23.3A: Lipid Molecules
- Page ID
- 8100
Fats and oils, which may be saturated or unsaturated, can be unhealthy but also serve important functions for plants and animals.
- Differentiate between saturated and unsaturated fatty acids
Key Points
- Fats provide energy, insulation, and storage of fatty acids for many organisms.
- Fats may be saturated (having single bonds) or unsaturated (having double bonds).
- Unsaturated fats may be cis (hydrogens in same plane) or trans (hydrogens in two different planes).
- Olive oil, a monounsaturated fat, has a single double bond whereas canola oil, a polyunsaturated fat, has more than one double bond.
- Omega-3 fatty acid and omega-6 fatty acid are essential for human biological processes, but they must be ingested in the diet because they cannot be synthesized.
Key Terms
- hydrogenation: The chemical reaction of hydrogen with another substance, especially with an unsaturated organic compound, and usually under the influence of temperature, pressure and catalysts.
- ester: Compound most often formed by the condensation of an alcohol and an acid, by removing water. It contains the functional group carbon-oxygen double bond joined via carbon to another oxygen atom.
- carboxyl: A univalent functional group consisting of a carbonyl and a hydroxyl functional group (-CO.OH); characteristic of carboxylic acids.
Fats have important functions, and many vitamins are fat soluble. Fats serve as a long-term storage form of fatty acids and act as a source of energy. They also provide insulation for the body.
Glycerol and Fatty Acids
A fat molecule consists of two main components: glycerol and fatty acids. Glycerol is an alcohol with three carbons, five hydrogens, and three hydroxyl (OH) groups. Fatty acids have a long chain of hydrocarbons with a carboxyl group attached and may have 4-36 carbons; however, most of them have 12-18. In a fat molecule, the fatty acids are attached to each of the three carbons of the glycerol molecule with an ester bond through the oxygen atom. During the ester bond formation, three molecules are released. Since fats consist of three fatty acids and a glycerol, they are also called triacylglycerols or triglycerides.
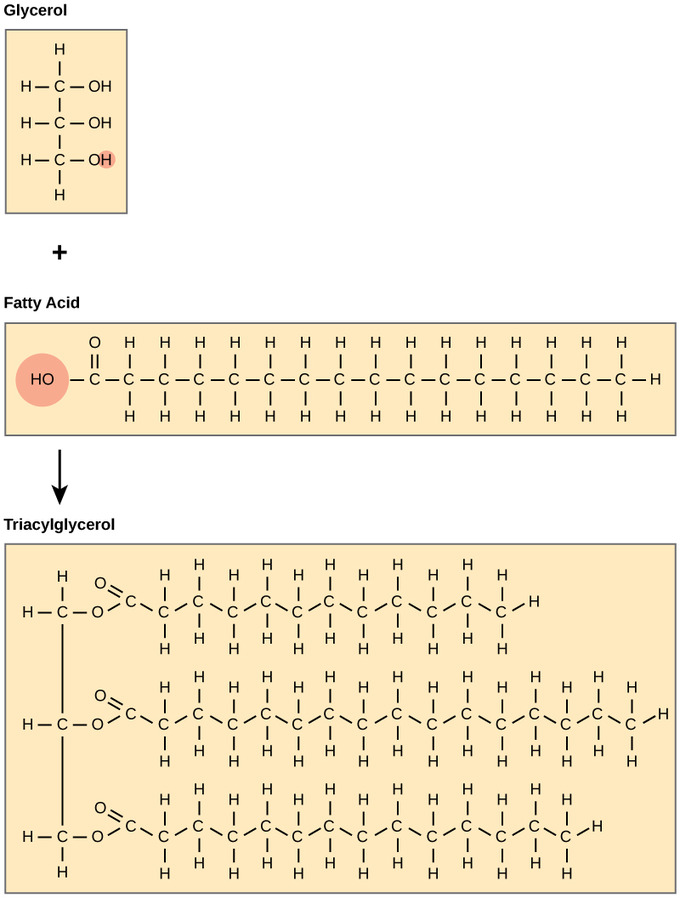
Triacylglycerols: Triacylglycerol is formed by the joining of three fatty acids to a glycerol backbone in a dehydration reaction. Three molecules of water are released in the process.
Saturated vs. Unsaturated Fatty Acids
Fatty acids may be saturated or unsaturated. In a fatty acid chain, if there are only single bonds between neighboring carbons in the hydrocarbon chain, the fatty acid is said to be saturated. Saturated fatty acids are saturated with hydrogen since single bonds increase the number of hydrogens on each carbon. Stearic acid and palmitic acid, which are commonly found in meat, are examples of saturated fats.
When the hydrocarbon chain contains a double bond, the fatty acid is said to be unsaturated. Oleic acid is an example of an unsaturated fatty acid. Most unsaturated fats are liquid at room temperature and are called oils. If there is only one double bond in the molecule, then it is known as a monounsaturated fat; e.g. olive oil. If there is more than one double bond, then it is known as a polyunsaturated fat; e.g. canola oil. Unsaturated fats help to lower blood cholesterol levels whereas saturated fats contribute to plaque formation in the arteries.
Unsaturated fats or oils are usually of plant origin and contain cis unsaturated fatty acids. Cis and trans indicate the configuration of the molecule around the double bond. If hydrogens are present in the same plane, it is referred to as a cis fat; if the hydrogen atoms are on two different planes, it is referred to as a trans fat. The cis double bond causes a bend or a “kink” that prevents the fatty acids from packing tightly, keeping them liquid at room temperature.
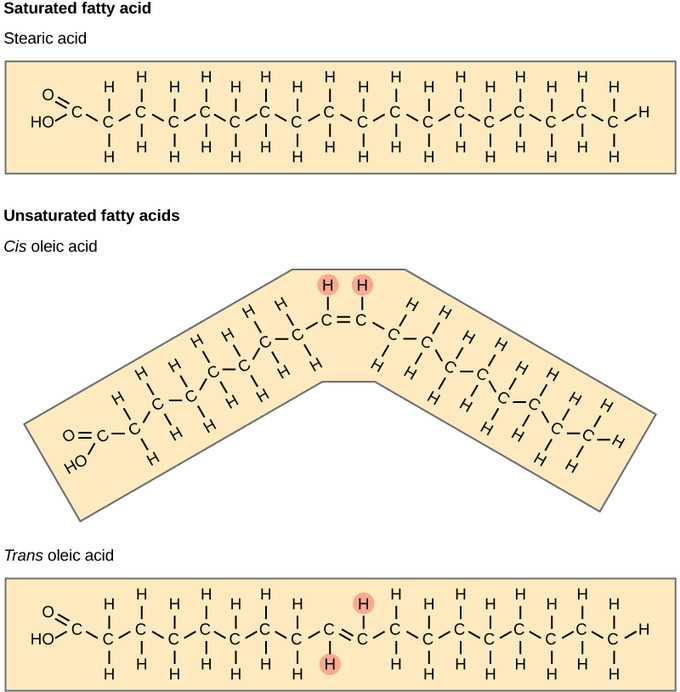
Fatty Acids: Saturated fatty acids have hydrocarbon chains connected by single bonds only. Unsaturated fatty acids have one or more double bonds. Each double bond may be in a cis or trans configuration. In the cis configuration, both hydrogens are on the same side of the hydrocarbon chain. In the trans configuration, the hydrogens are on opposite sides. A cis double bond causes a kink in the chain.
Trans Fats
In the food industry, oils are artificially hydrogenated to make them semi-solid and of a consistency desirable for many processed food products. During this hydrogenation process, gas is bubbled through oils to solidify them, and the double bonds of the cis-conformation in the hydrocarbon chain may be converted to double bonds in the trans-conformation.
Margarine, some types of peanut butter, and shortening are examples of artificially-hydrogenated trans fats. Recent studies have shown that an increase in trans fats in the human diet may lead to an increase in levels of low-density lipoproteins (LDL), or “bad” cholesterol, which in turn may lead to plaque deposition in the arteries, resulting in heart disease. Many fast food restaurants have recently banned the use of trans fats, and food labels are required to display the trans fat content.
Essential Fatty Acids
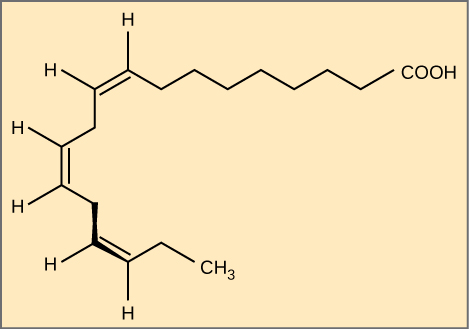
Omega Fatty Acids: Alpha-linolenic acid is an example of an omega-3 fatty acid. It has three cis double bonds and, as a result, a curved shape. For clarity, the carbons are not shown. Each singly bonded carbon has two hydrogens associated with it, also not shown.
Essential fatty acids are fatty acids required for biological processes, but not synthesized by the human body. Consequently, they have to be supplemented through ingestion via the diet and are nutritionally very important. Omega-3 fatty acid, or alpha-linoleic acid (ALA), falls into this category and is one of only two fatty acids known to be essential for humans (the other being omega-6 fatty acid, or linoleic acid). These polyunsaturated fatty acids are called omega-3 because the third carbon from the end of the hydrocarbon chain is connected to its neighboring carbon by a double bond. Salmon, trout, and tuna are good sources of omega-3 fatty acids.
Research indicates that omega-3 fatty acids reduce the risk of sudden death from heart attacks, reduce triglycerides in the blood, lower blood pressure, and prevent thrombosis by inhibiting blood clotting. They also reduce inflammation and may help reduce the risk of some cancers in animals.

