3.14: Surgery for Congenital Pyriform Aperture Stenosis
- Page ID
- 47879
OPEN ACCESS ATLAS OF OTOLARYNGOLOGY, HEAD & NECK OPERATIVE SURGERY
SURGERY FOR CONGENITAL NASAL PYRIFORM APERTURE STENOSIS (CNPAS)
John Wood, Graeme Van Der Meer

Figure 1: Axial CT demonstrates pyriform aperture stenosis (top) in contrast to bilateral choanal atresia (below)
Congenital nasal pyriform aperture stenosis (CNPAS) is a very rare condition of the newborn, in which the nasal pyriform aperture is narrowed by bony overgrowth of the nasal process of the maxilla (Figure 1). As the narrowest point of the nasal airway, a small change in its diameter may significantly increase nasal airway resistance. As infants are obligate nose breathers for the first three months of life (or longer if premature), such an increased airway resistance causes functional airway obstruction and respiratory distress.
While 1 in 5000 births has some form of congenital nasal airway obstruction, the majority is choanal atresia. The differential diagnosis includes nasal cavity masses, dacryocystocoeles, severe septal deviations and CNPAS.
Embryology
The palate develops from 3 structures: the primary palate that forms the hard palate anterior to the incisive fossa; and 2 lateral plates from outgrowths of the maxillary prominences. CNPAS can be explained by a deficiency of the fused medial nasal prominence, with subsequent faulty development of a small triangular primary palate, often abnormal incisors, and a narrow nasal cavity. During the 8 th week of gestation, maxillary ossification commences from the growth centers above the canine teeth. It has been suggested that over-ossification of the nasal process of the maxilla may be responsible, or it may be due to hypoplastic growth of the primary palate, suggesting that a local dysostosis of the maxilla with bony overgrowth is the abnormality, rather than “failed recanalization” of the nasal cavity.
Associated midline abnormalities can also occur, and may even be considered a minor form of holoprosencephaly (a spectrum of defects or malformations of the brain and face in which the forebrain fails to develop into 2 hemispheres). Abnormalities such as microcephaly, hypoplasia of the corpus callosum or olfactory bulbs can occur. Facial anomalies may include ocular abnormalities, hypotelorism and dental anomalies. Most serious is the occurrence of pituitary dysfunction, which occurs in 15 to 25% of patients. After initial blood testing, clinical monitoring for at least 12 months is recommended, with poor or slowing growth at one year a positive predictor requiring further investigation.
Diagnosis
History and Examination
CNPAS presents like other causes of nasal airway obstruction with cyclical cyanosis, respiratory distress, feeding difficulties and failure to thrive. Failure to pass a nasogastric tube suggests choanal atresia or CNPAS, the difference being the level of obstruction. Obstruction within the 1st 1 cm is typical of CNPAS, while obstruction beyond 3 cm is more likely to signify choanal atresia. Bilateral choanal atresia is typically diagnosed very early, but the diagnosis of CNPAS may be delayed. Some craniofacial syndromes e.g. Crouzon syndrome may present with similar signs and symptoms, but have stenosis affecting the entire length of the nose. They are not amenable to surgical correction and a tracheostomy should be offered instead.
Examination of the nasal cavity with a nasendoscope should generate a high degree of suspicion, especially when unable to pass the scope into the nose. Examination of the oral cavity may reveal an absent frenulum of the upper lip and may indicate the presence of a solitary median maxillary central incisor (SMMCI) syndrome. As a manifestation of holoprosencephaly, it should prompt investigation of other midline abnormalities of the brain, cranial bones and maxilla. Consequently, failure to pass a 5 Fr catheter or 1.9mm endoscope through the anterior third of the nose should prompt further investigation.
Investigations
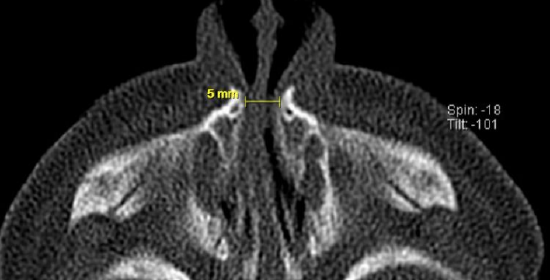
Figure 2: Axial CT scan demonstrating 5mm measurement of pyriform aperture stenosis
When CNPAS is suspected, a craniofacial CT scan with 2 mm slices will confirm the diagnosis. One study reported the average width of the pyriform aperture in CNPAS to be 8.5 mm, compared to 16.9 mm in a control group1. A pyriform aperture of >11 mm in a term infant is considered diagnostic of CNPAS (Figures 1, 2). This measurement however does not determine the need for surgical intervention, as this would depend on the clinical presentation.

Figure 3: Axial CT scan demonstrating solitary median maxillary central incisor

Figure 4: Median maxillary central incisor
Other radiological features should also be reviewed, including the presence of a midline bony ridge on the underside of the hard palate, and particularly the presence of a solitary median maxillary central incisor (Figures 3, 4). The presence of the latter is an indication for MRI scan and chromosomal analysis if available.
Management
Immediate

Figure 5: Oral airway secured with tape

Figure 6: Standard dummy with the tip cut off

Figure 7: Guedel oropharyngeal airway
The baby may present with varying degrees of respiratory distress. The patient should be admitted to a neonatal intensive care unit with continuous oxygen saturation monitoring. Significant distress may require an oral airway, as is used for newborns with bilateral choanal atresia (Figure 5). An oral airway is generally adequate to temporarily secure an airway. Other options include high flow oxygen administered via nasal prongs, a McGovern Nipple, a standard feeding bottle teat or dummy with the tip cut off (Figure 6), or a Guedel oropharyngeal airway (Figure 7).
Feeding can be established via an orogastric tube until nasal patency is achieved.
Neonatologists, geneticists, pediatric ENT and pediatricians should be consulted.
Conservative
Conservative management is the first line of treatment for CNPAS to allow the baby to grow beyond the 3 months’ age of obligate nasal breathing and to potentially avoid surgery. Measures may include nasal irrigation with saline, nasal decongestants, short-term xylometazoline drops, or intranasal dexamethasone drops. Children that respond to conservative treatment should be closely monitored following discharge from hospital for apneic episodes, cyanosis, sleep disordered breathing, respiratory infections and poor growth.
Failed conservative management typically declares itself within the first 3 weeks of treatment. Such children require surgery by the end of the first month.
Indications for surgery
Surgery is indicated if conservative management fails. Indications for surgical intervention include
- apneic episodes
- cyanosis
- poor growth
- failure to thrive
- inability to wean from airway assistance
Predicting failure of conservative management is difficult. One study found a pyriform aperture measurement of <5.7 mm had an 88% sensitivity and specificity for predicting surgical intervention2. It is important to note that studies have suggested that there may often be no correlation with the circumference of the nasal inlet, emphasizing that every baby has a different ability to recover from airway obstruction.
Surgical options
A sublabial approach is most common used as it permits lateral drilling of the nasal process of the maxilla. This technique is described below. Postoperative nasal stenting is the norm, although the optimal duration for stenting is a matter of debate.
Balloon dilatation has also been used3. A 7 mm airway balloon is inflated to a pressure of 10 atmospheres for 5 minutes, with subsequent nasal stenting with a 14 Fr nasopharyngeal airway. This provided a satisfactory nasal airway at 1, 2 and 12 months without further procedures being required. Although only a case report, this technique may provide at worst a temporizing measure until further equipment and expertise is sought as required.
Surgical technique
- A sublabial approach is used
- The procedure is done under general anesthesia with the baby intubated transorally
- Secure the endotracheal tube in a position that allows good access to the oral cavity
- A head ring and shoulder roll can be used
- Drape to protect the eyes, leaving the nose, midface and mouth accessible
- Prepare the nose with neurosurgical pledgets soaked in nasal decongestant

Figure 8: Blanching of alveolar mucosa following infiltration of local anesthetic
- Using a dental syringe, infiltrate local anesthetic (1% lidocaine with 1:100 000 adrenaline) into the upper gingival sulcus and pyriform aperture mucosa (Figure 8)

Figure 9: Sublabial incision and elevation of periosteum over maxilla with Cottle’s elevator
- Make a 1.5 cm sublabial incision with a 15 blade down to bone, taking care not to damage the soft bone overlying the dental buds (Figure 9)

Figure 10: Identification of right pyriform aperture, the anterior nasal spine and the nasal process of the maxilla
- Dissect subperiosteally to expose the anterior nasal spine and the floor of each nostril, remaining anterior to the inferior turbinates to avoid injury to the nasolacrimal system (Figures 9, 10)

Figure 11: 2 mm coarse diamond drill used to remove bone from nasal process of maxilla
- Use a 2 mm coarse diamond drill to widen the pyriform aperture laterally (Figure 11)
- Avoid drilling inferiorly to avoid the tooth buds
- Use the CT scan as a guide and the inferior turbinate as a surgical landmark to obtain an optimal depth of drilling
- Continue drilling until the nasal cavity can accommodate a 3.5 endotracheal tube bilaterally
- Liston dilators can be used once the anterior drilling is complete

Figure 12: Endotracheal tube and Liston dilator (16 Fr) in opposing nostrils
- As in choanal atresia repair, while dilating it is important to keep a Liston dilator in the opposite nasal cavity to prevent the nasal septum from being displaced (Figure 12)
- Replace the nasal mucosa
- Some surgeons recommend making a releasing incision in the mucosa along the floor of the nose
- Create stents by trimming size 3.5 endotracheal tubes
- Insert each stent into the nose
- Ensure the tips of the stents are in the oropharynx just below the soft palate
- To prevent the stents becoming dislodged, a suture is passed between the tips of the tubes as follows:
- Pass a small suction catheter down each tube so that the catheters protrude into the oropharynx
- Grasp the ends of the catheters and advance them out of the mouth
- Tie a single 3.0 silk suture to the end of each catheter
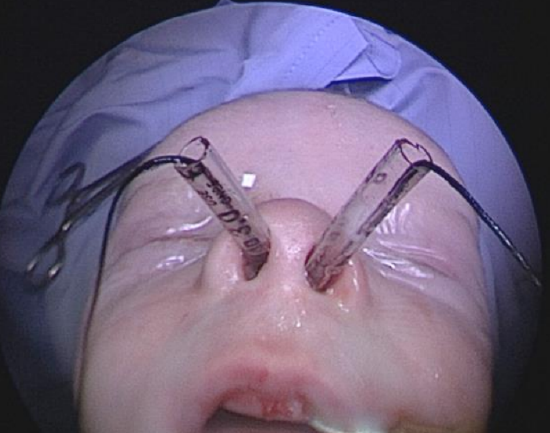
- Figure 13: Stent secured in each nasal cavity by a 3.0 silk suture looped around posterior end of the nasal septum
- Pull the catheters back along the endotracheal tubes and out of the nose, carrying the sutures with them (Figure 13)
- This loops the 3.0 silk suture around posterior end of the nasal septum and secures the stents in each nasal cavity
- It also obviates the need for posterior suturing of the tubes and allows for removal of the tubes in outpatients by simply cutting the anterior knot and pulling out the suture
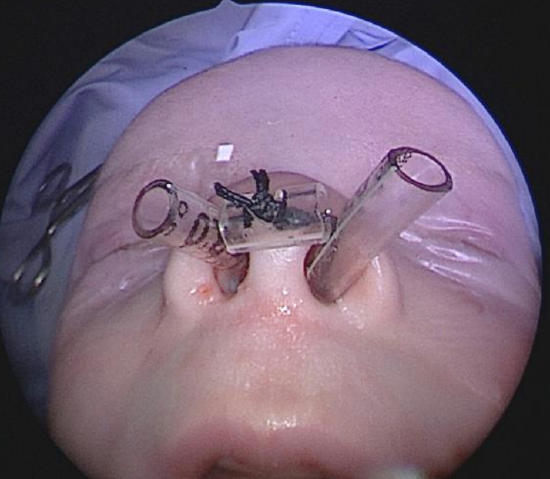
- Figure 14: Middle “bridge” formed from ETT to keep the stents from migrating and the suture from injuring the columella
- Separate the anterior ends of the stents by using a piece of endotracheal tube as a bridge and passing the suture through small holes made in the sides of the stents (Figure 14)
- Trim the stents to facilitate feeding (especially if breastfeeding)
- It is important to measure the length of each stent to allow for accurate suctioning to maintain patency of the stents
- There is no consensus regarding the duration of stenting and varies from a few days to > 4 weeks. If removed too early, tissue dehiscence or restenosis may occur while longer durations are associated with a higher risk of pressure injury to the anterior nose and septum and formation of granulation tissue
Postoperative care
- Monitoring in an intensive care unit for 24-48 hours is recommended to ensure nasal airway patency
- Care of the stents is critical with regular irrigation with saline and suctioning to maintain patency
- Suctioning should be done to the ends of the tubes (measured) to ensure no trauma to the posterior pharyngeal wall occurs
- Use a suction catheter twice the size of the ETT e.g. 7 Fr catheter for a 3.5 ETT
- Once the stents have been removed, use nasal decongestants in conjunction with normal saline drops to minimize edema and crusting. Intranasal corticosteroids such as fluticasone can be used over the ensuing weeks
Complications
- Take care intraoperatively to prevent injury to the nasal mucosa, tooth buds, nasolacrimal ducts and inferior turbinates
- Inspect the nostril daily for signs of pressure areas from the stents
- Feeding difficulties are not uncommon, even with adequate nasal patency
- Restenosis of the pyriform aperture is not uncommon and should be checked for if symptoms recur
Long term follow-up
Long term care should involve a general pediatrician. Consider airway and feeding difficulties, particularly requiring hospital readmission. If signs of restenosis do occur and are symptomatic, then re-expansion may be required e.g. with a balloon or Liston dilator as it is mostly a soft tissue stenosis.
If pituitary deficiency has been noted, then long-term endocrinology follow-up is required.
References
- Belden CJ, Mancuso AA, Schmalfuss I M. CT features of congenital nasal pyriform aperture stenosis: initial experience. Radiology 213, 1999; 495- 501
- Wormald R, Hinton-Bayre A, Bumbak P et al. Congenital nasal pyriform aperture stenosis 5.7mm or less is associated with surgical intervention: A pooled case series. Int J Ped Otorhinolaryngol. 2015;79(11) 1802-5
- Gungor AA, Reiersen DA. Balloon dilatation for congenital nasal piriform aperture stenosis (CNPAS): a novel conservative technique. Am J Otolaryngol. 2014; 35(3) 439-42
Authors
John Wood FRACS, MBBS, MSurg, BSocSci (Hons) BA(Hons)
Pediatric ORL Fellow
Starship Children’s Health
Grafton, Auckland, New Zealand
drjmwood@gmail.com
Graeme Van Der Meer
MBChB MMed (ENT)
Pediatric ORL Consultant
Starship Children’s Health
Grafton, Auckland, New Zealand
graemevdm@gmail.com
Pediatric Section Editor
Nico Jonas MBChB, FCORL, MMed
Pediatric ENT Consultant
Addenbrookes Hospital
Cambridge University Hospital NHS
Foundation Trust
Cambridge, United Kingdom
nicojonas@gmail.com
Editor
Johan Fagan MBChB, FCORL, MMed
Professor and Chairman
Division of Otolaryngology
University of Cape Town
Cape Town, South Africa
johannes.fagan@uct.ac.za


