Natural barriers against pathogens
The physical and chemical barriers that prevent pathogens from getting into our body tissues in the first place are often overlooked. As Figure 6 shows, the most comprehensive barrier is the waterproof layer of skin that covers the body’s surface. Human skin keeps most pathogens out as long as it remains intact. The speed with which a cut or graze can become infected is a reminder of the protection we normally get from our skin.
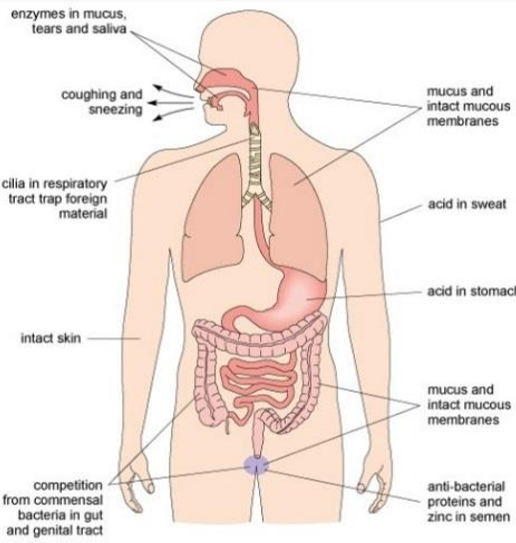
Figure \(\PageIndex{1}\). Physical and chemical barriers against infection
Vector-borne infections (e.g. malaria) are transmitted by biting invertebrates (e.g. mosquitoes) penetrating the skin when taking a blood meal. Humans are also vulnerable to invasion by pathogens in the air, food, water and soil, or during physical contact with infected people, some animals (e.g. pigs, dogs, poultry) or their feces. As Figure \(\PageIndex{1}\) illustrates, the inner surface of the respiratory system (nose, throat, airways and lungs), stomach, intestines, bladder and reproductive tract are lined with membranes that secrete jelly-like mucus, presenting a barrier against pathogens entering our tissues via these routes. Microscopic hairs called cilia [sill-ee-ah] line the respiratory system and ‘beat’ in unison to shunt mucus containing trapped pathogens towards the nose and mouth, where they can be expelled by coughing and sneezing, or swallowed into the stomach where acid destroys them.
Chemical barriers against infection include enzymes in tears, saliva and mucus that break down the surface of bacteria. The acid in sweat and in the stomach kills cellular pathogens and there are anti-bacterial proteins in semen (the fluid that contains male sperm). The more complex mechanisms of the immune system are only needed if pathogens breach these physical and chemical barriers.
The immune response to infection
The human immune system is an extremely complex network of interacting cells and biological molecules. The aim here is simply to give you an overview of how an immune response to infection develops, without going into too much detail. It occurs in three overlapping stages, the first of which is triggered when body cells are damaged. When tissues are injured, the damaged cells release chemicals that trigger the sequence of events described as inflammation. It occurs in response to any type of injury, such as a blow or a cut, an insect bite, or damage caused by pathogens multiplying in body tissues. Inflammation has four characteristic effects at the site of an injury, the first two of which are visible around the splinter shown in Figure \(\PageIndex{2}\):
- swelling
- redness
- heat
- pain
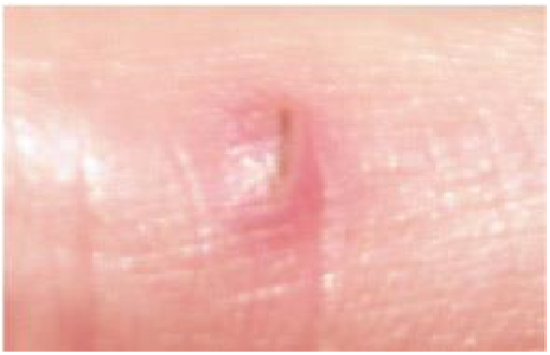
Figure \(\PageIndex{2}\). Inflammation of the tissues around a splinter
The inflamed area shows these signs because the local blood vessels dilate (get wider), increasing blood flow into the injury site, so it looks red as well as feeling warmer than the surrounding tissue. The walls of the blood vessels near the injury become leaky, allowing fluid, defensive proteins and immune system cells (described shortly) to flood into the area, which becomes swollen as a result. One of the proteins released during the inflammatory response also makes the area more sensitive to painful stimuli, so inflamed tissue is sore to touch.
Sites of tissue injury are vulnerable to invasion by pathogens so the benefits of inflammation generally outweigh the discomfort it causes. Flooding the area with fluid dilutes any pathogens that are already present, and the local concentration of immune system cells and defensive proteins enables an immune response to begin more quickly.
Leukocytes: the cells of the immune system
Once the barriers to infection have been breached and inflammation has begun, the active agents of the immune system, the leukocytes [loo-koh-sites], get to work. Leukocytes are often described as ‘white blood cells’ to distinguish them from the red blood cells that transport oxygen around the body; however, calling them ‘blood’ cells is misleading because leukocytes roam throughout the body tissues and only spend part of their lives in the bloodstream. In fact, they spend more time in the lymphatic system (Figure \(\PageIndex{3}\)), the network of fine tubules that collect tissue fluid from all over the body and return it to the bloodstream.
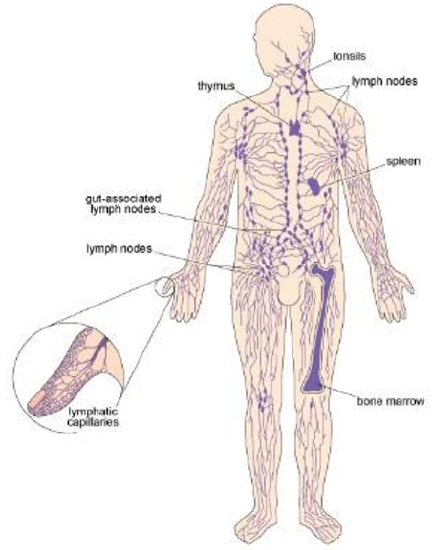
Figure \(\PageIndex{3}\). The human lymphatic system
The lymphatic system includes specialized organs and tissues where leukocytes develop. During an immune response to pathogens, we may become aware of swollen lymph nodes (popularly called ‘glands’) in the neck, armpits or groin, which enlarge when the leukocytes they contain are multiplying near a site of infection.
Leukocytes can distinguish between ‘self’, the cells and proteins generated by the organism whose body they patrol, and ‘non-self’ (or ‘foreign’) material such as pathogens that originated outside the host’s body. Leukocytes are self-tolerant, i.e. they do not normally attack the host’s own cells or body proteins, but direct their actions only against non-self material that may pose a threat.
Although we have referred to ‘the’ immune response, as if it was just one thing, in fact, there are two types of immune response, distinguished as innate and adaptive immunity.
Innate immunity
All animals, even those with much simpler bodies than our own (e.g. parasitic worms) respond to tissue damage in ways that resemble inflammation in humans. They have cells similar to leukocytes and defensive proteins that flood into areas of tissue damage or infection. These leukocytes and proteins can defend the organism from pathogens because they recognize common patterns of molecules that occur in the structures of many different types of pathogens. These pathogen ‘signature’ molecules are known as PAMPs, or pathogen-associated molecular patterns.
The fact that PAMPs are commonly found in unrelated pathogens means that the leukocytes that recognize them cannot tell one type of pathogen from another. This non-specific immune response against pathogens is so widespread among animals that it is described as innate immunity (‘innate’ means ‘inborn’). Some texts use the alternative term ‘natural’ immunity.
The leukocytes involved in innate immunity are of two general types, each with a different action against pathogens:
- Cytotoxic [sigh-toh-tox-ik] leukocytes, which simply means ‘cell poisoning’ (the prefix ‘cyto’ denotes a cell). These leukocytes have various methods of attaching to the outside of a pathogen and releasing destructive chemicals onto its surface. Worm larvae, bacteria and protists can all be killed this way.
- Phagocytic [fag-oh-sit-ik] leukocytes (the prefix ‘phago’ comes from a Greek word meaning ‘to eat’), often abbreviated to phagocytes [fag-oh-sigh-tz]. These leukocytes engulf small pathogens such as bacteria, drawing them into the cytosol where destructive chemicals break them down. This action is termed phagocytosis [fag-ohsigh-toh-siss].
The anti-pathogen activities of certain specialized proteins are important contributors to the innate immune response. They include proteins that accelerate inflammation, target leukocytes onto pathogens or make host cells resistant to invasion by viruses. Their concentration increases rapidly in the bloodstream during an infection and this rise can be detected in blood tests as a diagnostic sign of infection.
In summary, all animals have innate immunity based on cells similar to human leukocytes and defensive proteins that defend the organism against pathogens ‘in general’ in a nonspecific way, i.e. these defenses cannot distinguish between one type of pathogen and another. Humans and other warm-blooded animals have an additional defensive capability called adaptive immunity, which differentiates specifically between pathogens, as the next section describes.
Adaptive Immunity
Adaptive immunity is due to the actions of two types of specialized leukocytes, known as T cells and B cells. (If you are interested, the letters denote ‘thymus’ and ‘bone marrow’, the tissues where each of these leukocytes mature.) We will describe their individual contributions to the adaptive immune response shortly, but first we focus on the most striking difference between innate and adaptive immunity. The clue lies in the word ‘adaptive’.
T cells and B cells have recognition methods that distinguish between different pathogens (e.g. different species of bacteria), and they adapt during their first encounter with a particular pathogen. The second time they meet it in the body, the adaptive response begins earlier, lasts longer and is more effective than it was on the first occasion.
So, there is a much faster and increased response to a subsequent encounter with a pathogen and this demonstrates the adaptability of the immune system. This response is due to the production of long-lived memory cells that circulate in the body after the primary adaptive immune response subsides. These memory cells are specifically programmed to recognize the same pathogens that triggered the primary response if they ever get into the body again.
Antigens and the specific recognition of pathogens
Every type of pathogen has at least one (often many more) unique molecules known as antigens in their structure. In addition to the PAMPs (pathogen-associated molecular patterns) shared by many different pathogens, each type of pathogen also has its own unique distinctive antigens. Each individual T cell and B cell (the leukocytes responsible for adaptive immunity) is programmed to recognize just one specific antigen, so it follows that each T or B cell can usually recognize only one type of pathogen, or at most two or three closely related pathogens that have very similar antigens. Recognition of an antigen by these adaptive leukocytes triggers an immune response against only those pathogens with that antigen in their structure. The political slogan ‘One person, one vote’ springs to mind as an analogy for ‘One adaptive leukocyte, one target’!
Antibodies and B cells
Antibodies are very large proteins that contribute to adaptive immunity. A distinguishing feature of antibodies is that their structure includes at least two binding sites for antigens. B cells produce antibodies and also use them as their antigen receptors. The B cells carry antibodies embedded by the ‘tail’ in their outer cell membrane, with the binding sites facing outwards. This enables the B cell to bind to antigens that fit the binding sites in the antibodies it carries on its surface. This binding event is essential (but not sufficient on its own) to activate B cells into making a lot more antibody molecules that recognize the same antigen. These antibodies are released by the B cells and circulate in the bloodstream, tissue fluids and the lymphatic system. Antibodies are also abundant in the mucus membranes lining the respiratory system, the gut and the reproductive system, i.e. the sites in the body in contact with substances such as air, food, drinking water and sexual fluids that could contain pathogens.
Antibodies are often portrayed in the media as if they were ‘magic bullets’ that attack pathogens, but in fact they are more like ‘waving flags’ with a message that reads ‘here is a pathogen – come and destroy it’. When antibodies bind to a pathogen, they simply label it for destruction by leukocytes with the innate ability to phagocytose (engulf) it, or cytotoxic (cell-killing) leukocytes and defensive proteins. You can think of them as recruiting the cells and defensive proteins of the innate immune system to join the attack. We conclude this tour of adaptive immunity by describing the T cells.
T cells in adaptive immunity
There are two types of T cells with different roles in adaptive immunity. The cytotoxic T cells release destructive chemicals onto their target’s outer surface in much the same way as the cytotoxic leukocytes do in an innate immune response. But there is one crucial difference. Cytotoxic T cells are programmed to kill the body’s own cells that have become infected by viruses or by the few types of bacteria and single-celled pathogens that can ‘hide’ inside the cells of their host (Mycobacterium tuberculosis, the bacteria that cause TB, can do this). Without the cytotoxic T cells, we woud be particularly susceptible to infectious diseases caused by these pathogens.
The other T cell type is called the helper T cells. They send activation signals to all the other leukocytes involved in inflammation, phagocytosis, cytotoxicity or production of antibodies by B cells. Recognition of a pathogen by binding to it is only the first step. The other leukocytes cannot take action against the pathogens they encounter without activation signals from the helper T cells.
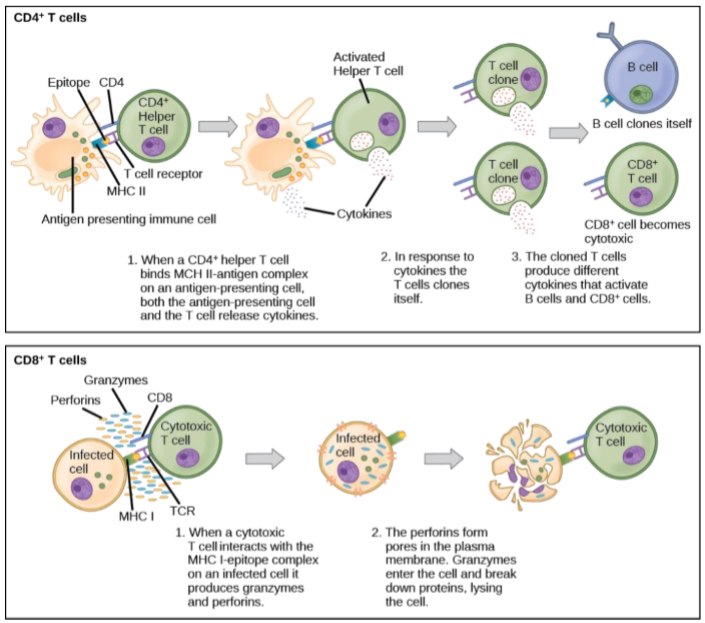
Figure \(\PageIndex{4}\). The Cell-Mediated Immune Response
Vaccination
Vaccination relies on the development of immunological memory for its protective effect. Vaccines contain killed pathogens, or extracts from pathogens, or modified strains of pathogens that are no longer harmful. For example, the MMR vaccine contains weakened (the technical term is ‘attenuated’) variants of the three viruses that cause measles, mumps and rubella (Figure \(\PageIndex{5}\)).
Figure \(\PageIndex{5}\). The MMR vaccine contains live attenuated strains of measles, mumps and rubella viruses.
Note that each of the memory cells and the T cells and B cells generated in an adaptive immune response are specifically directed against just one type of pathogen or a very closely related strain. Vaccination with MMR vaccine is only protective against measles, mumps and rubella infection.
Despite decades of effort, medical science has so far been unable to produce effective vaccines against a number of important infectious diseases, including HIV/AIDS and other sexually transmitted infections (although trials of several candidate vaccines are underway). Vaccines against malaria have shown some potential, but they only protected about half the vaccinated children in large-scale African trials. There are no effective vaccines against infections caused by any other single-celled or multicellular parasites.
Some vaccines are effective only for a relatively short time, e.g. cholera vaccines give protection for only around six months. Others, such as the tetanus vaccine, need repeated booster doses. Annual influenza vaccinations are offered to vulnerable groups in the population because influenza viruses alter their antigens over time.
Opposition to vaccination
Another major challenge is parental opposition to vaccination because of concerns about vaccine safety. Some parents are understandably anxious about taking a healthy child to be given a vaccine derived from infectious agents. Most vaccines are injected, which can be a frightening and painful experience for a child. Of even greater concern is that some children experience a negative reaction to a vaccine, most often inflammation around the injection site and/or a mild fever lasting a day or two, but rare instances of more severe reactions such as convulsions sometimes occur.
Hesitancy about vaccinating a child is increased if parents have no experience of the disease the vaccine prevents, because it has become rare in communities with high vaccine coverage. Some parents conclude that vaccination is unnecessary and might expose their children to an avoidable risk.
Negative rumors about a vaccine are another deterrent. For example, the myth that vaccination is a Western plot to sterilize Muslim children has deterred parents in northern Nigeria, Pakistan and Afghanistan from allowing their children to be vaccinated against polio (Figure \(\PageIndex{6}\)). Terrorist attacks have occurred against polio vaccination teams in all three countries – the only remaining locations where polio had not been eradicated by the WHO target of 2015.

Figure \(\PageIndex{6}\). Negative rumors about the oral polio vaccine did not deter this Nigerian child’s parents from bringing her for vaccination.
An inevitable consequence of falling vaccination rates in a community is an increase in infections, sometimes with fatal outcomes. In 2014, the WHO warned that progress on eliminating measles had stalled and the number of deaths had begun to rise – from 122 000 in 2012 to 145 700 in 2013.
Countries urgently need to prioritize maintaining and improving immunization coverage. Failure to reverse this alarming trend could jeopardize the momentum generated by a decade of achievements in reducing measles mortality.
Even in countries where over 90% of children are fully vaccinated, uptake rates can plummet under pressure from negative rumors. For example, false claims in 1998 that the MMR vaccine caused autism led to a sustained fall in vaccinations in the United States and United Kingdom, followed by a significant rise in measles cases and, to a lesser extent, mumps. The largest outbreak of measles around Wales in 2012–13 caused more than 1200 cases and one death (Public Health Wales, 2013). Once parents saw the reality of measles infection and the misery and discomfort it causes, a rapid increase in uptake of the MMR vaccine followed the outbreak. The memory of infection fades in well-protected communities, until an outbreak reminds everyone of how devastating infectious diseases can be.


