3.11: Supraglottoplasty for Laryngomalacia
- Page ID
- 18272
OPEN ACCESS ATLAS OF OTOLARYNGOLOGY, HEAD & NECK OPERATIVE SURGERY
LARYNGOMALACIA
Nico Jonas, Graeme van der Meer
Laryngomalacia is characterized by collapse of the supraglottic tissues on inspiration and is the most common cause of stridor in infancy. It generally becomes symptomatic 2-3 weeks after birth, and presents as characteristic high-pitched inspiratory, squeaking stridor. Classically symptoms are worse in the supine position, when the child cries, or is agitated. Males are affected twice as often as females. It usually resolves by the age of 2 years. Laryngomalacia may remain symptomatic beyond 2 years in patients with neuromascular disorders and should be considered in the differential diagnosis of inspiratory stridor even in older children. The etiology is not fully understood. There does appear to be a familial predisposition. Anatomically, redundant supraglottic soft tissues collapse on inspiration and cause supraglottic airway obstruction. Another theory is that of immature neuromuscular development causing supraglottic compromise. A common coexisting condition is that of acid reflux which may exacerbate laryngomalacia by causing posterior supraglottic edema.
Diagnosis
History
Diagnosis is usually suspected based on the history. The classic presentation is a neonate with inspiratory stridor starting 2-3 weeks after birth. Stridor is worse in the supine position and during feeding or with crying. The clinician should enquire about weight gain, cyanotic episodes, poor or prolonged feeding, aspiration and sleep disordered breathing. The voice should be unaffected, and the neonate should have a normal cry.
Clinical findings

Figure 1: Flexible laryngoscope with suction port
The diagnosis is confirmed by flexible fibreoptic laryngoscopy. This can be done in the awake, upright patient, using a flexible fibreoptic laryngoscope, ideally with a suction port (Figure 1).
Some clinicians prefer not to prepare the nose with local anesthetic as this permits feeding to comfort the neonate immediately following the procedure. The endoscope is passed via the nose or mouth. Limitations are that the subglottis and distal airway cannot be inspected.
Classic laryngoscopy findings
- Short aryepiglottic folds narrowing the supraglottis
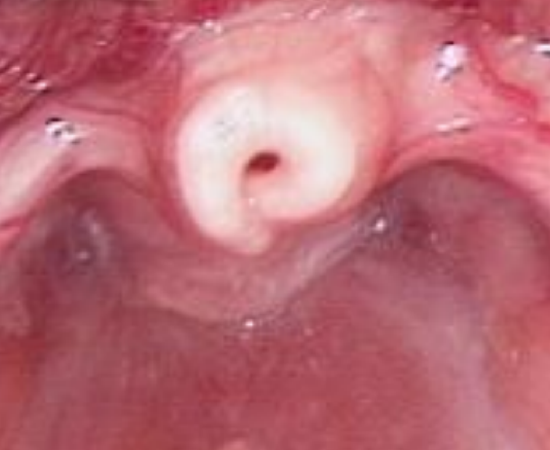
Figure 2: Curled-up, omega-shaped epiglottis
- Omega or tubular shaped epiglottis (Figure 2)
- Anterior prolapse of the corniculate and cuneiform cartilages

Figure 3: Omega-shaped epiglottis, short aryepiglottic folds (blue brackets) and prolapsing arytenoid mucosa (a: open during expiration, b: closed during inspiration)

Figure 4: Laryngomalacia secondary to prolapsing arytenoids: (a: open during expiration, b: closed during inspiration)
- There may be redundant soft tissue around the arytenoids. (Figures 3, 4)
- Intact vocal cord movement
Radiology is unnecessary unless concomitant pathology is suspected. Sleep studies or overnight saturation studies should be performed in patients when sleep disordered breathing and desaturations are suspected.
Direct laryngoscopy is occasionally required and indications include:
- Atypical history
- Suspicion of distal airway pathology
- Unable to make a diagnosis after flexible laryngoscopy
Classification
A variety of classification systems are used for laryngomalacia. The following system is pragmatic with specific treatments for each type:
Type 1: Foreshortened or taut aryepiglottic folds
Type 2: Presence of redundant supraglottic soft tissue
Type 3: Other underlying etiologies, such as neuromuscular disorders; this rarer type may present with the epiglottis prolapsing onto the vocal cords with inspiration
While these 3 types are not mutually exclusive, each should be considered as a separate pathological entity with a final common clinical presentation. Each type requires a specific surgical approach.
Non-surgical treatment
- Laryngomalacia usually resolves by the age of 2 years; no treatment other than parental reassurance is required in 99% of cases. Once the diagnosis is made the condition is explained to the parents. It is important to emphasize to parents that it is a self-limiting condition that will resolve in the majority of cases
- Patients are weighed every 2 weeks and the weight charted on a centile chart. They should be reviewed after 6 weeks to ensure adequate weight gain
- Patients are started on anti-reflux treatment (proton pump inhibitors or Histamine H2 receptor blockers); it is important that the dose of anti-reflux medication is adjusted whenever the body weight increases by 10% to ensure adequate dosage
Surgical treatment
Criteria that alert the clinician that surgical intervention may be required include:
- Dyspnea at rest
- Difficult and prolonged feeding (must stop feeding to catch his/her breath)
- Failure to thrive characterized by crossing two or more centiles on the growth chart
- Sleep disordered breathing/obstructive sleep apnea syndrome
Preoperative assessment
Once the decision has been made that surgery is required, a swallow assessment is done to exclude aspiration. Patients are also assessed to exclude underlying neurological conditions.
Anesthesia
- General anesthesia, preferably with spontaneous ventilation
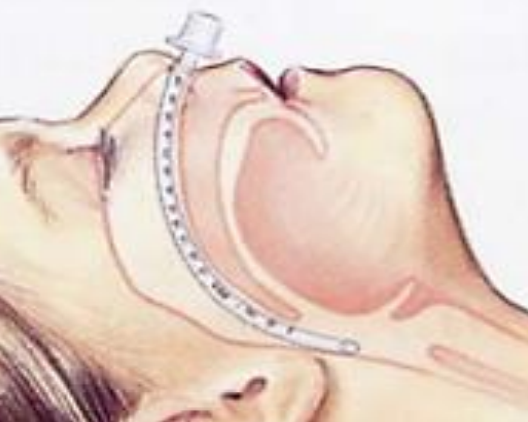
Figure 5: Endotracheal tube used as nasopharyngeal airway
- Anesthetic can be delivered via an endotracheal tube, used as a nasopharyngeal airway (Figure 5)
- Endotracheal intubation or laryngeal mask ventilation is occasionally required prior to diagnostic endoscopy and the surgical procedure
- Total intravenous anesthesia is preferred to avoid exposing operating staff to inhalant anesthetic gases
- Once the appropriate depth of anesthesia has been reached, the anesthetist removes the tube or mask and the surgeon inserts a laryngeal suspension laryngoscope, taking care not to injure the patient’s gums or teeth or to overextend the neck
- The patient may be allowed to breathe spontaneously with oxygen
- Anesthetic gases are delivered via a nasopharyngeal airway
- Dexamethasone (0.25 mg/kg) preoperatively minimizes postoperative inflammation and edema and may make the procedure more comfortable to the patient
Surgery: Diagnostic steps
- Diagnostic laryngoscopy, tracheoscopy and bronchoscopy are done to
- Confirm the diagnosis
- Exclude concomitant airway pathology
- Confirm normal vocal cord movement
- Position the patient in the “sniffing position” to allow the larynx and trachea to be visualized; a headrest and cushion behind the shoulders or a shoulder roll facilitate the procedure
- Ensure that the shared airway is not compromised by either the surgeon or the anesthetist. Good communication between the anesthetist and surgeon is essential

Figure 6: Pediatric laryngoscope
- Select the appropriate laryngoscope (different sizes according to age) that allows ventilation via a side port (Figure 6)
- Once the larynx is visualized, anesthetize it with lignocaine sprayed onto the larynx (max dose: 4 mg/kg)
- With the laryngoscope correctly in place, thoroughly inspect the larynx, trachea and bronchi with a 4 mm 0° Hopkin’s rod looking specifically for concomitant pathology such as a laryngeal cleft, subglottic/tracheal stenosis or tracheal defects (present in up to 15% of patients)
- Then conduct a thorough inspection of larynx to confirm the diagnosis of laryngomalacia
- It is essential to assess vocal cord movement as well as cricoarytenoid mobility
Supraglottoplasty
Tracheostomy is reserved only for exceptional cases of severe laryngomalacia causing airway obstruction when supraglottoplasty is contraindicated.
The aim of surgery is to correct the anatomical cause with minimal tissue damage.
Cold steel technique is recommended. (Other instruments have been used including laser and microdebriders). An advantage of a cold steel technique is that it can be performed in the presence of an endotracheal tube which secures the airway and makes the anesthetic more manageable especially in the presence of severe gastroesophageal reflux or lung pathology. Furthermore, the required equipment is readily available in ENT units and there are no expensive consumables. The operating microscope allows the surgeon to use both hands when performing the supraglottic surgery.
After confirming the diagnosis, the following approach to treatment is implemented:
Laryngomalacia Type I (shortened aryepiglottic folds)
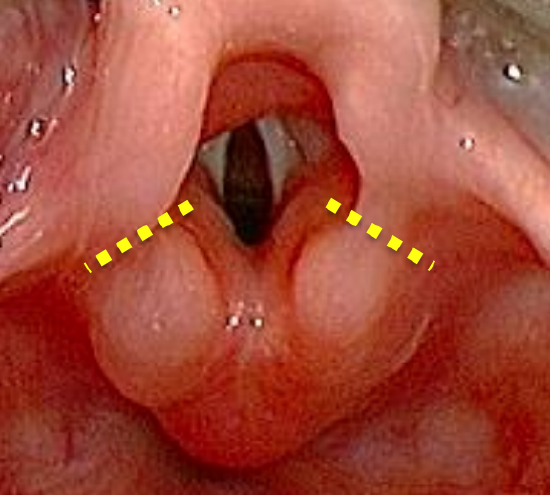
Figure 7: Short aryepiglottic folds; Yellow lines indicate where aryepiglottic cuts should be made

Figure 8: Endoscopic view illustrating improved supraglottic airway following cutting of shortened aryepiglottic folds
- Simple incision of the shortened aryepiglottic folds is the mainstay of treatment and has a success rate of 90% (Figures 7, 8)
- Apply adrenaline-soaked pledgets to the area to be incised with grasping forceps to provide tissue compression and diminish bleeding
- Tense the shortened aryepiglottic fold by grasping the mucosa at the back of the arytenoid and pulling it posteriorly
- Divide the shortened aryepiglottic fold with laryngeal microscissors down to the level of the false cord
- Control bleeding with adrenaline pledgets
- Repeat the procedure on the opposite side
- Some authors advocate excising a wedge, citing a slightly improved success rate
Laryngomalacia Type II (redundant supraglottic mucosa)
- Apply adrenaline-soaked pledgets to the area to be excised with grasping forceps to diminish bleeding
- Excise redundant mucosa using larynxgeal graspers and scissors
- Take care not to excise too much tissue
- It is very important to avoid traumatizing the mucosa on the medial surface of the arytenoid in order to prevent interarytenoid scarring and airway stenosis
Most cases are successfully treated by the above mentioned two procedures. Occasionally an epiglottopexy is required; this can be done using CO2 laser to burn the base of the lingual surface of the epiglottis or by placing a suture between the lingual surface of the epiglottis and the tongue base.
Postoperative care
- Extubate and monitor overnight in a pediatric ICU or high care unit
- Adequate analgesia
- Humidified air
- As patients not uncommonly aspirate following supraglottoplasty, prescribe antireflux medication
- Dexamathasone is occasionally given to reduce postoperative swelling and edema
Contraindications to supraglottoplasty
- Underlying neurological condition
- Severe aspiration
- Abnormal vocal cord movement with aspiration
Complications
- Aspiration can be a major problem especially with neurological variants of laryngomalacia
- Persistent dyspnea, sleep apnea, and/ or failure to thrive
- Granulomas
- Edema
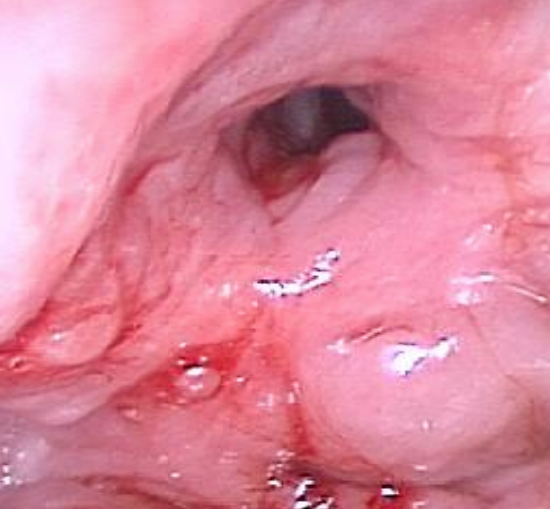
Figure 9: Severe supraglottic stenosis secondary to excessive lasering with supraglottic surgery
- Supraglottic scarring and fibrosis due to excessive removal of supraglottic tissues (Figure 9)
Complications are more frequent in children with associated congenital abnormalities; some surgeons recommend unilateral surgery in high risk cases to limit side effects. Most patients treated unilaterally have good outcomes although some patients do have to subsequently have a contralateral procedure done.
Important Tips
- Meticulous preparation and planning prior to surgery facilitates the procedure and minimizes complications
- Ensure correct patient positioning to achieve good surgical exposure
- Avoid excessive resection of supraglottic tissue
- Acid reflux disease control is important both pre- and postoperatively
Author and Pediatric Section Editor
Nico Jonas MBChB, FCORL, MMed
Pediatric Otolaryngologist
Addenbrooke’s Hospital
Cambridge, United Kingdom
nico.jonas@gmail.com
Author
Graeme van der Meer MBChB MMed
Otolaryngologist
Netcare Settlers Hospital
Grahamstown, South Africa
graemevdm@gmail.com
Editor
Johan Fagan MBChB, FCS(ORL), MMed
Professor and Chairman
Division of Otolaryngology
University of Cape Town
Cape Town, South Africa
johannes.fagan@uct.ac.za


