20.1E: Antimicrobial Proteins
- Page ID
- 7942
Antimicrobial peptides are an evolutionarily-conserved component of the innate immune response found among all classes of life.
- Describe the role of antimicrobial peptides in the innate immune system
Key Points
- Antimicrobial peptides include a net positive charge and hydrophilic and hydrophobic ends, which allow them to adhere to the lipid membranes of bacterial cell membranes.
- The positive charge makes antimicrobial peptides selective, so they only adhere to negatively-charged bacterial cell membranes instead of host cell membranes.
- The modes of action by which antimicrobial peptides kill bacteria are varied and include disrupting membranes, interfering with metabolism, and targeting cytoplasmic components.
- Many bacteria have developed antimicrobial resistance, in which a component of their cell membranes or enzyme secretions is altered to prevent the peptides from binding to the bacteria, or by inhibiting the peptides directly.
Key Terms
- antimicrobial resistance: Any mechanism that enables bacteria to evade or inhibit antimicrobial action.
- peptide: A class of organic compounds consisting of various numbers of amino acids in which the amine of one is reacted with the carboxylic acid of the next to form anamide bond.
- amphipathic: A molecule with both hydrophobic and hydrophilic groups that allow it to adhere to lipid structures more easily.
Antimicrobial peptides (also called host defense peptides) are an evolutionarily-conserved component of the innate immune response found among all known species. These peptides are found in many of the mucus membranes across the human body and are therefore considered to be part of the barrier immune system. The function of these peptides is to kill microbial pathogens and prevent them from entering the body.
Structure
Antimicrobial peptides are a unique and diverse group of molecules. As peptides, they consist of chains of amino acids that determine their composition and structure. These peptides have a stronger positive than negative charge, which is an important component of their selectivity. They also include hydrophobic and hydrophilic groups that enable them to latch onto other molecules (often lipids) through intermolecular forces, such as the lipid bilayer that forms cell membranes.
The secondary structures of these molecules follow four themes, including i) α-helical, ii) β-stranded due to the presence of two or more disulfide bonds, iii) β-hairpin or loop due to the presence of a single disulfide bond and/or cyclization of the peptide chain, and iv) extended. Many of these peptides are unstructured and inactive in free solution, and fold into their final configuration upon reaching mucus membranes. The amphipathicity (hydrophilic and hydrophobic ends) and positive charge of peptides are their defining structural features.
Antimicrobial Action
The modes of action by which antimicrobial peptides kill bacteria are varied and include disrupting cell membranes, interfering with metabolism, and damaging organelles. The initial contact between the peptide and target organism is electrostatic due to the force of negative and positive ionic charge. The amino acid composition, amphipathicity, cationic charge, and size allow them to attach to and insert into membrane bilayers to form pores by barrel-stave, carpet, or toroidal-pore mechanisms.
The peptides are selective and thus more likely to adhere to bacterial cell membranes than to cell membranes of the host cells. The peptides have a greater positive charge than negative charge, while bacterial cell membranes have a greater negative charge than host cell membranes. This causes the peptide to bind to bacterial membranes instead of host cell membranes.
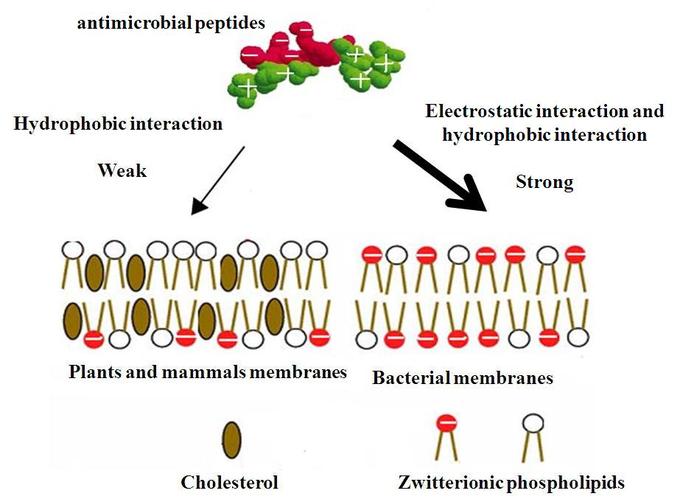
Mechanism of Selectivity of Antimicrobial Peptides: Cell membranes of bacteria are different from the cell membranes of plants and animals and are preferentially targeted by the antimicrobial proteins.
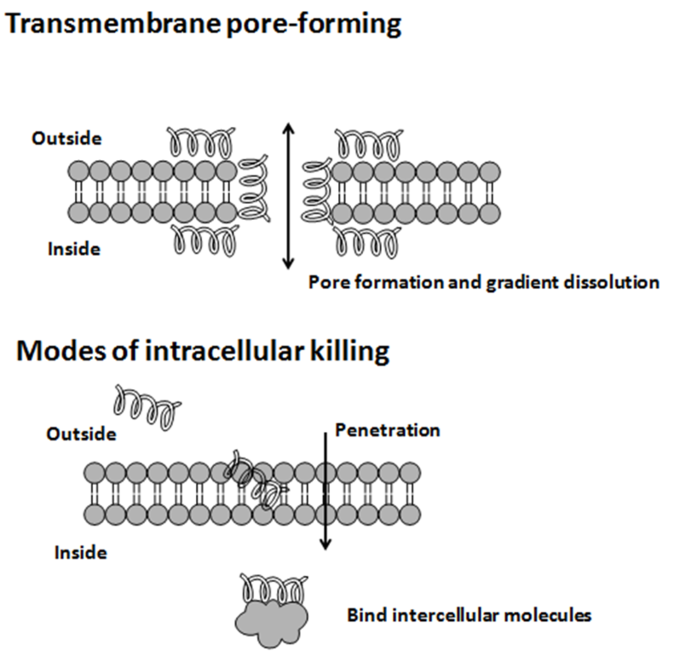
Modes of Action by Antimicrobial Peptides: Antimicrobial peptides multiple various modes of action.
Other antimicrobial mechanisms include intracellular binding models. These involve inhibition of cell wall synthesis, alteration of the cytoplasmic membrane, activation of autolysin, inhibition of DNA, RNA, and protein synthesis, and inhibition of certain bacterial enzymes. However, in many cases, the exact killing mechanism is unknown. In general, the antimicrobial activity of these peptides is determined by measuring the minimal inhibitory concentration (MIC), the lowest concentration of drug that inhibits bacterial growth and an indicator of the antimicrobial strength of that peptide.
Antimicrobial Resistance
Despite the efficiency of antimicrobial peptides to inhibit and kill bacteria, they are still able to get inside the body and cause infections. Bacteria can develop resistance to antimicrobial peptides (as well as separate resistances to antibiotics and other antimicriobials). Bacteria like staphylococcus aureas, which forms the highly resistant MRSA strain, can reduce the negativity of the charge of its cell membrane by bringing amino acids from the cytoplasm into its cell membrane so antimicrobial peptides won’t bind to it. Other forms of antimicrobial resistance include producing enzymes that inhibit the antimicrobial peptides, altering the hydrophobic forces on the cell membrane, and capturing antimicrobial peptides in vesicles on the cell membrane to remove them from the bacterium.
Additionally, commensal bacteria have developed antimicrobial resistance to peptides, but they are normal flora of the body. Most never act as pathogens, though some may be opportunistic pathogens or only act as pathogens in people with certain genetic characteristics.

