15.3: Intravenous Solutions
- Page ID
- 52498
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)When patients experience deficient fluid volume, intravenous (IV) fluids are often prescribed. IV fluid restores fluid to the intravascular compartment, and some IV fluids are also used to facilitate the movement of fluid between compartments due to osmosis. There are three types of IV fluids: isotonic, hypotonic, and hypertonic.
Isotonic Solutions
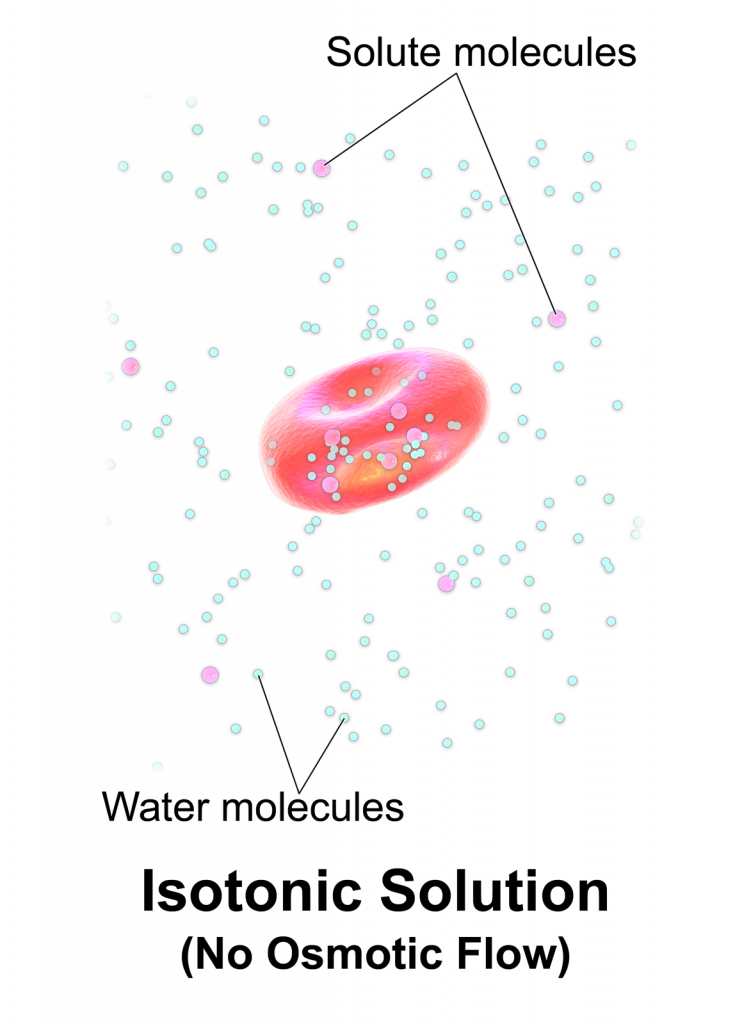
Isotonic solutions are IV fluids that have a similar concentration of dissolved particles as blood. An example of an isotonic IV solution is 0.9% Normal Saline (0.9% NaCl). Because the concentration of the IV fluid is similar to the blood, the fluid stays in the intravascular space and osmosis does not cause fluid movement between compartments. See Figure 15.8[1] for an illustration of isotonic IV solution administration with no osmotic movement of fluid with cells. Isotonic solutions are used for patients with fluid volume deficit (also called hypovolemia) to raise their blood pressure. However, infusion of too much isotonic fluid can cause excessive fluid volume (also referred to as hypervolemia).
Hypotonic Solutions
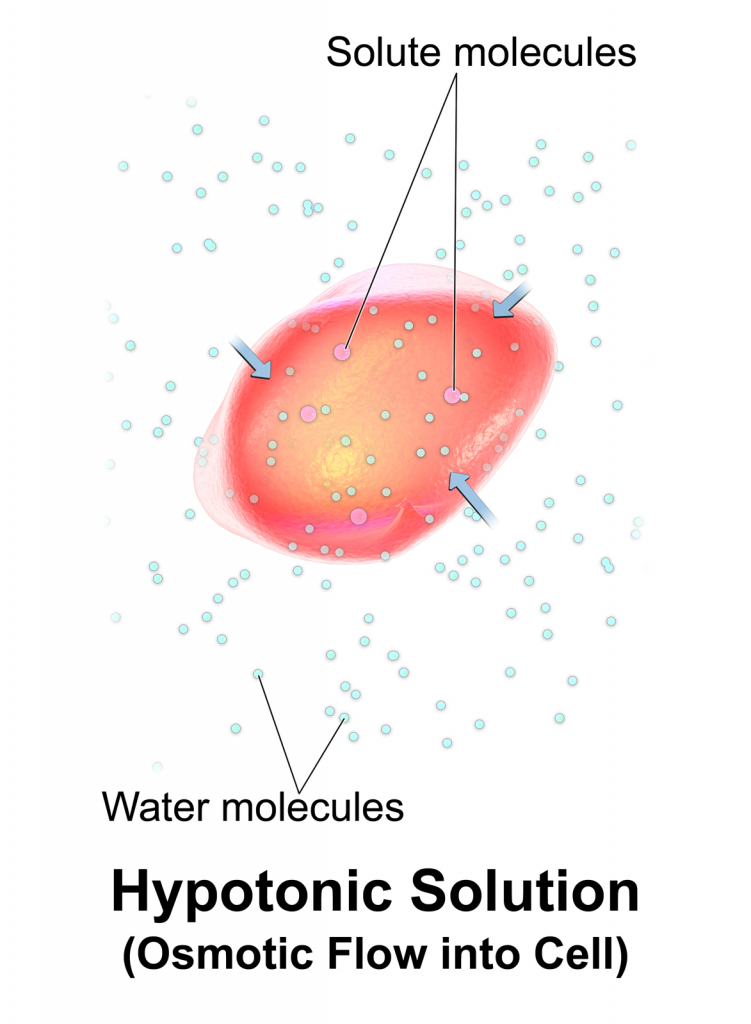
Hypotonic solutions have a lower concentration of dissolved solutes than blood. An example of a hypotonic IV solution is 0.45% Normal Saline (0.45% NaCl). When hypotonic IV solutions are infused, it results in a decreased concentration of dissolved solutes in the blood as compared to the intracellular space. This imbalance causes osmotic movement of water from the intravascular compartment into the intracellular space. For this reason, hypotonic fluids are used to treat cellular dehydration. See Figure 15.9[2] for an illustration of the osmotic movement of fluid into a cell when a hypotonic IV solution is administered, causing lower concentration of solutes (pink molecules) in the bloodstream compared to within the cell.
However, if too much fluid moves out of the intravascular compartment into cells, cerebral edema can occur. It is also possible to cause worsening hypovolemia and hypotension if too much fluid moves out of the intravascular space and into the cells. Therefore, patient status should be monitored carefully when hypotonic solutions are infused.
Hypertonic Solutions
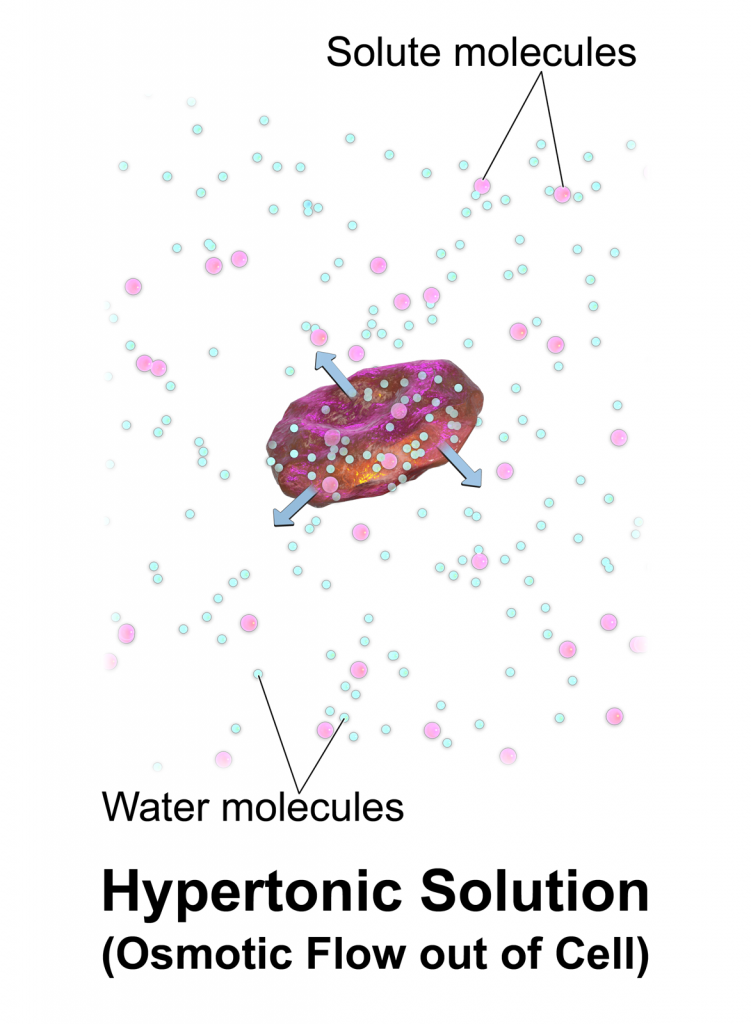
Hypertonic solutions have a higher concentration of dissolved particles than blood. An example of hypertonic IV solution is 3% Normal Saline (3% NaCl). When infused, hypertonic fluids cause an increased concentration of dissolved solutes in the intravascular space compared to the cells. This causes the osmotic movement of water out of the cells and into the intravascular space to dilute the solutes in the blood. See Figure 15.10[3] for an illustration of osmotic movement of fluid out of a cell when hypertonic IV fluid is administered due to a higher concentration of solutes (pink molecules) in the bloodstream compared to the cell.
When administering hypertonic fluids, it is essential to monitor for signs of hypervolemia such as breathing difficulties and elevated blood pressure. Additionally, if hypertonic solutions with sodium are given, the patient’s serum sodium level should be closely monitored.[4] See Table 15.3 for a comparison of types of IV solutions, their uses, and nursing considerations.
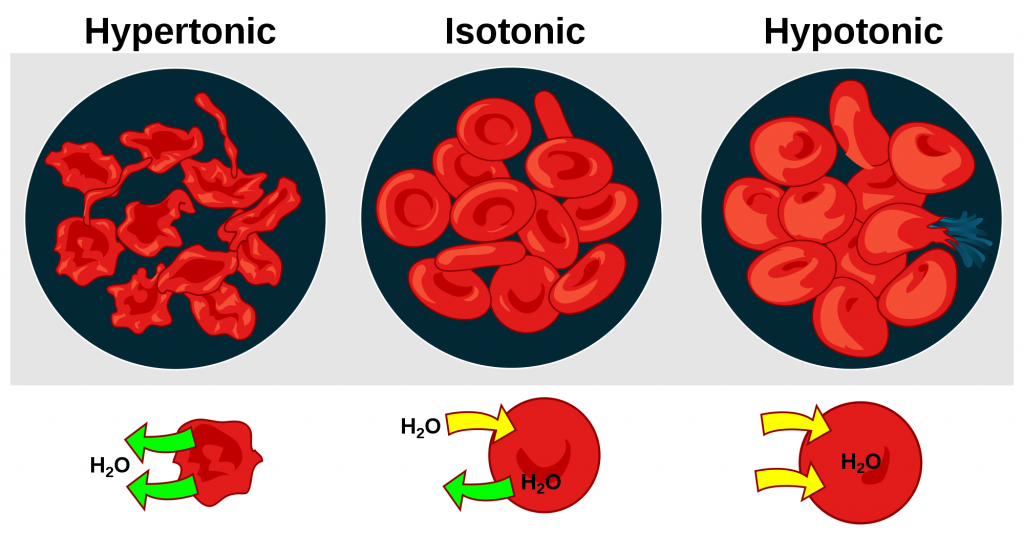
See Figure 15.11[5] for an illustration comparing how different types of IV solutions affect red blood cell size.
| Type | IV Solution | Uses | Nursing Considerations |
|---|---|---|---|
| Isotonic | 0.9% Normal Saline (0.9% NaCl) | Fluid resuscitation for hemorrhaging, severe vomiting, diarrhea, GI suctioning losses, wound drainage, mild hyponatremia, or blood transfusions. | Monitor closely for hypervolemia, especially with heart failure or renal failure. |
| Isotonic | Lactated Ringer’s Solution (LR) | Fluid resuscitation, GI tract fluid losses, burns, traumas, or metabolic acidosis. Often used during surgery. | Should not be used if serum pH is greater than 7.5 because it will worsen alkalosis. May elevate potassium levels if used with renal failure. |
| Isotonic | 5% Dextrose in Water (D5W) *starts as isotonic and then changes to hypotonic when dextrose is metabolized | Provides free water to help renal excretion of solutes, hypernatremia, and some dextrose supplementation. | Should not be used for fluid resuscitation because after dextrose is metabolized, it becomes hypotonic and leaves the intravascular space, causing brain swelling. Used to dilute plasma electrolyte concentrations. |
| Hypotonic | 0.45% Sodium Chloride (0.45% NaCl) | Used to treat intracellular dehydration and hypernatremia and to provide fluid for renal excretion of solutes. | Monitor closely for hypovolemia, hypotension, or confusion due to fluid shifting into the intracellular space, which can be life-threatening. Avoid use in patients with liver disease, trauma, and burns to prevent hypovolemia from worsening. Monitor closely for cerebral edema. |
| Hypotonic | 5% Dextrose in Water (D5W) | Provides free water to promote renal excretion of solutes and treat hypernatremia, as well as some dextrose supplementation. | Monitor closely for hypovolemia, hypotension, or confusion due to fluid shifting out of the intravascular space, which can be life-threatening. Avoid use in patients with liver disease, trauma, and burns to prevent hypovolemia from worsening. Monitor closely for cerebral edema. |
| Hypertonic | 3% Sodium Chloride (3% NaCl) | Used to treat severe hyponatremia and cerebral edema. | Monitor closely for hypervolemia, hypernatremia, and associated respiratory distress. Do not use it with patients experiencing heart failure, renal failure, or conditions caused by cellular dehydration because it will worsen these conditions. |
| Hypertonic | 5% Dextrose and 0.45% Sodium Chloride (D50.45% NaCl) | Used to treat severe hyponatremia and cerebral edema. | Monitor closely for hypervolemia, hypernatremia, and associated respiratory distress. Do not use it with patients experiencing heart failure, renal failure, or conditions caused by cellular dehydration because it will worsen these conditions. |
| Hypertonic | 5% Dextrose and Lactated Ringer’s (D5LR)
D10 |
Used to treat severe hyponatremia and cerebral edema. | Monitor closely for hypervolemia, hypernatremia, and associated respiratory distress. Do not use it with patients experiencing heart failure, renal failure, or conditions caused by cellular dehydration because it will worsen these conditions. |
Osmolarity is defined as the proportion of dissolved particles in an amount of fluid and is generally the term used to describe body fluids. As the dissolved particles become more concentrated, the osmolarity increases. Osmolality refers to the proportion of dissolved particles in a specific weight of fluid. The terms osmolarity and osmolality are often used interchangeably in clinical practice.
- “Blausen_0685_OsmoticFlow_Isotonic.png” by BruceBlaus.com staff is licensed under CC BY 3.0↵
- “Blausen_0684_OsmoticFlow_Hypotonic.png” by BruceBlaus.com staff is licensed under CC BY 3.0↵
- “Blausen_0683_OsmoticFlow_Hypertonic.png” by BruceBlaus.com staff is licensed under CC BY 3.0↵
- Harris, H. (2011). I.V. fluids: What nurses need to know. Nursing2017, 41(5), 30-38. ↵
- “Osmotic pressure on blood cells diagram.svg” by LadyofHats is in the Public Domain↵
- Harris, H. (2011). I.V. fluids: What nurses need to know. Nursing2017, 41(5), 30-38. ↵


