5.8: Detailed Mechanism of Cross-bridge Formation
- Page ID
- 131897
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)A neuromuscular junction exists between the axon terminal and the motor end plate of a muscle fiber where neurotransmitters are released.
- Outline the steps of the cross-bridge cycle.
- Identify the roles of calcium ions and ATP in regulating cross-bridge cycling and muscle fiber shortening.
- Explain the anatomical limit of shortening.
Cross-Bridge Formation is a key event in the molecular mechanism of the sliding filament model of muscle contraction. It occurs when the head of a myosin molecule temporarily attaches to a binding site on an actin filament within a sarcomere. This attachment forms a “cross-bridge” between the thick (myosin) and thin (actin) filaments, creating a physical link that allows the muscle to contract.
Cross-Bridge Cycle: The Molecular Mechanism of Sarcomere Shortening
Cross-bridge formation is essential, but it does not occur automatically. The binding sites on actin must first be “unlocked.” This regulation is controlled by two proteins:
-
Tropomyosin: a long protein that wraps around actin filaments, covering the myosin-binding sites.
-
Troponin: a protein attached to tropomyosin that acts as a calcium sensor.
Together, they form the troponin–tropomyosin complex, which prevents myosin from binding to actin when the muscle is relaxed.

Step 1: Calcium Unlocks the Binding Sites
When a motor neuron stimulates a muscle fiber, calcium ions (Ca²⁺) flood into the sarcoplasm. Then:
-
Ca²⁺ binds to troponin.
-
Troponin changes shape and shifts tropomyosin away from the binding sites on actin.
-
Once the binding site is exposed, myosin heads can attach to actin, forming cross-bridges.
Step 2: The Cross-Bridge Cycle (The Pulling Action)
Myosin heads then go through a repeated cycle of binding, pulling, releasing, and resetting — like tiny oars rowing a boat.
Each cycle involves four main steps:
-
Cross-bridge formation: A myosin head binds to an exposed binding site on actin (only possible when Ca²⁺ is present).
-
Power stroke: The myosin head pivots, pulling the actin filament toward the M-line (the center of the sarcomere).
-
Cross-bridge detachment: ATP binds to the myosin head, causing it to release from actin.
-
Reactivation: ATP is hydrolyzed into ADP and Pi, releasing energy that re-cocks the myosin head into its high-energy position, ready for another cycle.

Because each pull is brief, the cycle must repeat many times in rapid succession. The repeated attachment, pivoting, and release of myosin heads slide the thin (actin) filaments past the thick (myosin) filaments, shortening the sarcomere. The force produced by one sarcomere is small, but when multiplied across all sarcomeres within a myofibril, all myofibrils within a muscle fiber, and all fibers within a muscle, the total force and movement generated become significant. The coordinated action of millions of myosin heads working together generates a strong, steady contraction of the entire muscle.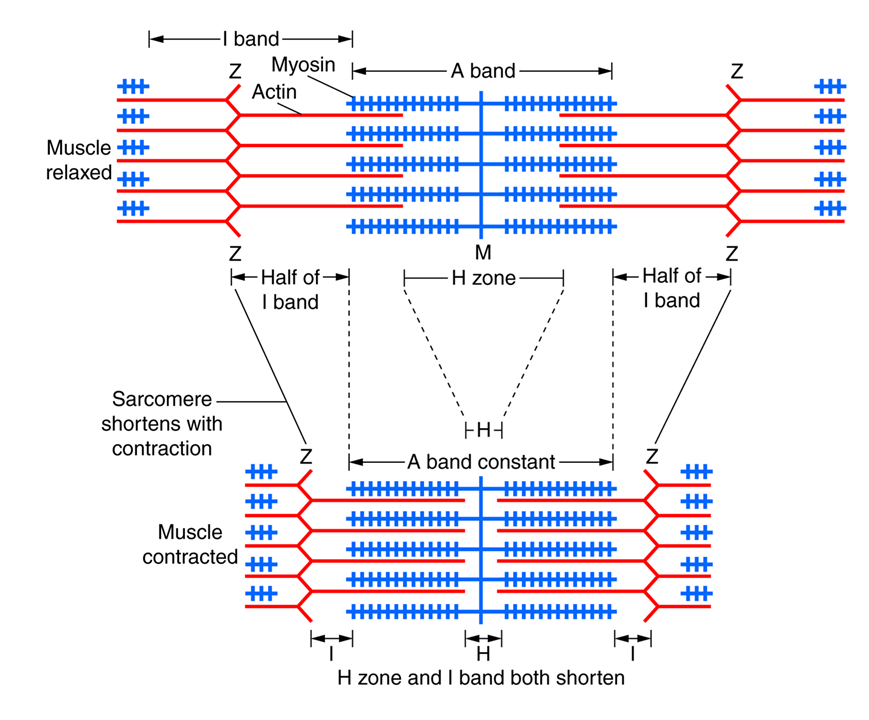
A muscle (or more specifically: a sarcomere) keeps shortening as long as
- Calcium is present to bind to troponin and uncover the binding sites on actin; and
- ATP is available to fuel the pulling action of myosin; or
- Until the thick filaments (myosin) bump up against the Z-lines, which is the built-in stop point (see image on the right).
Expansion and contraction of the sarcomere occur only within the I band and H zone (both shortening or disappearing). The myofilaments themselves do not change in length, so the A band remains constant.
👉 In summary: The sliding filament theory is the conceptual framework that explains how sarcomeres shorten through changes in the H and I bands. The cross-bridge cycle is the microscopic engine — the molecular mechanism — by which myosin and actin filaments slide over each other.
Watch this short video to review the mechanism of muscle contraction:


