7.3A: Vitamin B₁ (Thiamine)
- Page ID
- 1258
Thiamine, also known as thiamin or vitamin B1, is a vitamin found in food and used as a dietary supplement. As a supplement it is used to treat and prevent thiamine deficiency and disorders that result from it, including beriberi, Korsakoff's syndrome, and Korsakoff's psychosis. Other uses include maple syrup urine disease and Leigh's disease. It is taken by mouth or by injection. Side effects are generally few. Allergic reactions including anaphylaxis may occur. Thiamine is in the B complex family. It is needed for the metabolism of carbohydrates.[1] As people are unable to make it, thiamine is an essential nutrient. Food sources include whole grains, meat, and fish.
Thiamine was discovered in 1897 and is on the World Health Organization's List of Essential Medicines, the most effective and safe medicines needed in a health system. Thiamine is available as a generic medication and over the counter. The wholesale cost in the developing world is about 2.17 USD per one gm vial. In the United States a month of replacement is less than 25 USD. Some countries require its addition to certain foods such as grains. Thiamine is used to treat thiamine deficiency which can prove fatal. In less-severe cases, nonspecific signs include malaise, weight loss, irritability and confusion.
Thiamine is a colorless organosulfur compound with a chemical formula \(\ce{C12H17N4OS}\) (Figure \(\PageIndex{1}\)). Its structure consists of an aminopyrimidine and a thiazole ring linked by a methylene bridge. The thiazole is substituted with methyl and hydroxyethyl side chains. Thiamine is soluble in water, methanol, and glycerol and practically insoluble in less polar organic solvents. It is stable at acidic pH, but is unstable in alkaline solutions. Thiamine, which is a N-heterocyclic carbene, can be used in place of cyanide as a catalyst for benzoin condensation. Thiamine is unstable to heat, but stable during frozen storage.[citation needed] It is unstable when exposed to ultraviolet light[9] and gamma irradiation.
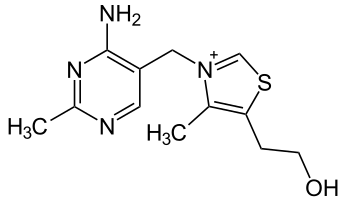
Figure \(\PageIndex{1}\): Structure of thiamine
Thiamin is found in a wide variety of processed and whole foods, with edible seeds, legumes, rice and processed foods, such as breakfast cereals, having among the highest contents. Some other foods naturally rich in thiamin are corn flour, pork, pecans and spinach.
Dietary Reference Intakes
The Food and Nutrition Board of the U.S. Institute of Medicine updated Estimated Average Requirements (EARs) and Recommended Dietary Allowances (RDAs) for thiamine in 1998. The current EARs for thiamine for women and men ages 14 and up are 0.9 mg/day and 1.0 mg/day, respectively; the RDAs are 1.1 and 1.2 mg/day. RDAs are higher than EARs so as to identify amounts that will cover people with higher than average requirements. RDA for pregnancy equals 1.4 mg/day. RDA for lactation equals 1.4 mg/day. For infants up to 12 months the Adequate Intake (AI) is 0.2-0.3 mg/day. and for children ages 1–13 years the RDA increases with age from 0.5 to 0.9 mg/day.
As for safety, the Food and Nutrition Board of the U.S. Institute of Medicine sets Tolerable Upper Intake Levels (known as ULs) for vitamins and minerals when evidence is sufficient. In the case of thiamine there is no UL, as there is no human data for adverse effects from high doses. The European Food Safety Authority reviewed the same safety question and also reached the conclusion that there was not sufficient evidence to set a UL for thiamine. Collectively the EARs, RDAs and ULs are referred to as Dietary Reference Intakes.
For U.S. food and dietary supplement labeling purposes the amount in a serving is expressed as a percent of Daily Value (%DV). For thiamine labeling purposes 100% of the Daily Value was 1.5 mg, but as of May 2016 it has been revised to 1.2 mg. A table of the pre-change adult Daily Values is provided at Reference Daily Intake. Food and supplement companies have until July 28, 2018 to comply with the change.
Antagonists
Thiamine in foods can be degraded in a variety of ways. Sulfites, which are added to foods usually as a preservative, will attack thiamine at the methylene bridge in the structure, cleaving the pyrimidine ring from the thiazole ring. The rate of this reaction is increased under acidic conditions. Thiamine is degraded by thermolabile thiaminases (present in raw fish and shellfish). Some thiaminases are produced by bacteria. Bacterial thiaminases are cell surface enzymes that must dissociate from the membrane before being activated; the dissociation can occur in ruminants under acidotic conditions. Rumen bacteria also reduce sulfate to sulfite, therefore high dietary intakes of sulfate can have thiamine-antagonistic activities.
Plant thiamine antagonists are heat-stable and occur as both the ortho- and para-hydroxyphenols. Some examples of these antagonists are caffeic acid, chlorogenic acid, and tannic acid. These compounds interact with the thiamine to oxidize the thiazole ring, thus rendering it unable to be absorbed. Two flavonoids, quercetin and rutin, have also been implicated as thiamine antagonists.
Absorption
Thiamine is released by the action of phosphatase and pyrophosphatase in the upper small intestine. At low concentrations, the process is carrier-mediated, and, at higher concentrations, absorption occurs via passive diffusion. Active transport is greatest in the jejunum and ileum; but, active transport can be inhibited by alcohol consumption and by folic deficiency. Decline in thiamine absorption occurs at intakes above 5 mg/day. The cells of the intestinal mucosa have thiamine pyrophosphokinase activity, but it is unclear as to whether the enzyme is linked to active absorption. The majority of thiamine present in the intestine is in the pyrophosphorylated form ThDP, but when thiamine arrives on the serosal side of the intestine it is often in the free form. The uptake of thiamine by the mucosal cell is likely coupled in some way to its phosphorylation/dephosphorylation. On the serosal side of the intestine, evidence has shown that discharge of the vitamin by those cells is dependent on Na+-dependent ATPase.
Uptake of thiamine by cells of the blood and other tissues occurs via active transport and passive diffusion. The brain requires much more thiamine than other tissues of the body. Much of ingested thiamine never reaches the brain because of passive diffusion and the blood–brain barrier. About 80% of intracellular thiamine is phosphorylated and most is bound to proteins. In some tissues, thiamine uptake and secretion appears to be mediated by a soluble thiamine transporter that is dependent on Na+ and a transcellular proton gradient.
Human storage of thiamine is about 25 to 30 mg, with the greatest concentrations in skeletal muscle, heart, brain, liver, and kidneys. ThMP and free (unphosphorylated) thiamine is present in plasma, milk, cerebrospinal fluid, and, it is presumed, all extracellular fluids. Unlike the highly phosphorylated forms of thiamine, ThMP and free thiamine are capable of crossing cell membranes. Thiamine contents in human tissues are less than those of other species.
Contributors and Attributions
- Wikipedia. Content is copyrighted under a CC-BY-SA 4.0 license.

