qualities as tissues of the oropharynx, has a reasonable amount of bulk, and preserves mobility (Figure 41).
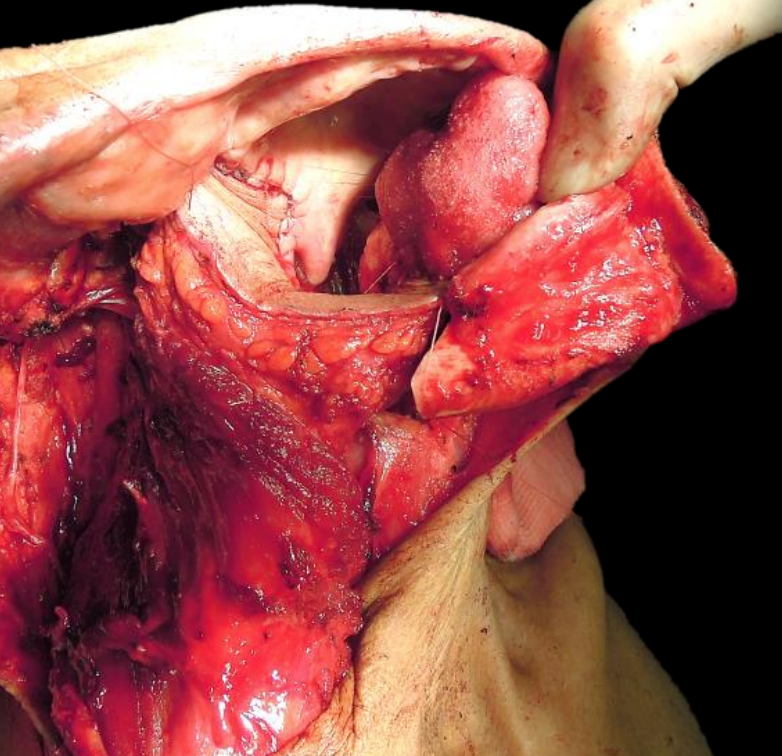
Figure 42: Pectoralis major flap used to reconstruct an oropharyngeal defect and to fill in the bony defect of a segmental mandibulectomy
Pectoralis major flap: (See chapter: Pectoralis major flap) Although this is a reasonable option to use, it may drag down on the soft palate causing palatal incompetence. It is a useful soft tissue filler of a lateral segmental mandibular defect (Figure 42).
Buccal fat pad flap: (See chapter: Buccal fat pad flap) This is a useful flap to cover exposed mandible or maxillary bone, or to augment a thinned out soft palate, or to close a small soft palate defect.
Temporalis muscle flap: (See chapter: Temporalis muscle flap) This is commonly used in some centers for oropharyngeal defects. The muscle is pedicled on the coronoid process which is immediately adjacent to the oropharynx. It requires that the deep temporal arteries be preserved.
Other pedicled flaps to consider are the Submental artery island flap and Supraclavicular flap
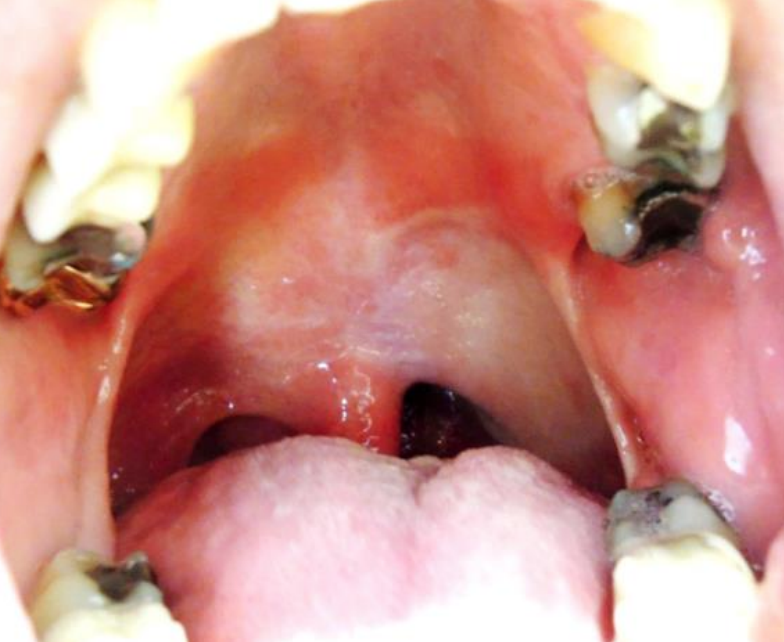
Figure 43: Soft palate and lateral pharyngeal wall reconstruction with RFFF
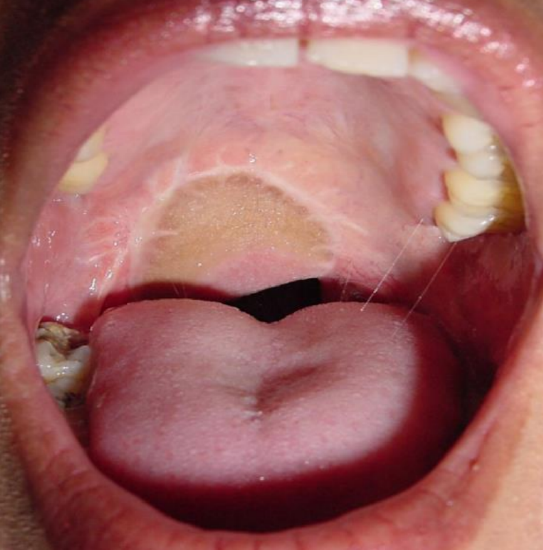
Figure 44: Soft palate reconstruction with RFFF
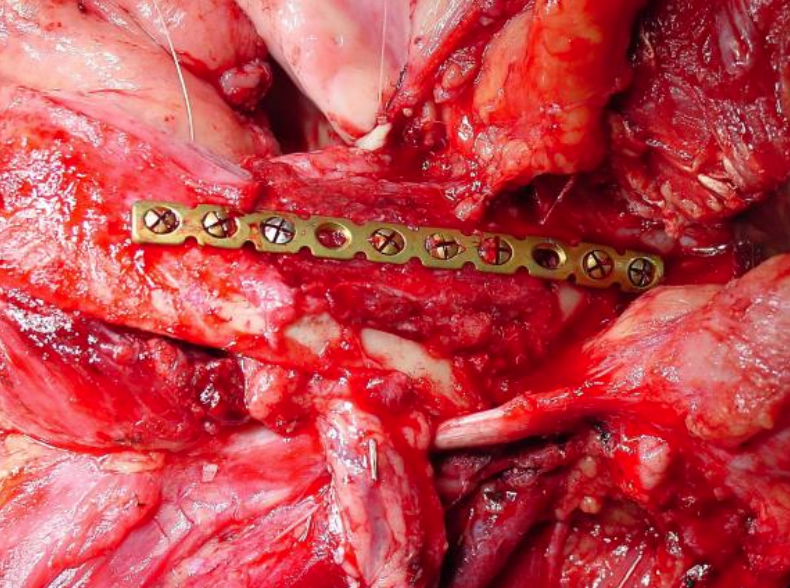
Figure 45: Marginal mandibulectomy with onlay radial osseocutaneous flap
Radial free forearm flap (RFFF): (See chapter: Radial free forearm flap) This is a thin, pliable flap and is therefore an excellent option for oropharyngeal repair (Figures 43-45). It can also be used as an onlay osseocutaneous flap following marginal mandibulectomy.
Anterolateral free thigh flap: (See chapter: Anterolateral free thigh flap) Muscle harvested with the flap can be tailored to conform to the volume of the defect to be filled. However, it is less pliable than the radial free forearm flap and is therefore less suited to soft palate reconstruction.
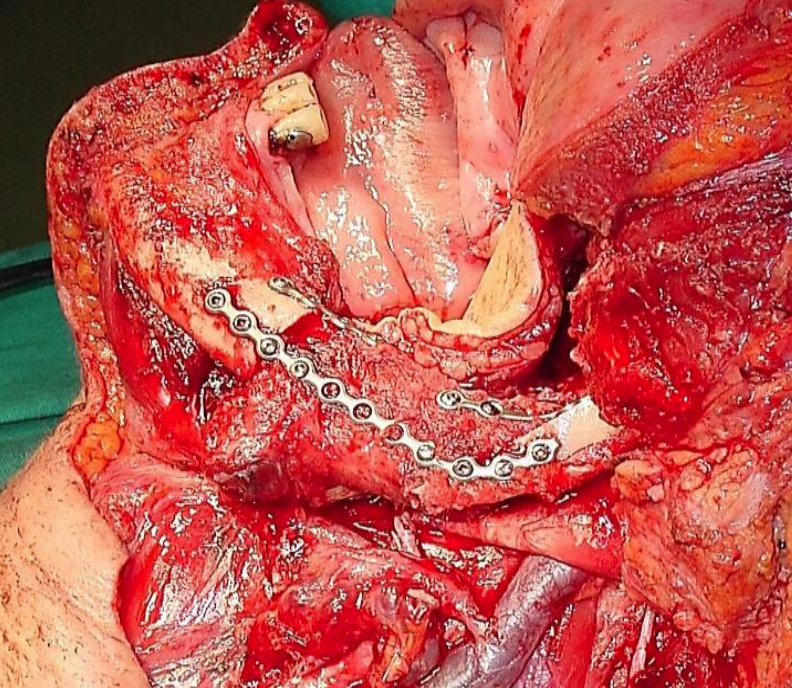
Figure 46: FFF graft following segmental mandibulectomy; skin of leg used to reconstruct oropharynx defect
Free fibula flap (FFF): (See chapter: Vascularized free fibula flap). This is the workhorse flap for patients requiring reconstruction of mandible (Figure 46).
Postoperative management
- Antibiotics are administered for 24hrs
- A temporary tracheostomy is done; once the airway appears adequate, an uncuffed tracheostomy tube is inserted, the tracheostomy tube is corked for 24hrs and if tolerated, is removed
- A nasogastric feeding tube is inserted; oral feeding is commenced on Day 5
- Suction drains are removed when drainage is <50mL/24hrs
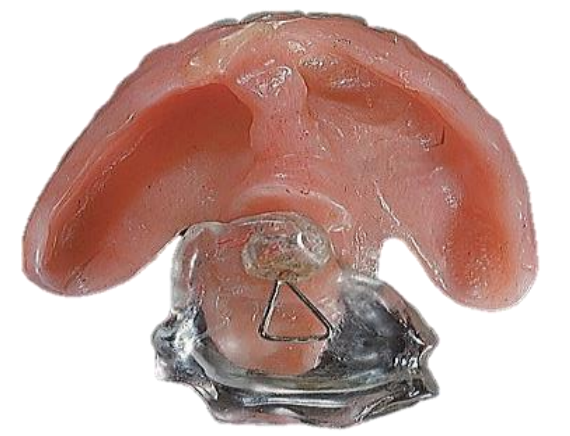
Figure 47: Palatal prosthesis
- Prosthetic devices can be used to correct palatal incompetence (Figure 47)
- Dental rehabilitation may involve new dentures or implants (Figures 48a, b)
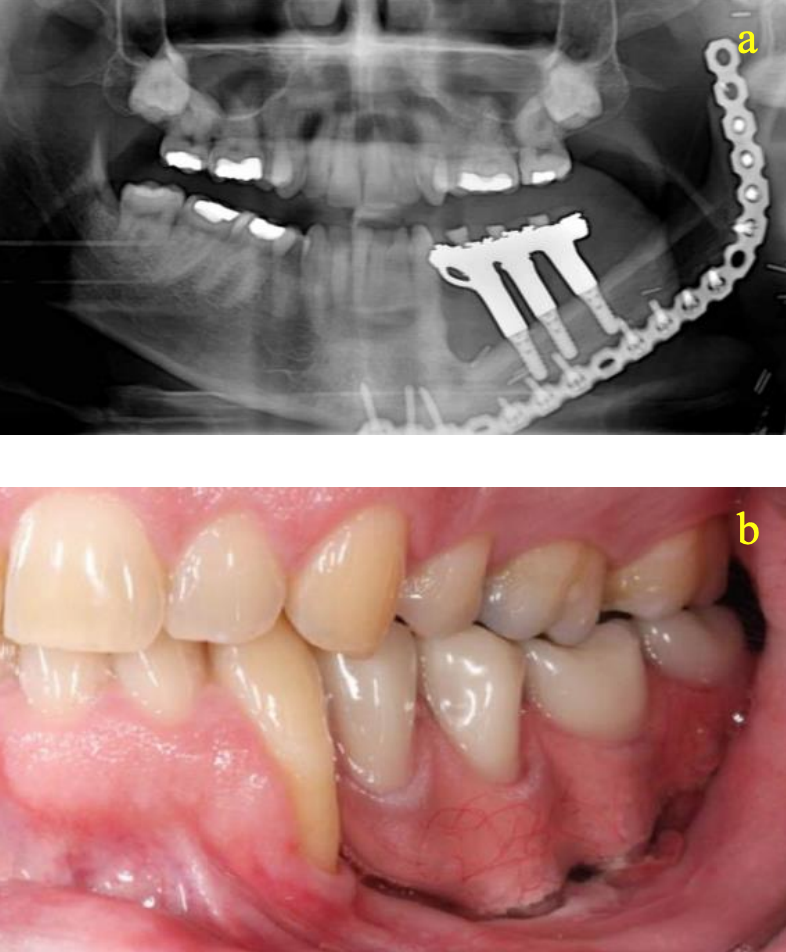
Figures 48a, b: Dental implants and rehabilitation following FFF reconstruction
Final comments
Resecting tumors of the oropharynx is challenging particularly in terms of access and optimizing swallowing and speech. One should not compromise resection margins for function. The surgical team must master an array of surgical approaches and reconstructive techniques to ensure the best oncological and functional outcomes.
Author and Editor
Johan Fagan MBChB, FCS(ORL), MMed
Professor and Chairman
Division of Otolaryngology
University of Cape Town
Cape Town, South Africa
johannes.fagan@uct.ac.za
Author
Eugene N Myers MD, FACS, FRCS Edin (Hon)
Distinguished Professor & Emeritus Chair
Department of Otolaryngology Eye and Ear Institute, Suite 519
200 Lothrop Street
Pittsburgh, PA 15213, USA
myersen@upmc.edu


