4.2: Basic Concepts
- Page ID
- 92815
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Indications for CVAD
Common indications for CVAD placement include delivery of medication, fluids, and nutrition, especially for clients requiring long-term therapy. CVADs are required for infusion of high osmolarity solutions and vesicant medications. They may also be placed for emergency venous access for clients requiring fluid resuscitation and hemodynamic monitoring.[1] Many clients who require CVADs are older adults, very young children, or those with chronic health conditions.[2]
High osmolarity solutions refer to a highly concentrated solution expressed as the total number of solute particles per liter. High osmolarity solutions, such as total parenteral nutrition and hypertonic IV fluids, are irritating to peripheral vessels and increase the client’s risk for phlebitis, thrombosis, and occlusion. Additionally, vesicant medications (such as certain antineoplastic drugs, antibiotics, electrolytes, and vasopressors) can cause severe tissue injury or destruction if they extravasate. Extravasation refers to leakage of fluid into the tissues around the IV site, causing tissue injury when the catheter has dislodged from the blood vessel but is still in the nearby tissue. For this reason, infusions of high osmolarity solutions and vesicant medications are administered through a CVAD into a large vein such as the superior vena cava. When these solutions enter this larger vessel, the solution is hemodiluted, thus minimizing the risk of these complications from occurring.
Fluid resuscitation refers to infusing a large volume of fluid through the intravenous venous access to restore hemodynamics and optimize tissue perfusion and, ultimately, tissue oxygen delivery. Hemodynamic monitoring is often in place when a client requires fluid resuscitation. Hemodynamic monitoring is the assessment of a critically ill client’s circulatory status and includes measurements of central venous pressure, cardiac output, and blood volume.[3] Central venous pressure (CVP) reflects the pressure in the central veins as they enter the right atrium and is often monitored during fluid resuscitation as measure of preload (i.e., volume status).
Types of Central Venous Access Devices and Locations
There are several types of CVADs, and the selection of which type is used depends upon the specific client’s clinical situation, indication, and duration of treatment. The type of CVAD selected is based on the specific client’s clinical situation. The decision process for selecting an appropriate CVAD involves collaboration among the provider, the client, and the health care team while considering the treatment requirements. Special considerations include examining the expected length of treatment; the specific prescribed treatment; and the client’s vascular characteristics, age, cognitive level, medical history, infusion therapy history, and, if appropriate, their preference for the CVAD site location. Generally, other venous access or alternative delivery methods should be considered prior to inserting a CVAD due to its invasiveness and associated risks. Indications for CVAD insertion and its associated risks should be explained to the client by the provider and documented in their medical record.[4]
CVADs may be inserted centrally or peripherally. A centrally inserted central venous catheter is typically placed into the client’s internal jugular, subclavian, or femoral vein. Peripherally inserted central venous catheters (referred to as PICC lines) are primarily placed through the basilic, cephalic, or brachial veins. Insertion is more successful with fewer complications when guided ultrasound is used for placement.[5]
Only specially trained health care clinicians can select and insert CVADs. The determination of which area (peripheral, midline, or central vein) used to insert a CVAD is based on a suitable venous pathway, optimal vein characteristics, risk of nerve injury, and anatomical variations.[6]
Table 4.2a outlines various types of CVADs, their uses, expected durations, site considerations, and client safety considerations.
| Device | Type of Therapy | Expected Duration | Site Considerations | Rationale and Safety Considerations |
|---|---|---|---|---|
| Peripherally Inserted Central Catheter (PICC)(See Figure 4.2[7]) | Long-term use. May be used to infuse high osmolarity solutions or antibiotic therapy. Power ports may be used for high pressure rapid infusions. | Up to six months. | Utilize median cubital, cephalic, basilic, or brachial veins with sufficient diameter size. | Avoid in clients with end-stage renal disease requiring vein preservation for fistulas and grafts or those with a history of thrombosis, hypercoagulability states, or decreased peripheral vascular flow. |
| Non-Tunneled CVAD(See Figure 4.3[8]) | May be used to infuse high osmolarity solutions. | Days to several weeks. | Insertion sites may be subclavian, external/internal jugular, or femoral veins. | The subclavian vein is favored in adult clients due to decreased risk of catheter-related thrombosis and/or infection. |
| External Tunneled CVAD (Hickman, Broviac, Groshong)(See Figure 4.4[9]) | Long-term intravenous therapy, such as chemotherapy or hemodialysis. | May be long-term or permanent. | Inserted in the chest area via a subclavian or jugular vein. Tunneled subcutaneously from the proximal end of the insertion site to an exit site. | Surgery is required to tunnel the catheter so that part of the catheter lies in the subcutaneous tunnel. This helps prevent organisms from entering the bloodstream by separating where the catheter exists the skin from where it enters the vein. Passing the catheter under the skin also helps keep it better secured. |
| Implanted Venous Access Device (IVAD), also referred to as an Implanted Port(See Figure 4.5[10]) | Long-term medication or IV therapy such as chemotherapy. | May be long-term or permanent. | Placed in a subclavian or jugular vein and attached to a reservoir pocket. The reservoir pocket is a small plastic or metal cylinder, usually placed just below the collar bone, that releases medication into the bloodstream. An IVAD is less obvious than a tunneled catheter and requires little daily care, with less impact on a person’s activities than a PICC line or a tunneled catheter. | Surgery is required to place the port. The port is accessed with a noncoring needle and can be used immediately after placement. |




Lumens
CVADs may have a single lumen (opening), double lumen, or multiple lumens that exit at various places along the central catheter. See Figure 4.6[11] for an image of a CVAD with two lumens.

Table 4.2b describes different types of lumens based on their exit points, their size, and their uses.
| Lumens | Proximal Lumen | Middle Lumen | Distal Lumen |
|---|---|---|---|
| Size | 18 gauge | 18 gauge | 16 gauge |
| Uses | Fluids
TPN/Lipids Medications |
Medications | Blood draw
Blood administration Central venous pressure (CVP) monitoring |
Insertion of a CVAD
The insertion of a CVAD is an invasive medical procedure requiring informed consent from the client. The insertion should be performed only by a trained, credentialed health professional. Accurate placement of the CVAD tip is confirmed according to agency policy by fluoroscopy during insertion, post-procedure chest X-ray, or a magnet tip locator. Fluoroscopy is an imaging technique that uses X-rays to obtain real-time moving images of the interior of an object within the body. If real-time fluoroscopy is used during the procedure to confirm tip placement, a post-procedure chest X-ray is not required. If fluoroscopy is not used, a post-procedure X-ray is used to confirm tip placement, as well as to check for a possible pneumothorax that can inadvertently occur during insertion. After the tip location is verified, it is essential to document the location in the client’s medical record.[12]
Securement of CVADs
After the placement of the CVAD tip is confirmed, the CVAD must be stabilized and secured to the client. Dislodgement and premature removal of the CVAD increase complications such as infections, vessel injury, and treatment delays. Depending on the location and type of CVAD, it may be stabilized with sutures or a sutureless engineered stabilization device. See Figure 4.7[13] for illustrations of stabilization devices.
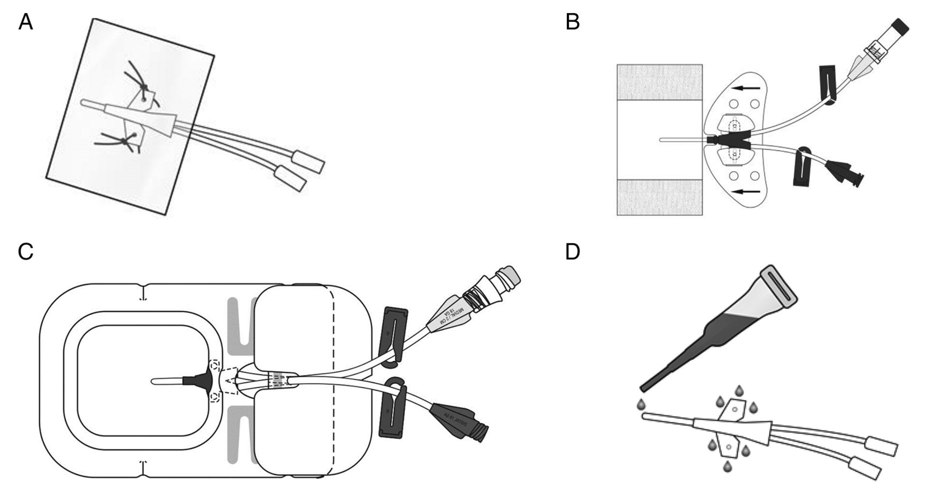
The goal of the securement device is to maintain a secure hold on the central line and to prevent it from moving in and out of the insertion site. Sutureless devices, if appropriate, have less risk of infection because they maintain intact skin. Adhesive devices have the risk of causing a skin-related injury such as skin tear or a local reaction to the adhesive. Applying a prophylactic skin barrier prior to applying an adhesive device decreases this risk to the client.
After the catheter is secured, a sterile, transparent semipermeable dressing is applied to cover the insertion site. Some transparent dressings have an impregnated chlorhexidine gel or a biopatch that is directly placed over the insertion site to reduce the growth of microorganisms. In some cases, if there is blood or exudate leaking from the insertion site after initial placement, a sterile gauze dressing may be used to absorb the fluid until the leaking resolves.
Prevention of Central Line-Associated Bloodstream Infections (CLABSI)
When a client has a CVAD, it is crucial to follow evidence-based guidelines regarding its insertion, care, and maintenance to prevent the development of a life-threatening infection. Clients with CVADs are at risk for developing central line-associated bloodstream infections (CLABSI). CLABSI is diagnosed when pathogens are found in the client’s blood in the absence of another source of infection and the client has had a central line in place for more than two calendar days before the infection occurs. CLABSI continues to be one of the most deadly and costly hospital-acquired infections in the United States.[14]
A group of evidence-based practice standards has been formulated by the Institute of Healthcare Improvement (IHI) to reduce the risk of infection related to the insertion and management of CVADs and improve quality of care. CLABSI prevention strategies encompass three areas: clinical indications, insertion, and care and maintenance. These strategies include the following elements[15],[16]:
- Hand hygiene as directed by CDC guidelines
- Maximal barrier precautions during insertion
- Skin antiseptic using chlorhexidine
- Optimal site selection, such as avoiding femoral vein access, when possible, for central venous access in adults
- Daily assessment of the necessity of the central line and prompt removal if deemed unnecessary
- Routine disinfection of catheter hubs, connectors, and injection ports
- Changing dressings over the site every two days for gauze dressings, every seven days for semipermeable dressings, or as needed if it becomes damp, loose, or visibly soiled
There have been improvements in CLABSI rates with these IHI practice standards, but, unfortunately, CLABSI continues to be an issue in hospitals despite these prevention measures. Read more information regarding the CDC recommendations to prevent CLABSIs in the following box.
Read the Infection Control Guidelines for Central Lines from the CDC’s web page.
Potential Complications and Unexpected Outcomes of CVADs
Nursing management of clients with CVADs requires strict asepsis, knowledge of the purpose and functions of these devices, and routine interventions to prevent complications. Ongoing assessment and monitoring of the client and the CVAD system are essential for safety, quality care, and positive client outcomes. The following table summarizes possible complications associated with CVADs, their assessments, related prevention actions, and associated nursing interventions.
| Potential Complication | Assessment | Prevention | Nursing Intervention |
|---|---|---|---|
| Occlusion due to clot formation or malpositioning | Perform recommended site and CVAD system care, including equipment function and checking for blood return and the ability to infuse fluid. Assess for pain and edema at the insertion site (i.e., shoulder, ear, neck, or arm). If an implantable port is in place, assess the noncoring needle for correct placement. | Flush the catheter routinely as recommended and according to agency policy. Do not flush against resistance. Keep the catheter free from kinks. Do not mix incompatible medications during infusion that can cause precipitation within the catheter. | Initially, reposition the client. Raise the client’s arm overhead, ask the client to cough and deep breathe, or assist them to stand up and sit down. If appropriate, administer thrombolytics as ordered by the provider. A clogged CVAD may require removal by trained health care personnel per provider order. |
| Catheter damage or breakage | Assess the site every shift and with flushing. Observe for leaks, tears, pinholes, or drainage after flushing. | Using a 10-mL syringe is preferred for CVADs to avoid increased pressure that can cause a potential rupture. Never flush against resistance. Use needleless system devices and avoid sharp objects such as scissors near the catheter. Follow agency policy regarding the proper clamping procedure if the access device has a closed catheter system. | Clamp the catheter near the insertion site and place sterile gauze over the break or hole until it is repaired. If repair of the catheter is safe and appropriate, use only a repair kit that is recommended by the manufacturer. The CVAD may require removal by trained health care personnel per provider order. |
| Infection (CLABSI) | Assess the catheter insertion site and surrounding area for redness, edema, drainage, and tenderness. Monitor pertinent laboratory results (e.g., WBC). | Maintain and utilize aseptic technique. Comply with guidelines and agency policies regarding CVAD and follow the recommended CLABSI prevention bundle. | Notify the health care provider and anticipate a blood culture order for suspected CLABSI. Follow agency sepsis prevention and implementation protocol. The CVAD may require removal by trained health care personnel per provider order. Anticipate obtaining a culture of the CVAD tip using sterile scissors and a sterile specimen cup. Diagnosed CLABSIs should be treated as life-threatening. Antibiotics specific to the organism should be initiated. Infection preventionists, vascular access specialists, providers involved with device insertion, and primary nursing staff should review each case in detail, looking for potential contributing factors.[17] |
| Dislodgment | Measure and document the catheter length per agency policy. Assess for dislodgement by identifying any edema at or around the catheter insertion site. Palpate for coiling of the catheter under the skin. | Ensure the catheter is secured at all times and the dressing is intact. Avoid pulling and manipulating the catheter. | If the catheter is completely dislodged, cover the insertion site and apply direct manual pressure while asking a colleague to call the rapid response team. The client will require monitoring for possible air embolus and reinsertion of a CVAD for critical medications. |
| Catheter migration (i.e., the catheter moved from its original position) | Assess the patency of the catheter, noting local irritation, swelling, or the inability to aspirate blood. Assess for edema of the arm and hand and distended neck veins. May be able to hear “gurgling” sounds from the catheter. If the catheter tip has advanced into the heart, cardiac dysrhythmias may occur. | Avoid site insertion of a CVAD in areas near the site of a local infection, disrupted skin integrity, or scar tissue. | Notify the provider. A catheter that has migrated externally from its original placement should not be readvanced. A catheter that has migrated internally should be retracted to the original insertion length by a trained health professional. |
| Skin erosion | Assess the skin at and around the CVAD insertion site. Note any skin separation from the catheter exit site, drainage, contusions, or any indication of skin involvement. | Maintain optimal client nutritional status. For implanted ports, avoid using the same insertion “hole” when accessing it multiple times because this increases the risk of tissue and port breakdown. | Plan for removal of the CVAD per order. Provide effective skin care. Improve nutrition as appropriate to the client’s condition. |
| Air embolism[18] | An air embolism is the result of a pressure gradient that allows air to enter the bloodstream when flushing or removing the catheter. An air embolism can subsequently occlude blood flow. Signs and symptoms include sudden dyspnea, continuous coughing, and chest pain. Neurological symptoms include seizures, loss of consciousness, altered mental status, and hemiparesis. | Catheter hubs should not be open to air. Ensure all clamps are engaged appropriately for the device. | Call the rapid response team if an air embolism is suspected. Prevent further air embolism if a clamp is not engaged or a hub is open to air. Administer high-flow oxygen and place the client on their left side with their head down. Begin CPR if indicated. |
| Pneumothorax[19] | Pneumothorax may inadvertently occur during insertion of a CVAD if the needle in the CVAD placed in the neck or chest goes through the vein or misses the vein and pierces the lung, causing it to collapse. Symptoms of a pneumothorax include sharp, stabbing chest pain that worsens when trying to breathe in; shortness of breath; cyanosis; tachypnea; and a dry, hacking cough. | Not applicable. | Call for assistance and ask a colleague to call the rapid response team and notify the provider. Stay with the patient and administer high-flow oxygen. Anticipate placement of a chest tube if the client is hypoxic. |
| Infiltration or extravasation | Palpate over the catheter insertion site dressing and around the surrounding area for sponginess and observe for redness or swelling. Note any labored breathing exhibited by the client or complaints of pain with infusions. Observe IV flow rate for free-flowing fluid. Aspirate for blood return. | Stop the infusion and/or administration of the vesicant solution. If extravasation occurs, aspirate any remaining medication from the catheter after disconnection to prevent further damage to vessels. To maintain skin integrity, administer antidote or therapeutic medication as appropriate per protocol.[20] | Discontinue IV solutions. Apply warm/cold compresses as recommended by agency policy. Notify the provider and anticipate an order for a chest X-ray to evaluate catheter integrity and placement. |
| Incorrect placement | Assess for inadequate blood withdrawal, blood flowing back into the tubing, hypotension, cardiac dysrhythmias, and neck vein distension. | Verification of catheter placement by chest X-ray after insertion or during real-time guided fluoroscopy. | Stop all fluid and medication administration. Anticipate orders for an X-ray and electrocardiogram. The CVAD may require removal or withdrawal to the correct position by trained health care personnel per provider order. |
![]() PICC Line Care[21]
PICC Line Care[21]
- Avoid venipuncture, peripheral intravenous cannula insertion, and taking blood pressure on the same arm where the PICC is located. Place reminder signs for the health care team members according to agency policy.
- Ensure the PICC line dressing stays dry during showering.
- Avoid bandages or tight coverings over the PICC line insertion point. Tight elastic coverings can increase the risk of compressing the vein, leading to vein wall irritation, phlebitis, or thrombosis.
- This work is a derivative of StatPearls by Tse and Schick and is licensed under CC BY 4.0 ↵
- Broadhurst, D., Moureau, N., & Ullman, A. J. The World Congress of Vascular Access (WoCoVA) Skin Impairment Management Advisory Panel. (2017). Management of central venous access device-associated skin impairment: An evidence-based algorithm. Journal of Wound, Ostomy, and Continence Nursing, 44(3), 211-220. https://journals.lww.com/jwocnonline/fulltext/2017/05000/management_of_central_venous_access.2.aspx ↵
- McCarthy, C. J., Behravesh, S., Naidu, S. G., & Oklu, R. (2016). Air embolism: Practical tips for prevention and treatment. Journal of Clinical Medicine, 5(11), 93. https://doi.org/10.3390/jcm5110093 ↵
- NSW Agency for Clinical Innovation. (2021). Central venous access devices (CVAD): Clinical practice guide. Agency for Clinical Innovation. https://aci.health.nsw.gov.au/__data/assets/pdf_file/0010/239626/ACI-CVAD-clinical-practice-guide.pdf ↵
- Chopra, V. (2022). Central venous access devices and approach to device and site selection in humans. UpToDate. Retrieved November 28, 2022, from https://www.uptodate.com/ ↵
- NSW Agency for Clinical Innovation. (2021). Central venous access devices (CVAD): Clinical practice guide. Agency for Clinical Innovation. https://aci.health.nsw.gov.au/__data/assets/pdf_file/0010/239626/ACI-CVAD-clinical-practice-guide.pdf ↵
- "Blausen_0193_Catheter_PICC.png" by Blausen Medical Communications, Inc. is licensed under CC BY 3.0 ↵
- "Blausen_0181_Catheter_CentralVenousAccessDevice_NonTunneled.png" by Blausen.com staff (2014) for "Medical gallery of Blausen Medical 2014" is licensed under CC BY 3.0 ↵
- "Tunneled_venous_access_device.png" by Glynda Rees Doyle and Jodie Anita McCutcheon is licensed under CC BY 4.0 ↵
- "Venous_Access_Port_Catheter.png" by BruceBlaus is licensed under CC BY-SA 4.0 ↵
- "xvthfl4exkgi9wdyt2twbf18gfpi3hdf.jpg" by unknown author used on the basis of Fair Use. Access original image at https://www.bd.com/en-us/products-and-solutions/products/product-families/powerline-central-venous-catheter#eifuresources. ↵
- NSW Agency for Clinical Innovation. (2021). Central venous access devices (CVAD): Clinical practice guide. Agency for Clinical Innovation. https://aci.health.nsw.gov.au/__data/assets/pdf_file/0010/239626/ACI-CVAD-clinical-practice-guide.pdf ↵
- "F1.medium.png" by Amanda Ullman et al., courtesy of BMJ Open is licensed under CC BY-NC 4.0. Access for free at https://bmjopen.bmj.com/content/6/6/e011197 ↵
- Institute for Healthcare Improvement. (n.d.). Central line infection. https://www.ihi.org/Topics/CentralLineInfection/Pages/default.aspx ↵
- Institute for Healthcare Improvement. (n.d.). Central line infection. https://www.ihi.org/Topics/CentralLineInfection/Pages/default.aspx ↵
- The Joint Commission. (n.d.) CLABSI toolkit - Chapter 3. https://www.jointcommission.org/resources/patient-safety-topics/infection-prevention-and-control/central-line-associated-bloodstream-infections-toolkit-and-monograph/clabsi-toolkit---chapter-3/ ↵
- DeVries, M. (2019). Revisiting CLABSI prevention strategies: Part 2 Learn about central line care and maintenance. American Nurse Today, 14(6), 44-47. www.myamericannurse.com/wp-content/uploads/2019/06/ant6-INFECTION-CLABSI-2-521a.pdf ↵
- McCarthy, C. J., Behravesh, S., Naidu, S. G., & Oklu, R. (2016). Air embolism: Practical tips for prevention and treatment. Journal of Clinical Medicine, 5(11), 93. https://doi.org/10.3390/jcm5110093 ↵
- Tsotsolis, N., Tsirgogianni, K., Kioumis, I., Pitsiou, G., Baka, S., Papaiwannou, A., Karavergou, A., Rapti, A., Trakada, G., Katsikogiannis, N., Tsakiridis, K., Karapantzos, I., Karapantzou, C., Barbetakis, N., Zissimopoulos, A., Kuhajda, I., Andjelkovic, D., Zarogoulidis, K., & Zarogoulidis, P. (2015). Pneumothorax as a complication of central venous catheter insertion. Annals of Translational Medicine, 3(3), 40. https://doi.org/10.3978/j.issn.2305-5839.2015.02.11 ↵
- Kim, J. T., Park. J. Y., Lee, H. J., & Cheon, Y. J. (2020). Guidelines for the management of extravasation. Journal of Educational Evaluation for Health Professions, 17:21.www.ncbi.nlm.nih.gov/pmc/articles/PMC7431942/ ↵
- Cancer Institute NSW. (2021, July 19). Central venous access devices. https://www.eviq.org.au/clinical-resources/central-venous-access-devices-cvads/112-central-venous-access-devices#key-practice-points-for-clinical-procedures ↵

