20.3C: Maturation of T Cells
- Page ID
- 7947
T cells originate from hematopoietic stem cells in the bone marrow and undergo positive and negative selection in the thymus to mature.
- Describe T cell maturation
Key Points
- Hematopoietic progenitors derived from hematopoietic stem cells populate the thymus and expand by cell division to generate a large population of immature thymocytes. After the thymus becomes inactive later in life, existing immature T cells will proliferate through clonal expansion.
- About 98% of thymocytes die during the development processes in the thymus by failing either positive selection or negative selection, while the other 2% survive and leave the thymus to become mature immunocompetent T cells.
- During positive selection, only T cells that can bind to MHC are kept alive. The rest are killed by an apoptotic signal so that non-functional T cells don’t get into the body and crowd out functional T cells.
- During negative selection, most T cells that bind too easily to self antigens are killed. Some are kept alive and differentiate into T reg cells, which help prevent overactive cell mediated immune responses.
- Autoimmune diseases may be caused either by antibodies or T cells that can bind to self antigens, causing damage to self organs and tissues. They may be caused by failed negative selection and often have a genetic component.
Key Terms
- thymus: A ductless gland consisting mainly of primary lymphatic tissue that is the site of lymphocyte maturation and selection.
- negative selection: The process by which T cells are screened so that those with a high affinity for binding to self antigens (and potentially causing autoimmunity) are destroyed.
- Positive selection: The process by which T cells are screened so that only those capable of binding to MHC are kept alive.
T cells belong to a group of white blood cells known as lymphocytes and play a central role in the cell-mediated branch of the adaptive immune system. They are distinguished from other lymphocytes, such as B cells and natural killer cells (NK cells), by the presence of a T cell receptor (TCR) on the cell surface. T cells are produced in the bone marrow but travel to the thymus to mature. T cells can be either helper T cells or cytoxic T cells based on whether they express CD4 (helper) or CD8 ( cytotoxic ) glycoprotein.
Maturation of T Cells
All T cells originate from hematopoietic stem cells in the bone marrow, which are capable of differentiating into any type of white blood cell. Immature T cells that migrate to the thymus are called thymocytes. The earliest thymocytes express neither CD4 nor CD8, and are therefore classed as double-negative (CD4-CD8-) cells. As they progress through their development they become double-positive thymocytes (CD4+CD8+) and finally mature to single-positive (CD4+CD8- or CD4-CD8+) thymocytes that are released from the thymus to peripheral tissues. Typically, these mature thymocytes are still referred to as either “immature” or “naive” because they have not been presented with an antigen. They travel to sites that contain secondary lymphoid tissue, such as the lymph nodes and tonsils, where antigen presentation. This facilitates the development of antigen-specific adaptive immunity.
The thymus contributes fewer cells as a person ages. As its functional mass shrinks by about 3% a year throughout middle age, there is a corresponding fall in the thymic production of naive T cells, leaving clonal expansion of immature T cells to play a greater role in protecting older subjects. The thymus is thus thought to be important in building a large stock of naive T cells soon after birth that can later function without thymus support.
Positive Selection of T Cells
During thymocyte maturation, 98% of T cells are discarded by selection, thich is a mechanism designed to ensure that T cells function without major problems. Positive selection designates T cells capable of interacting with MHC. Double-positive thymocytes (CD4+/CD8+) move deep into the thymic cortex tissue where they are presented with self-antigens. These are expressed by thymic cortical epithelial cells that express both MHC I and MHC II molecules on the surface of cortical cells. Only those thymocytes that interact with MHC I or MHC II will receive a vital “survival signal.” Thosethat can’t interact will undergo apoptosis (cell death). This insures T cell functionality since T cells with non-functional receptors cannot receive antigens and are thus useless to the immune system. If non-functional T cells were allowed into circulation, they would crowd out functional T cells and slow down the rate at which adaptive immune responses are formed. The vast majority of thymocytes die during this process.
A thymocyte’s differentiation into either a helper or cytotoxic version is also determined during positive selection. Double-positive cells (CD4+/CD8+) that are positively selected on MHC class II molecules will eventually become CD4+ helper T cells, while cells positively selected on MHC class I molecules mature into CD8+ cytotoxic T cells. A T cell is then signaled by the thymus to become a CD4+ cell by reducing expression of its CD8 cell surface receptors. If the cell does not lose its signal, it will continue reducing CD8 and become a CD4+, single positive cell. But if there is a signal interruption, it will instead reduce CD4 molecules, eventually becoming a CD8+, single positive cell. This process does not remove thymocytes that may become sensitized against self-antigens, which causes autoimmunity. The potentially autoimmune cells are removed by the process of negative selection.
Negative Selection of T Cells
Negative selection removes thymocytes that are capable of strongly binding with self-antigens presented by MHC. Thymocytes that survive positive selection migrate towards the boundary of the thymic cortex and thymic medulla (the part of the thymus where T cells enter circulation). While in the medulla, they are again presented with self-antigen in complex with MHC molecules on thymic epithelial cells. Thymocytes that interact too strongly with the antigen receive an apoptotic signal that leads to cell death.
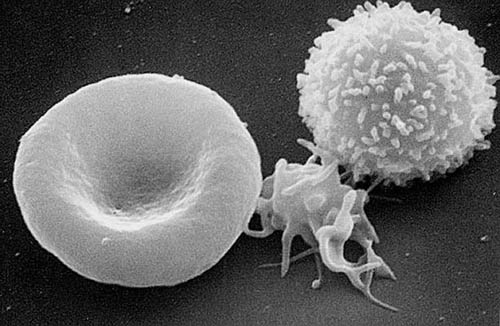
Blood cells: Scanning electron micrograph of T lymphocyte (right), a platelet (center), and a red blood cell (left).
However, some cells are selected to become T-reg cells, which retain their ability to bind to self-antigens in order to suppress overactive immune responses. These cells may be protective against autoimmunity. The remaining cells exit the thymus as mature naive T cells. This process is an important component of central tolerance, a process that prevents the formation of self-reactive T cells that are capable of inducing autoimmune diseases in the host. Autoimmune diseases reflect a loss of central tolerance in which the body’s own B and T cells become sensitized towards self-antigens. Many autoimmune disorders are primarily antibody-mediated, but some are T cell mediated. One example of the latter is Crohn’s disease, in which T cells attack the colon. These autoimmune disorders may be caused by problems in negative selection and tend to have genetic components.

