12.6: Polymers
- Page ID
- 15310
- To draw structures for monomers that can undergo addition polymerization and for four-monomer-unit sections of an addition polymer.
The most important commercial reactions of alkenes are polymerizations, reactions in which small molecules, referred to in general as monomers (from the Greek monos, meaning “one,” and meros, meaning “parts”), are assembled into giant molecules referred to as polymers (from the Greek poly, meaning “many,” and meros, meaning “parts”). A polymer is as different from its monomer as a long strand of spaghetti is from a tiny speck of flour. For example, polyethylene, the familiar waxy material used to make plastic bags, is made from the monomer ethylene—a gas.
There are two general types of polymerization reactions: addition polymerization and condensation polymerization. In addition polymerization, the monomers add to one another in such a way that the polymer contains all the atoms of the starting monomers. Ethylene molecules are joined together in long chains. The polymerization can be represented by the reaction of a few monomer units:
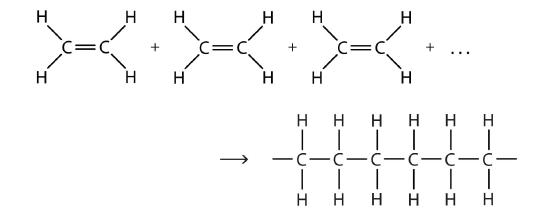
The bond lines extending at the ends in the formula of the product indicate that the structure extends for many units in each direction. Notice that all the atoms—two carbon atoms and four hydrogen atoms—of each monomer molecule are incorporated into the polymer structure. Because displays such as the one above are cumbersome, the polymerization is often abbreviated as follows:
nCH2=CH2 → [ CH2CH2 ] n
Many natural materials—such as proteins, cellulose and starch, and complex silicate minerals—are polymers. Artificial fibers, films, plastics, semisolid resins, and rubbers are also polymers. More than half the compounds produced by the chemical industry are synthetic polymers.
Some common addition polymers are listed in Table \(\PageIndex{1}\). Note that all the monomers have carbon-to-carbon double bonds. Many polymers are mundane (e.g., plastic bags, food wrap, toys, and tableware), but there are also polymers that conduct electricity, have amazing adhesive properties, or are stronger than steel but much lighter in weight.
| Monomer | Polymer | Polymer Name | Some Uses |
|---|---|---|---|
| CH2=CH2 | ~CH2CH2CH2CH2CH2CH2~ | polyethylene | plastic bags, bottles, toys, electrical insulation |
| CH2=CHCH3 | 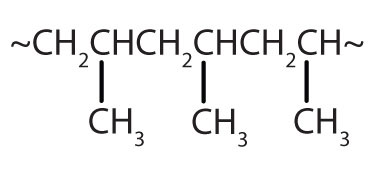 |
polypropylene | carpeting, bottles, luggage, exercise clothing |
| CH2=CHCl | 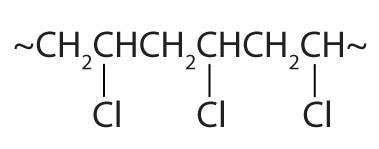 |
polyvinyl chloride | bags for intravenous solutions, pipes, tubing, floor coverings |
| CF2=CF2 | ~CF2CF2CF2CF2CF2CF2~ | polytetrafluoroethylene | nonstick coatings, electrical insulation |
An interesting use of polymers is the replacement of diseased, worn out, or missing parts in the body. For example, about a 250,000 hip joints and 500,000 knees are replaced in US hospitals each year. The artificial ball-and-socket hip joints are made of a special steel (the ball) and plastic (the socket). People crippled by arthritis or injuries gain freedom of movement and relief from pain. Patients with heart and circulatory problems can be helped by replacing worn out heart valves with parts based on synthetic polymers. These are only a few of the many biomedical uses of polymers.
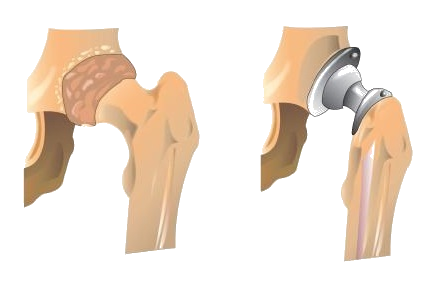 Figure \(\PageIndex{1}\): Hip Joint Replacement. Synthetic polymers are an important part of a hip joint replacement. The hip is much like a ball-and-socket joint, and total hip replacements mimic this with a metal ball that fits in a plastic cup.
Figure \(\PageIndex{1}\): Hip Joint Replacement. Synthetic polymers are an important part of a hip joint replacement. The hip is much like a ball-and-socket joint, and total hip replacements mimic this with a metal ball that fits in a plastic cup.
Key Takeaway
- Molecules having carbon-to-carbon double bonds can undergo addition polymerization.

