3.10: Surgery for Nasal Dermoids
- Page ID
- 18271
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)OPEN ACCESS ATLAS OF OTOLARYNGOLOGY, HEAD & NECK OPERATIVE SURGERY
SURGERY FOR NASAL DERMOIDS
Hiba Al-Reefy, Claire Hopkins, Nico Jonas
Midline masses are rare congenital anomalies, with an incidence reported to be between 1 in 20,000 to 1 in 40,000 live births. Nasal dermoid cysts are the most common of the congenital midline nasal masses, a group that also includes nasal gliomas and encephalocoeles, and much less frequently haemangiomas, lipomas, lymphangiomas and other very rare lesions. Cases are usually sporadic, but rare familial series have been reported. They are distinguished by the tissues of origin, with dermoids developing from trapped ectoderm and mesoderm; hence they may contain adnexal structures such as skin, hair follicles and sebaceous glands. By contrast, encephaloceles are herniations of the meninges, and may also contain brain tissue, while gliomas contain glial cells.
Clinical presentation
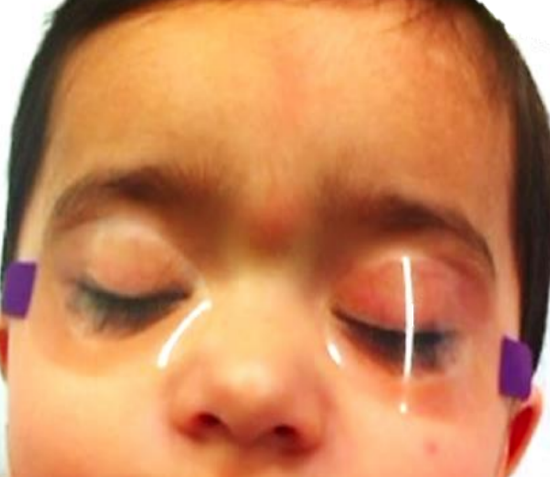
Figure 1: Dermoid cyst presenting as midline nasal mass

Figure 2: Nasofrontal sinus
Many dermoids present at birth, but some present later in childhood or even adulthood when they become symptomtic. They typically present as an isolated mass, a midline nasal deformity (Figure 1) or as a sinus tract opening onto the skin, (Figure 2). A midline nasal pit, fistula, or infected mass may be located anywhere from the glabella to the nasal columella. It usually terminates in a single subcutaneous tract which may have hair at its opening; this is said to be pathognomonic of a dermoid. It may secrete sebaceous material or pus, become intermittently inflamed, form an abscess, cause osteomyelitis, or present with nasal obstruction, or with broadening of the nasal root or bridge.
Occasionally it presents with intracranial complications such as meningitis or a cerebral abscess. Connection with the central nervous system has been variably reported to occur in 4-45%. Suspicion of in intracranial involvement should remain high, and suspected lesions should not be biopsied before radiological evaluation has been undertaken.
Although there is no known syndromic association, associated congenital anomalies occur in 5-41%; this includes aural atresia, mental retardation, spinal column abnormalities, hydrocephalus, hypertelorism, hemifacial microsomia, albinism, corpus callosum agenesis, cerebral atrophy, lumbar lipoma, dermal cyst of the frontal lobe, cleft lip and palate, tracheoesophageal fistula, and coronary artery, cardiac, genital, and cerebral anomalies.
Embryology
The nose is formed from the frontonasal process and two nasal placodes which develop dorsal to the stomodaeum (primitive mouth) during the fourth week of embryological life. The nasal placodes consist of medial and lateral processes and become more prominent. The medial processes approach one another and eventually fuse in the midline. The lateral processes become less prominent as the maxillary process fuses with them. A deep groove in this region, called the nasal-maxillary groove becomes the nasolacrimal duct. As the external nose is develops, other neural crest cells migrate through the frontonasal process to form the posterior septum, ethmoid bone, and sphenoid. The nasal septum develops around week five from the frontonasal process, growing in an anterior-posterior direction.

Figure 3: (a) Fonticulus nasofrontalis and prenasal space; (b) Fonticulus frontalis closes, foramen caecum is formed, and a projection of dural diverticulum contacts the tip of the nose; (c) Dural diverticulum retracts and prenasal space is obliterated. (From: Barkovich AJ, et al. Congenital nasal masses: CT and MR imaging features in 16 cases. Am J Neuroradiol 1991; 12:105-16)
During formation of the skull base and nose, mesenchymal structures are formed from several centers that eventually fuse and begin to ossify. Before they fuse there are recognized spaces between them that are important in the development of congenital midline nasal masses. These include the fonticulus nasofrontalis, the prenasal space, and the foramen caecum (Figures 3a-c). The fonticulus frontalis is the space between the frontal and nasal bones. The prenasal space is located between the nasal bones and the nasal capsule (precursor of nasal septum and nasal cartilages). During fetal development these spaces are close by fusing and ossifying. Abnormal development of these structures is thought to be involved in the formation of nasal dermoids.
A widely accepted theory of dermoid sinus cyst development is the prenasal space theory. According to this theory, during normal development a projection of dura protrudes through the fonticulus frontalis or inferiorly into the prenasal space. This projection normally regresses but if it does not, the dura then remains attached to the epidermis and result in trapped ectodermal elements.
Pre-operative evaluation
Clinical evaluation
Encephalocoeles are pulsatile, compressible masses that expand on crying and on bilateral compression of the internal jugular veins (Furstenberg test); neither gliomas nor dermoids expand with crying or the Furstenberg test. However, a negative Furstenberg test does not exclude intracranial extension; hence imaging is essential.
Radiological Imaging
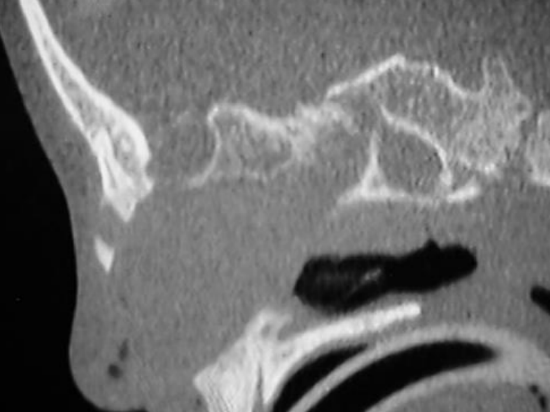
Figure 4: CT scan of nasal dermoid cyst
If a dermoid is suspected, imaging is mandatory to determine the extent of the cyst or tract, to exclude an intracranial connection and to plan surgery. A CT scan delineates the bony anatomy and may indicate an intracranial connection (Figure 4).

Figure 5: MRI scan of nasal dermoid cyst
Features such as an intracranial soft tissue mass, a bifid crista galli, orbital widening or a cribiform plate defect are indicators of intracranial extension. A normal crista galli makes an intracranial connection unlikely. However, in order to exclude this, MRI scanning is required and is becoming the imaging method of choice, as both false-positives and false-negatives for intracranial involvement are found with CT. It is prudent in many cases (especially when a child requires general anesthesia or sedation for imaging) to either obtain both images at a single sitting or to proceed directly to MRI scan (Figure 5).
Surgery
Early surgical intervention is recommended to avoid further distortion of the nose or bony atrophy caused by growth of the mass or recurrent infection. Biopsy is contraindicated due to risk of CSF leakage in cases with intracranial connections. The surgical objective is complete surgical excision at the first operation. Two factors determine the surgical approach
- Is there an intracranial connection?
- What is the extent of the lesion?
The ideal surgical approach for extensive dermoid cysts and tracts should permit
- Access to the whole cyst/tract
- Medial and lateral osteotomies if required
- Rapid repair of cribriform defects and CSF rhinorrhoea
- Reconstruction of the nasal dorsum with a minimally visible scar
Several surgical approaches have been described; occasionally more than one incision is required especially in the presence of a nasal pit or skin breakdown
- Transverse rhinotomy
- Septorhinoplasty approach
- Vertical rhinotomy
- Horizontal nasofrontal incision with eyebrow extensions
- Endoscopic approaches
Transverse rhinotomy: This can be used for small to moderate-sized lesions without intracranial extension. The sinus punctum is excised within a transversely oriented ellipse of skin and the tract is cannulated with a lacrimal probe and dissected. Medial or lateral osteotomies may be performed if necessary. If placed in a natural nasal skin crease it leaves a very favorable scar.

Figure 6: Stepped columella incision with open septorhinoplasty approach

Figure 7: Full exposure of dermoid cyst
Open septorhinoplasty approach: This provides wide exposure, but with a concealed, aesthetically pleasing scar, for larger lesions and for patients with damaged bone and cartilage from prior surgery or recurrent infection or with intracranial extension. A separate excision of a sinus opening may be required; with intracranial extension a combined intracranial approach may be required. A stepped columella incision is made to release the columella followed by bilateral marginal incisions (Figure 6); the upper and lower lateral cartilages are delineated. A dermoid cyst may be adherent to the upper lateral cartilages, dorsal septum and nasal bones; full exposure is mandatory to ensure complete excision and to prevent recurrence (Figure 7). A nasal pit can be removed using a small elliptical skin incision on the nasal dorsum.
An endoscope or operating microscope may be used to facilitate more cephalad dissections and to achieve complete resection. The open septorhinoplasty approach allows for the lower lateral cartilages to be realigned if displaced laterally by a dermoid cyst extending to the nasal tip.
If a large defect remains after surgery, a dorsal graft can be placed to restore volume without the presence of an overlying incision line, thereby reducing risks of infection and extrusion. Grafts are however often not required as the defect fills with scar tissue.

Figure 8: Extended alar base incision

Figure 9a: Further exposure allows complete excision

Figure 9b: Excised dermoid cyst
Extension of this approach to involve the alar base allows for further exposure of the nasal dorsum as the skin envelope can be raised more cephalad (Figures 8, 9a, 9b).
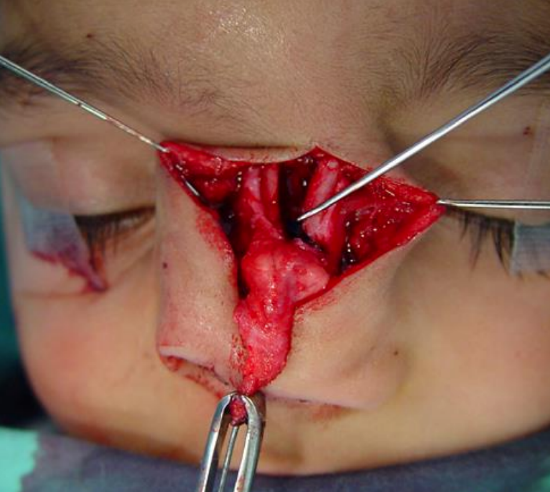
Figure 10: Vertical midline incision
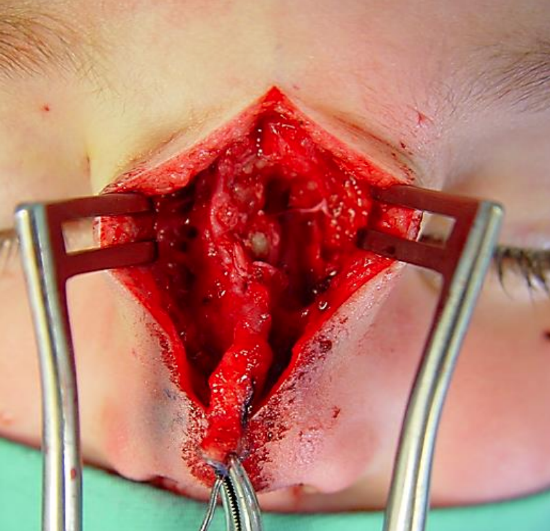
Figure 11: Nasal dermoid tract
Midline vertical incision: This provides excellent exposure as it can be extended to allow access to the skull base (Figures 10, 11).

Figure 12: Horizontal nasofrontal incision with eyebrow extensions

Figure 13a: Dermoid cyst visible in wound

Figure 13b: Skull exposed following excision; specimen (inset)

Figure 14: Bony window marked on outer table of skull

Figure 15: Small cutting burr used to cut out bony window
Horizontal nasofrontal incision: This is reserved for cysts limited to the nasofrontal area or to facilitate a small craniotomy to remove the tract with intracranial extension. The incision can be extended along the eyebrows to allow additional exposure (Figure 12). Following removal of a cyst (Figures 13a, b) a bony window can be marked (Figure 14) to include the tract and allow access to the intracranial component. A small cutting bur can be used to cut out the bony window and allow direct access to the intracranial portion (Figure 15).

Figure 16: Closed horizontal nasofrontal incision
All facial incisions mentioned heal with excellent cosmesis (Figure 16); the choice of external surgical approach is therefore dictated principally by the size and site of the dermoid, presence of a pit, and experience of the surgeon.
Endoscopic approach: An endonasal endoscopic approach is recommended when a dermoid is located within the nasal cavity with little or no cutaneous involvement. It can be combined with a small external midline excision of the cutaneous punctum. Although there are reports of adequate visualization of the skull base through intercartilagenous incisions to allow passage of an endoscope and instruments, intracranial extension is a relative contraindication to endoscopic approaches.
Dermoids with intracanial extension
Midline masses with an intracranial connection usually require a combined approach with the help of a pediatric neurosurgeon. A frontal craniotomy is done via a bicoronal incision with elevation of a pericranial flap to facilitate reconstruction. A subcranial approach via transglabellar or nasofrontal incisions with eyebrow extensions and minicraniotomy as described above may be preferred to avoid frontal lobe retraction. Once the intracranial component has been excised, dural and bony defects are repaired. The extracranial mass may require a separate approach using the incisions described previously to permit complete removal.
Where intracranial communication has been neither excluded nor confirmed, an external approach is initially undertaken. The tract may be followed to its dural attachment at the foramen caecum where biopsies can be taken. If dermal or epidermal elements are identified in the biopsy, frontal craniotomy is indicated; if only fibrous tissue is present then excision is complete.
Postoperative complications
- Tissue defect: Resecting a large dermoid may leave a significant soft tissue defect, as well as splayed nasal bones. Although osteotomies may be used to close the defect, there is often remarkable filling-in of the defect by scar tissue. It is therefore the authors’ practice to splint the nose with plaster of Paris, as following rhinoplasty, and to delay reconstructive procedures. This has the advantage of having no graft material in situ should for revision surgery for recurrence be required.
- Complications associated with intracranial extension
- Meningitis
- Cerebrospinal fluid leak
- Cavernous sinus thrombosis
- Sepsis: Soft tissue, osteomyelitis
- Anosmia: With frontal craniotomy
- Recurrence: This may occur as a delayed phenomenon if the cyst or the tract was not fully excised. Imaging should be repeated, paying particular attention to the most cephalic extension of the cyst. Rates of 4-12% in some larger series, and up to 100%, have been reported
Key Points
- The primary aim is complete surgical excision at the first operation
- Biopsy is contraindicated due to the risk of CSF leakage in cases with intracranial connections
- Early surgical intervention is recommended to avoid further nasal distortion and bony atrophy due to growth of the mass or recurrent infection
- The surgical approach is determined by the extent of the lesion and the presence of an intracranial connection
Authors
Hiba Al-Reefy MBChB, DOHNS, FRCS (ORL-HNS)
Specialist Registrar
Otolaryngology, Head and Neck Surgery
Guys and St. Thomas’ Hospital
London, United Kingdom
drhibaalreefy@hotmail.com
Claire Hopkins BMBCh, MA (Oxon), FRCS (ORL-HNS), DM Rhinologist, Skull Base Surgeon
Guys and St. Thomas’ Hospital
London, United Kingdom
clairehopkins@yahoo.com
Author and Pediatric Section Editor
Nico Jonas MBChB, FCORL, MMed
Pediatric Otolaryngologist
Addenbrooke’s Hospital
Cambridge, United Kingdom
nico.jonas@gmail.com
Editor
Johan Fagan MBChB, FCS (ORL), MMed
Professor and Chairman
Division of Otolaryngology
University of Cape Town
Cape Town, South Africa
johannes.fagan@uct.ac.za


