15.2: Basic Fluid and Electrolyte Concepts
- Page ID
- 52497
Before learning about how to care for patients with fluid and electrolyte imbalances, it is important to understand the physiological processes of the body’s regulatory mechanisms. The body is in a constant state of change as fluids and electrolytes are shifted in and out of cells within the body in an attempt to maintain a nearly perfect balance. A slight change in either direction can have significant consequences on various body systems.
Body Fluids
Body fluids consist of water, electrolytes, blood plasma and component cells, proteins, and other soluble particles called solutes. Body fluids are found in two main areas of the body called intracellular and extracellular compartments. See Figure 15.1[1] for an illustration of intracellular and extracellular compartments.
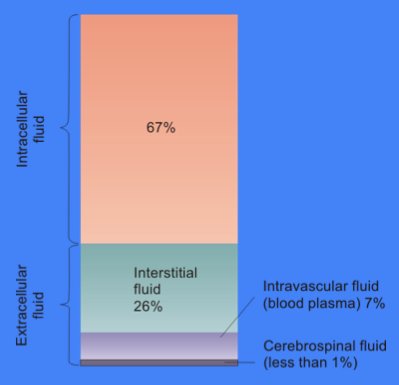
Intracellular fluids (ICF) are found inside cells and are made up of protein, water, electrolytes, and solutes. The most abundant electrolyte in intracellular fluid is potassium. Intracellular fluids are crucial to the body’s functioning. In fact, intracellular fluid accounts for 60% of the volume of body fluids and 40% of a person’s total body weight![2]
Extracellular fluids (ECF) are fluids found outside of cells. The most abundant electrolyte in extracellular fluid is sodium. The body regulates sodium levels to control the movement of water into and out of the extracellular space due to osmosis.
Extracellular fluids can be further broken down into various types. The first type is known as intravascular fluid that is found in the vascular system that consists of arteries, veins, and capillary networks. Intravascular fluid is whole blood volume and also includes red blood cells, white blood cells, plasma, and platelets. Intravascular fluid is the most important component of the body’s overall fluid balance.
Loss of intravascular fluids causes the nursing diagnosis Deficient Fluid Volume, also referred to as hypovolemia. Intravascular fluid loss can be caused by several factors, such as excessive diuretic use, severe bleeding, vomiting, diarrhea, and inadequate oral fluid intake. If intravascular fluid loss is severe, the body cannot maintain adequate blood pressure and perfusion of vital organs. This can result in hypovolemic shock and cellular death when critical organs do not receive an oxygen-rich blood supply needed to perform cellular function.
A second type of extracellular fluid is interstitial fluid that refers to fluid outside of blood vessels and between the cells. For example, if you have ever cared for a patient with heart failure and noticed increased swelling in the feet and ankles, you have seen an example of excess interstitial fluid referred to as edema.
The remaining extracellular fluid, also called transcellular fluid, refers to fluid in areas such as cerebrospinal, synovial, intrapleural, and gastrointestinal system.[3]
Fluid Movement
Fluid movement occurs inside the body due to osmotic pressure, hydrostatic pressure, and osmosis. Proper fluid movement depends on intact and properly functioning vascular tissue lining, normal levels of protein content within the blood, and adequate hydrostatic pressures inside the blood vessels. Intact vascular tissue lining prevents fluid from leaking out of the blood vessels. Protein content of the blood (in the form of albumin) causes oncotic pressure that holds water inside the vascular compartment. For example, patients with decreased protein levels (i.e., low serum albumin) experience edema due to the leakage of intravascular fluid into interstitial areas because of decreased oncotic pressure.
Hydrostatic pressure is defined as pressure that a contained fluid exerts on what is confining it. In the intravascular fluid compartment, hydrostatic pressure is the pressure exerted by blood against the capillaries. Hydrostatic pressure opposes oncotic pressure at the arterial end of capillaries, where it pushes fluid and solutes out into the interstitial compartment. On the venous end of the capillary, hydrostatic pressure is reduced, which allows oncotic pressure to pull fluids and solutes back into the capillary.[4],[5] See Figure 15.2[6] for an illustration of hydrostatic pressure and oncotic pressure in a capillary.
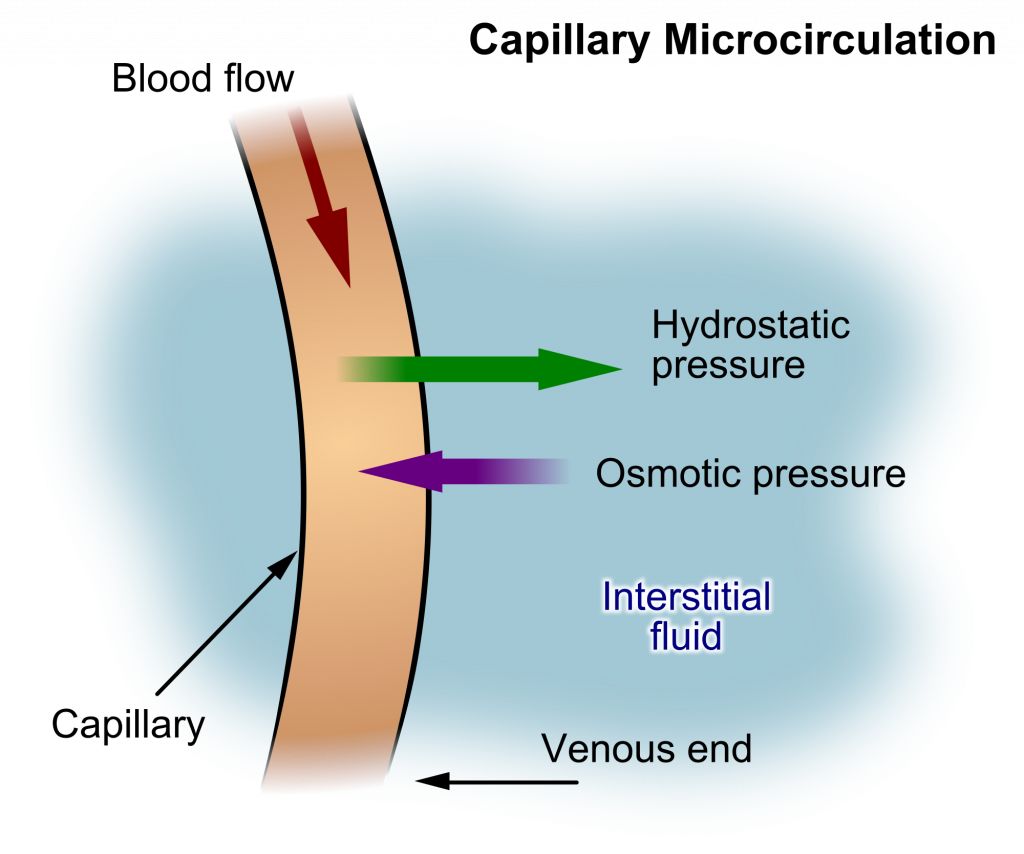
Filtration occurs when hydrostatic pressure pushes fluids and solutes through a permeable membrane so they can be excreted. An example of this process is fluid and waste filtration through the glomerular capillaries in the kidneys. This filtration process within the kidneys allows excess fluid and waste products to be excreted from the body in the form of urine.
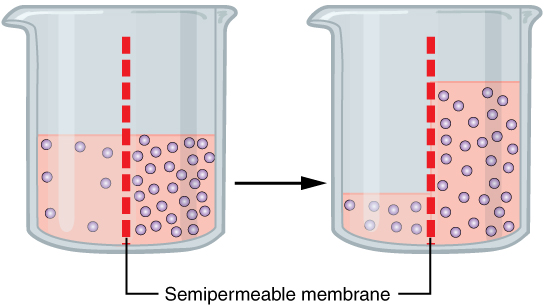
Fluid movement is also controlled through osmosis. Osmosis is water movement through a semipermeable membrane, from an area of lesser solute concentration to an area of greater solute concentration, in an attempt to equalize the solute concentrations on either side of the membrane. Only fluids and some particles dissolved in the fluid are able to pass through a semipermeable membrane; larger particles are blocked from getting through. Because osmosis causes fluid to travel due to a concentration gradient and no energy is expended during the process, it is referred to as passive transport.[7] See Figure 15.3[8] for an illustration of osmosis where water has moved to the right side of the membrane to equalize the concentration of solutes on that side with the left side.
Osmosis causes fluid movement between the intravascular, interstitial, and intracellular fluid compartments based on solute concentration. For example, recall a time when you have eaten a large amount of salty foods. The sodium concentration of the blood becomes elevated. Due to the elevated solute concentration within the bloodstream, osmosis causes fluid to be pulled into the intravascular compartment from the interstitial and intracellular compartments to try to equalize the solute concentration. As fluid leaves the cells, they shrink in size. The shrinkage of cells is what causes many symptoms of dehydration, such as dry, sticky mucous membranes. Because the brain cells are especially susceptible to fluid movement due to osmosis, a headache may occur if adequate fluid intake does not occur.
Solute Movement
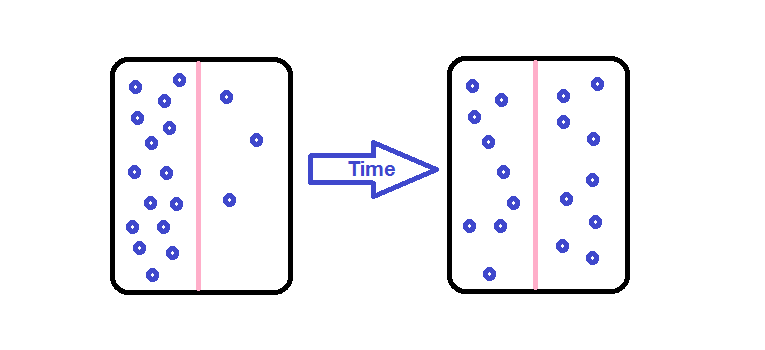
Solute movement is controlled by diffusion, active transport, and filtration. Diffusion is the movement of molecules from an area of higher concentration to an area of lower concentration to equalize the concentration of solutes throughout an area. (Note that diffusion is different from osmosis because osmosis is the movement of fluid whereas diffusion is the movement of solutes.) See Figure 15.4[9] for an image of diffusion. Because diffusion travels down a concentration gradient, the solutes move freely without energy expenditure. An example of diffusion is the movement of inhaled oxygen molecules from alveoli to the capillaries in the lungs so that they can be distributed throughout the body.
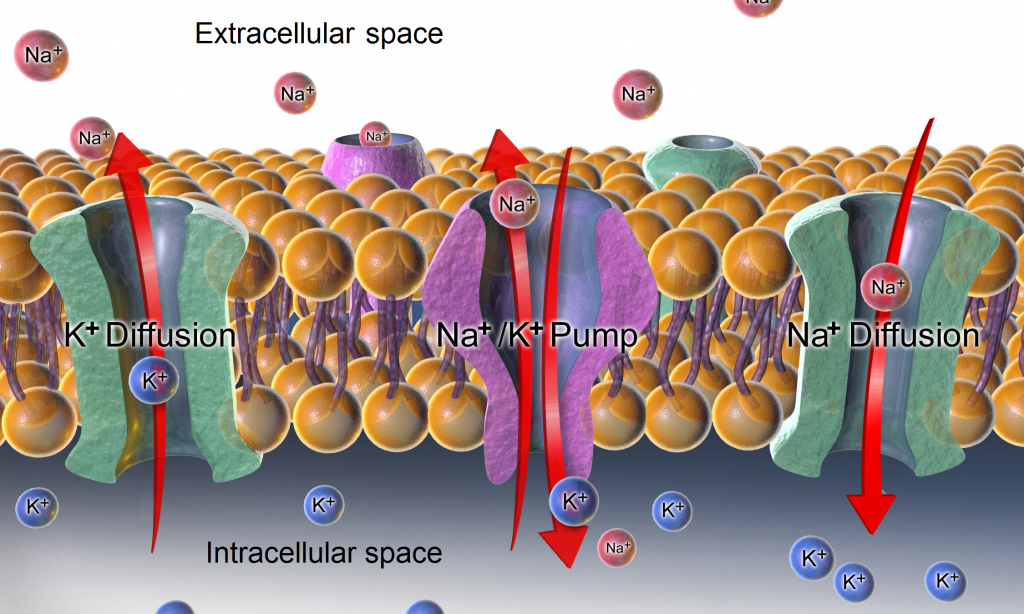
Active transport, unlike diffusion, involves moving solutes and ions across a cell membrane from an area of lower concentration to an area of higher concentration. Because active transport moves solutes against a concentration gradient to prevent an overaccumulation of solutes in an area, energy is required for this process to take place.[10] An example of active transport is the sodium-potassium pump, which uses energy to maintain higher levels of sodium in the extracellular fluid and higher levels of potassium in the intracellular fluid. See Figure 15.5[11] for an image of diffusion and the sodium-potassium pump regulating sodium and potassium levels in the extracellular and intracellular compartments. Recall that sodium (Na+) is the primary electrolyte in the extracellular space and potassium (K+) is the primary electrolyte in the intracellular space.
Fluid and Electrolyte Regulation
The body must carefully regulate intravascular fluid accumulation and excretion to prevent fluid volume excesses or deficits and maintain adequate blood pressure. Water balance is regulated by several mechanisms including ADH, thirst, and the Renin-Angiotensin-Aldosterone System (RAAS).
Fluid intake is regulated by thirst. As fluid is lost and the sodium level increases in the intravascular space, serum osmolality increases. Serum osmolality is a measure of the concentration of dissolved solutes in the blood. Osmoreceptors in the hypothalamus sense increased serum osmolarity levels and trigger the release of ADH (antidiuretic hormone) in the kidneys to retain fluid. The osmoreceptors also produce the feeling of thirst to stimulate increased fluid intake. However, individuals must be able to mentally and physically respond to thirst signals to increase their oral intake. They must be alert, fluids must be accessible, and the person must be strong enough to reach for fluids. When a person is unable to respond to thirst signals, dehydration occurs. Older individuals are at increased risk of dehydration due to age-related impairment in thirst perception. The average adult intake of fluids is about 2,500 mL per day from both food and drink. An increased amount of fluids is needed if the patient has other medical conditions causing excessive fluid loss, such as sweating, fever, vomiting, diarrhea, and bleeding.
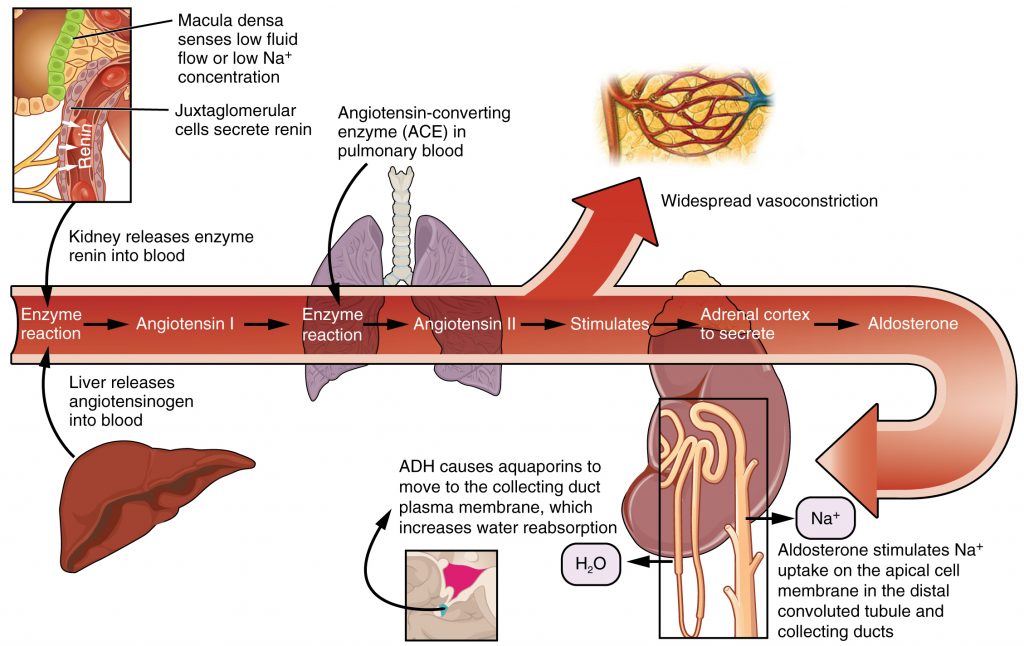
The Renin-Angiotensin-Aldosterone System (RAAS) plays an important role in regulating fluid output and blood pressure. See Figure 15.6[12] for an illustration of the Renin-Angiotensin-Aldosterone System (RAAS). When there is decreased blood pressure (which can be caused by fluid loss), specialized kidney cells make and secrete renin into the bloodstream. Renin acts on angiotensinogen released by the liver and converts it to angiotensin I, which is then converted to angiotensin II. Angiotensin II does a few important things. First, angiotensin II causes vasoconstriction to increase blood flow to vital organs. It also stimulates the adrenal cortex to release aldosterone. Aldosterone is a steroid hormone that triggers increased sodium reabsorption by the kidneys and subsequent increased serum osmolality in the bloodstream. As you recall, increased serum osmolality causes osmosis to move fluid into the intravascular compartment in an effort to equalize solute particles. The increased fluids in the intravascular compartment increase circulating blood volume and help raise the person’s blood pressure. An easy way to remember this physiological process is “aldosterone saves salt” and “water follows salt.”[13]
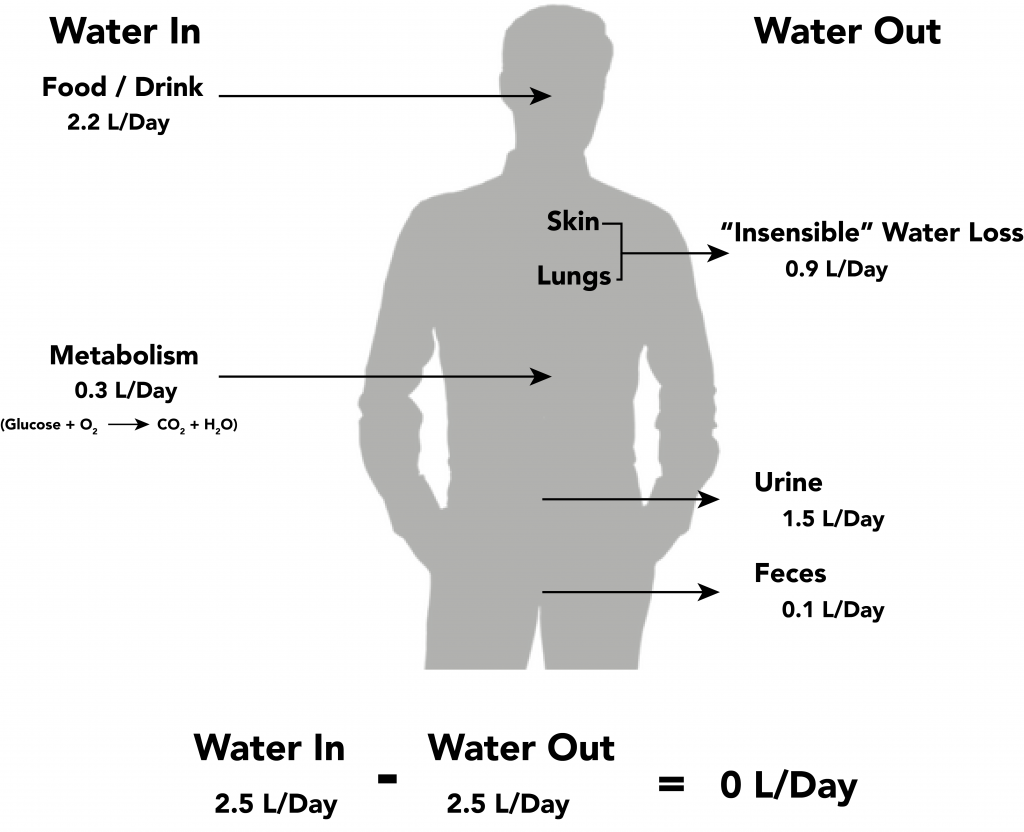
Fluid output occurs mostly through the kidneys in the form of urine. Fluid is also lost through the skin as perspiration, through the gastrointestinal tract in the form of stool, and through the lungs during respiration. Forty percent of daily fluid output occurs due to these “insensible losses” through the skin, gastrointestinal tract, and lungs and cannot be measured. The remaining 60% of daily fluid output is in the form of urine. Normally, the kidneys produce about 1,500 mL of urine per day when fluid intake is adequate. Decreased urine production is an early sign of dehydration or kidney dysfunction. It is important for nurses to assess urine output in patients at risk. If a patient demonstrates less than 30 mL/hour (or 0.5 mL/kg/hour) of urine output over eight hours, the provider should be notified for prompt intervention. See Figure 15.7[14] for an illustration of an average adult’s daily water balance of 2,500 mL fluid intake balanced with 2,500 mL fluid output.
Fluid Imbalance
Two types of fluid imbalances are excessive fluid volume (also referred to as hypervolemia) and deficient fluid volume (also referred to as hypovolemia). These imbalances primarily refer to imbalances in the extracellular compartment, but can cause fluid movement in the intracellular compartments based on the sodium level of the blood.
Excessive Fluid Volume
Excessive fluid volume (also referred to as hypervolemia) occurs when there is increased fluid retained in the intravascular compartment. Patients at risk for developing excessive fluid volume are those with the following conditions:
- Heart Failure
- Kidney Failure
- Cirrhosis
- Pregnancy[15]
Symptoms of fluid overload include pitting edema, ascites, and dyspnea and crackles from fluid in the lungs. Edema is swelling in dependent tissues due to fluid accumulation in the interstitial spaces. Ascites is fluid retained in the abdomen.
Treatment depends on the cause of the fluid retention. Sodium and fluids are typically restricted and diuretics are often prescribed to eliminate the excess fluid. For more information about the nursing care of patients with excessive fluid volume, see the “Applying the Nursing Process” section.
Deficient Fluid Volume
Deficient fluid volume (also referred to as hypovolemia or dehydration) occurs when loss of fluid is greater than fluid input. Common causes of deficient fluid volume are diarrhea, vomiting, excessive sweating, fever, and poor oral fluid intake. Individuals who have a higher risk of dehydration include the following:
- Older adults
- Infants and children
- Patients with chronic diseases such as diabetes mellitus and kidney disease
- Patients taking diuretics and other medications that cause increased urine output
- Individuals who exercise or work outdoors in hot weather[16]
In adults, symptoms of dehydration are as follows:
- Feeling very thirsty
- Dry mouth
- Headache
- Dry skin
- Urinating and sweating less than usual
- Dark, concentrated urine
- Feeling tired
- Changes in mental status
- Dizziness due to decreased blood pressure
- Elevated heart rate[17]
In infants and young children, additional symptoms of dehydration include the following:
- Crying without tears
- No wet diapers for three hours or more
- Being unusually sleepy or drowsy
- Irritability
- Eyes that look sunken
- Sunken fontanel[18]
Dehydration can be mild and treated with increased oral intake such as water or sports drinks. Severe cases can be life-threatening and require the administration of intravenous fluids.
For more information about water balance and fluid movement, review the following video.
- “Cellular_Fluid_Content.jpg” by Welcome1To1The1Jungle is licensed under CC BY 3.0↵
- Fluid. (2012). In Britannica. https://www.britannica.com/science/fluid-biology↵
- This work is a derivative of StatPearls by Brinkman, Dorius, and Sharma and is licensed under CC BY 4.0↵
- This work is a derivative of StatPearls by Brinkman, Dorius, and Sharma and is licensed under CC BY 4.0↵
- "Hydrostatic Pressure" by Ann Lawrie is licensed under CC BY-NC 2.0↵
- “Capillary_microcirculation.jpg” by Kes47 is in the Public Domain↵
- BBC. (n.d.) Movement across cell membranes. https://www.bbc.co.uk/bitesize/guides/zc9tyrd/revision/5↵
- “0307_Osmosis.jpg” by OpenStax is licensed under CC BY 4.0. Access for free at https://openstax.org/books/anatomy-and-physiology/pages/3-1-the-cell-membrane↵
- “Simple_Diffusion.png” by Elizabeth2424 is licensed under CC BY-SA 3.0↵
- BBC. (n.d.) Movement across cell membranes. https://www.bbc.co.uk/bitesize/guides/zc9tyrd/revision/5↵
- “Sodium-potassium_pump_and_diffusion.png” by BruceBlaus.com staff is licensed under CC BY 3.0↵
- “2626_Renin_Aldosterone_Angiotensin.jpg” by OpenStax is licensed under CC BY 3.0. Access for free at https://openstax.org/books/anatomy-and-physiology/pages/25-4-microscopic-anatomy-of-the-kidney↵
- This work is a derivative of StatPearls by Fountain and Lappin and is licensed under CC BY 4.0↵
- "Water_balance.png" by David Walsh and Alan Sved is licensed under CC BY-SA 4.0↵
- Lewis, J. L., III. (June 2020). Volume overload. Merck Manual Professional Version. https://www.merckmanuals.com/professional/endocrine-and-metabolic-disorders/fluid-metabolism/volume-overload↵
- MedlinePlus [Internet]. Bethesda (MD): National Library of Medicine (US); Dehydration; [updated 2020, Oct 1; reviewed 2016, Apr 15; cited 2020, Aug 5]. https://medlineplus.gov/dehydration.html↵
- MedlinePlus [Internet]. Bethesda (MD): National Library of Medicine (US); Dehydration; [updated 2020, Oct 1; reviewed 2016, Apr 15; cited 2020, Aug 5]. https://medlineplus.gov/dehydration.html↵
- MedlinePlus [Internet]. Bethesda (MD): National Library of Medicine (US); Dehydration; [updated 2020, Oct 1; reviewed 2016, Apr 15; cited 2020, Aug 5]. https://medlineplus.gov/dehydration.html↵
- Forciea, B. (2017, April 21). Fluids and electrolytes: Water. [Video]. YouTube. All rights reserved. Video used with permission. https://youtu.be/VMxmDeduKR0↵


