15.6: Properties of Monosaccharides
- Page ID
- 15356
- To identify the physical and chemical properties of monosaccharides.
Monosaccharides such as glucose and fructose are crystalline solids at room temperature, but they are quite soluble in water, each molecule having several OH groups that readily engage in hydrogen bonding. The chemical behavior of these monosaccharides is likewise determined by their functional groups.
An important reaction of monosaccharides is the oxidation of the aldehyde group, one of the most easily oxidized organic functional groups. Aldehyde oxidation can be accomplished with any mild oxidizing agent, such as Tollens’ reagent or Benedict’s reagent. With the latter, complexed copper(II) ions are reduced to copper(I) ions that form a brick-red precipitate [copper(I) oxide; Figure \(\PageIndex{1}\)].
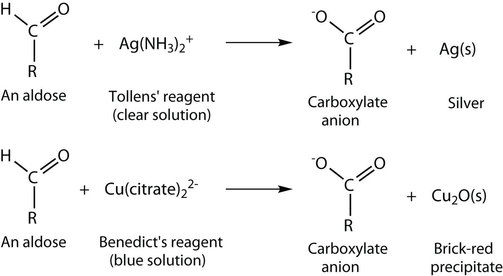
Any carbohydrate capable of reducing either Tollens’ or Benedict’s reagents without first undergoing hydrolysis is said to be a reducing sugar. Because both the Tollens’ and Benedict’s reagents are basic solutions, ketoses (such as fructose) also give positive tests due to an equilibrium that exists between ketoses and aldoses in a reaction known as tautomerism.
 Figure \(\PageIndex{1}\): Benedict’s Test. Benedict’s test was performed on three carbohydrates, depicted from left to right: fructose, glucose, and sucrose. The solution containing sucrose remains blue because sucrose is a nonreducing sugar.
Figure \(\PageIndex{1}\): Benedict’s Test. Benedict’s test was performed on three carbohydrates, depicted from left to right: fructose, glucose, and sucrose. The solution containing sucrose remains blue because sucrose is a nonreducing sugar.
These reactions have been used as simple and rapid diagnostic tests for the presence of glucose in blood or urine. For example, Clinitest tablets, which are used to test for sugar in the urine, contain copper(II) ions and are based on Benedict’s test. A green color indicates very little sugar, whereas a brick-red color indicates sugar in excess of 2 g/100 mL of urine.
Summary
Monosaccharides are crystalline solids at room temperature and quite soluble in water. Monosaccharides are reducing sugars; they reduce mild oxidizing agents, such as Tollens’ or Benedict’s reagents.

