11.1C: Spinal Cord Grey Matter and Spinal Roots
- Page ID
- 7613
The grey matter of the spinal cord contains neuronal cell bodies, dendrites, axons, and nerve synapses.
- Describe the grey matter and spinal roots of the spinal cord
Key Points
- Each segment of the spinal cord is associated with a pair of ganglia called dorsal root ganglia, situated just outside of the spinal cord.
- The dorsal root ganglia contain the cell bodies of sensory neurons. Axons of these sensory neurons travel into the spinal cord via the dorsal roots.
- The grey matter in the center of the cord contains interneurons and the cell bodies of motor neurons, axons, and dendrites.
- Projections of the grey matter (the “wings”) are called horns. Together, the grey horns and the grey commissure form the H-shaped grey matter.
- The dorsal root ganglia develops in the embryo from neural crest cells. The spinal ganglia can thus be regarded as grey matter of the spinal cord that was translocated to the periphery.
Key Terms
- neural crest: A strip of ectodermal material in the early vertebrate embryo inserted between the prospective neural plate and the epidermis.
- grey matter: A major component of the central nervous system consisting of neuronal cell bodies, neuropil (dendrites and unmyelinated axons), glial cells (astroglia and oligodendrocytes), and capillaries.
- neural tube: The embryonic precursor to the central nervous system (CNS).
EXAMPLES
- The spine acts as the conduit to relay information to and from the brain from the rest of the body.
- Damage to the grey matter (eg, the ventral gray horn) may lead to tingling and muscle weakness.
The spinal cord is the main pathway for information connecting the brain and peripheral nervous system. The spinal cord is much shorter in length than the bony spinal column. The human spinal cord extends from the foramen magnum of the occipital bone of the skull and continues to the conus medullaris near the second lumbar vertebra, terminating in a fibrous extension known as the filum terminale.
Spinal Cord Topography and Roots
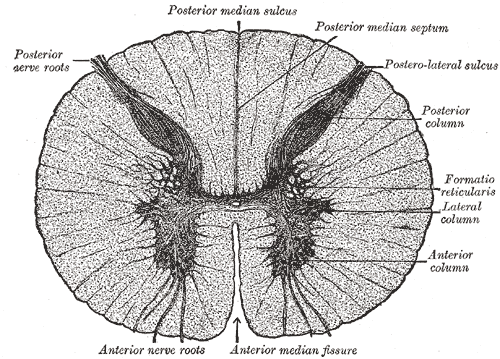
Cross-Section of Spinal Cord, Mid-Thoracic Level: Note the commissure, rendered in grey.
The spinal cord is compressed dorsoventrally, giving it an elliptical shape. The cord has grooves in the dorsal and ventral sides. The posterior median sulcus is the groove in the dorsal side, and the anterior median fissure is the groove in the ventral side.
Each segment of the spinal cord is associated with a pair of ganglia called dorsal root ganglia, situated just outside of the spinal cord. These ganglia contain cell bodies of sensory neurons. Axons of these sensory neurons travel into the spinal cord via the dorsal roots.
The grey matter, in the center of the cord, is shaped like a butterfly and consists of cell bodies of interneurons and motor neurons, as well as neuroglia cells and unmyelinated axons. Projections of the grey matter (the “wings”) are called horns. Together, the grey horns and the grey commissure form the H-shaped grey matter.
Dorsal and Ventral Roots
The dorsal root ganglia lie along the vertebral column by the spine. The dorsal root ganglia develops in the embryo from neural crest cells, not the neural tube. Hence, the spinal ganglia can be regarded as grey matter of the spinal cord that became translocated to the periphery.
The axons of dorsal root ganglion neurons are known as afferents. In the peripheral nervous system, afferents refer to the axons that relay sensory information into the central nervous system. These neurons are of the pseudo-unipolar type, meaning that they have an axon with two branches that act as a single axon, often referred to as distal and proximal processes. Ventral roots consist of axons from motor neurons, which bring information to the periphery from cell bodies within the CNS. Dorsal roots and ventral roots come together and exit the intervertebral foramina as they become spinal nerves.
The nerve endings of dorsal root ganglion neurons have a variety of sensory receptors that are activated by mechanical, thermal, chemical, and noxious stimuli. In these sensory neurons, a group of ion channels thought to be responsible for somatosensory transduction has been identified. Compression of the dorsal root ganglion by a mechanical stimulus lowers the voltage threshold needed to evoke a response and causes action potentials to be fired. This firing may even persist after the removal of the stimulus.
Impulse Transmission
The dendrite receives information from another neuron’s axon at the synapse, and the axon sends information to the next neuron’s dendrites. Unlike the majority of neurons found in the CNS, an action potential in a dorsal root ganglion neuron may initiate in the distal process in the periphery, bypass the cell body, and continue to propagate along the proximal process until reaching the synaptic terminal in the dorsal horn of the spinal cord.
The distal section of the axon may either be a bare nerve ending or encapsulated by a structure that helps relay specific information to nerve. For example, a Meissner’s corpuscle or a Pacinian corpuscle may encapsulate the nerve ending, rendering the distal process sensitive to mechanical stimulation, such as stroking or vibration.
Ion Channels
Two distinct types of mechanosensitive ion channels have been found in the dorsal root ganglia, broadly classified as either high-threshold (HT) or low-threshold (LT). As their names suggest, they have different thresholds as well as different sensitivities to pressure. These are cationic channels whose activity appears to be regulated by the proper functioning of the cytoskeleton and cytoskeleton-associated proteins. The presence of these channels in the dorsal root ganglion gives reason to believe that other sensory neurons may contain them as well.
High-threshold channels have a possible role in nociception. These channels are found predominantly in smaller sensory neurons in the dorsal root ganglion cells and are activated by higher pressures, two attributes that are characteristic of nociceptors. Also, the threshold of HT channels was lowered in the presence of PGE2 (a compound that sensitizes neurons to mechanical stimuli and mechanical hyperalgesia), which further supports a role for HT channels in the transduction of mechanical stimuli into nociceptive neuronal signals.

