5.5: More complex designs
- Page ID
- 13162
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)5.1 Two groups of unequal size
Sections 3 and 4 considered the simplest situation where the two groups to be com- pared are of equal size. Sometimes, there may be reasons for wishing to allocate more individuals to one group than to the other. For example, if an experimental drug is very expensive, it may be desired to minimize the number of patients allocated to the
drug, and so the trial may be arranged so that there are two or three patients given the old drug for every patient given the new drug. In order to maintain the same power as in the equal allocation scheme, a larger total trial size will be needed, but the number given the new drug will be smaller. Conversely, in a trial of a new vaccine, it may be decided to allocate twice as many participants to the vaccinated group as are included in the placebo group, in order to increase the size of the safety database for the new vac- cine, before it goes into public health programmes.
Let the size of the smaller of the two groups be n1, and suppose the ratio of the two sample sizes to be k, so that there will be kn1 individuals in the other group (k >1).Then, to achieve approximately the same power and precision as in a trial with an equal number n in each group, n1 should be chosen as:
\[
n_{1}=n(k+1) /(2 k)
\]
Table 5.5 Trial size necessary to achieve approximately the same power in a trial with two groups, one of which contains k times as many individuals as the other
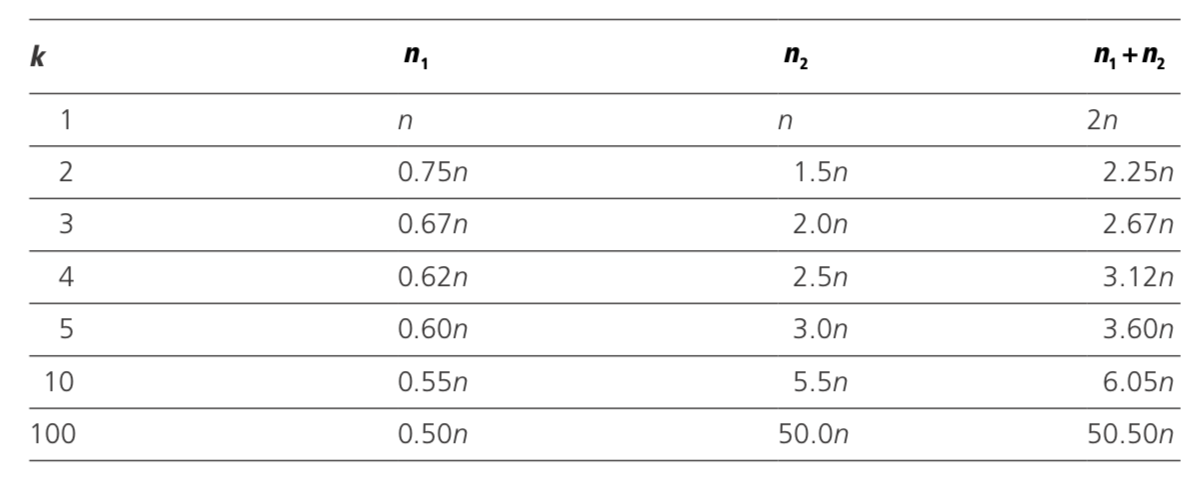
Examples are shown in Table 5.5 for various values of k. Notice that the number al- located to the smaller group can never be reduced below half the number required with equal groups. Little is gained by increasing k beyond 3 or 4, since, beyond this point, even a substantial increase in n2 achieves only a small reduction in n1.
5.2 Comparison of more than two groups
Field trials comparing two groups (for example, intervention and control, or treatment A and treatment B) are by far the commonest. However, in some trials, three or more groups may be compared. For example, in a trial of a new vaccine, there may be four trial groups receiving different doses of the vaccine. It is unusual for field trials to have more than four groups, because of logistical constraints or trial size limitations.
It is suggested that, in designing a trial with three or more groups, the investigator should decide which pair-wise comparisons between groups are of central interest. The methods of Sections 3 and 4 can then be used to decide on the trial size required in each group. Where there is one control group for comparison with several intervention groups, it is likely that the main pair-wise comparisons will be between each interven- tion group and the control group. Note, however, that direct comparisons between the intervention groups may then be inadequately powered, since, if each of the interven- tions has some effect, differences between the intervention groups may be smaller than when each is compared with the control group.
5.3 Factorial designs
As discussed in Chapter 4, Section 3.2, some trials are designed to look simultaneously at the effects of two interventions, using a factorial design. In a \(2×2\) factorial trial of two interventions A and B, for example, participants are randomly allocated between four trial groups receiving A only, B only, both A and B, or a control group receiving neither intervention. If the effects of A and B can be assumed independent, so that the effect of A is the same in the presence or absence of B and vice versa, then this trial design allows us to measure the effects of the two interventions for roughly the price of a single two-group trial measuring the effect of one intervention.
Under these conditions of independence, the main change to the calculation of sample size for a \(2×2\) factorial trial is that the expected outcome in the intervention and control groups for intervention A has to be adjusted for the expected effect of intervention B. This is explained with an example.
For example, suppose we are interested in the effects of iron supplements (intervention A) and anti-malarial prophylaxis (intervention B) on anaemia during pregnancy. Suppose that the prevalence of anaemia in the control group that receives neither A nor B is expected to be 30%, that each intervention is expected to reduce the prevalence proportionally by 20%, and that these effects are independent. Then the expected prevalences in the four arms of the trial will be: control—30%; A only—24%; B only—24%; \(A+B\)—19.2%. In this factorial trial, the effect of intervention A will be estimated by comparing the prevalence between groups \(A+B\) and B only, and between group A only and the control group. The overall prevalence in the two groups given intervention A will be \(21.6%[=(24+19.2)/2]\) and in the two groups not given A \(27%[=(30+24)/2]\) Since the difference in prevalences is slightly smaller than in a simple two-group trial, the total sample size will be somewhat larger for the factorial design.
In some factorial trials, we may wish to look explicitly at whether the effects of the two interventions are independent. This requires a test for interaction or effect modification, since we are interested in whether the effect of A, for example, differs according to the presence or absence of B. Testing for interaction generally requires a much larger sample size than a simple comparison of two groups. As a rough guideline, the total sample size for a \(2×2\) factorial trial would need to be multiplied by at least four to detect a substantial interaction (of similar size to the main effects of the interventions) between the effects of two interventions.
5.4 Equivalence and non-inferiority trials
In most field trials, the objective is to determine whether a new intervention is superior to a control intervention, for example, an existing intervention. In some cases, however, we may wish to demonstrate that a new intervention is equivalent, or at least not inferior, to an existing intervention. For example, suppose the current treatment for some condition is known to be highly effective, but it is also expensive and has some unpleasant side effects. Now suppose that a new treatment has been developed which is less costly and has fewer side effects. This would probably be considered for implementation, as long as it is as effective as the old treatment. In this case, we may decide to conduct an equivalence trial aimed at determining whether the two treatments have similar efficacy.
For a full discussion of such trials, the reader is referred to Blackwelder (1982) or Wang and Bakhai (2006). However, a simple example is given to illustrate the required sample size calculations.
Example: Suppose that the current treatment for TB has a cure rate of around 90% but requires a prolonged course of treatment. A new shorter-course regimen has been developed which would have advantages, in terms of cost, convenience, and adherence. We wish to carry out a trial to determine whether the cure rate for the short-course regimen is equivalent to that of the current regimen. We would usually do this by defining a lower limit for the cure rate, below which we would no longer consider the treatments to be ‘equivalent’. If we set this at 85%, the trial would need to be powered to demonstrate that the difference in cure rates is no more than 5%. The null hypothesis is now that the new treatment is inferior to the old treatment, and we power the trial to reject this null hypothesis and declare equivalence of the two treatments if the new treatment has a cure rate that is not inferior to the standard treatment by more than the specified 5%.
Modifying the first equation in Section 4.1 appropriately, we need n patients in each group, where:
\[\boldsymbol { n } = \left[ \left( z _ { 1 } + z _ { 2 } \right) ^ { 2 } 2 p ( 1 - p ) \right] / D ^ { 2 }\]
In this equation, p is the expected cure rate of 90% in both groups, assuming equivalence, and D is the acceptable margin of inferiority, which is 5% in this example. Thus, for 90% power and a two-sided significance test withp=0.05,p=0.05,we have:
\[n = \left[ ( 1.96 + 1.28 ) ^ { 2 } \times 2 \times 0.90 \times 0.10 \right] / 0.05 ^ { 2 } = 756\]
In general, large sample sizes are needed to test equivalence.

