1.37: Base of Tongue Cancer Resection
- Page ID
- 17671
OPEN ACCESS ATLAS OF OTOLARYNGOLOGY, HEAD & NECK OPERATIVE SURGERY
RESECTION OF CANCERS OF THE BASE OF TONGUE
Johan Fagan
Cancers of the base of tongue (BOT) may be treated with primary surgery, and/or irradiation, and/or chemoradiation therapy. Both the oncology team and patient need to carefully weigh up morbidity vs. cure of surgical and nonsurgical options, both of which may cause significant morbidity. Patients need to be carefully assessed relating to their ability to deal with a measure of aspiration, access to speech and swallowing services and to PEG feeding should they not resume oral feeding.
Surgical approaches
Surgeons have to be au fait with the full range of surgical approaches and reconstructive options so as to ensure complete resection, minimize morbidity, and optimize speech and swallowing function.
Surgical approaches include the following:
- Transoral
- Electrocautery (Bovie)
- CO2 laser
- Transoral robotic (TORS)
- External
- Mandibulotomy with mandibular swing
- Suprahyoid
- Lateral pharyngotomy
This chapter focuses on surgical management of BOT cancer other than CO2 laser and TORS.
Surgical Anatomy
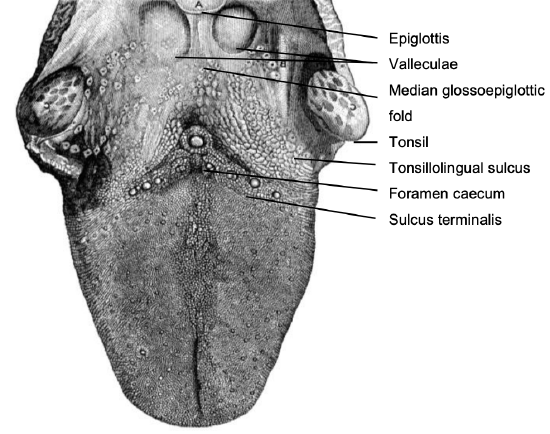
Figure 1: Topography of BOT
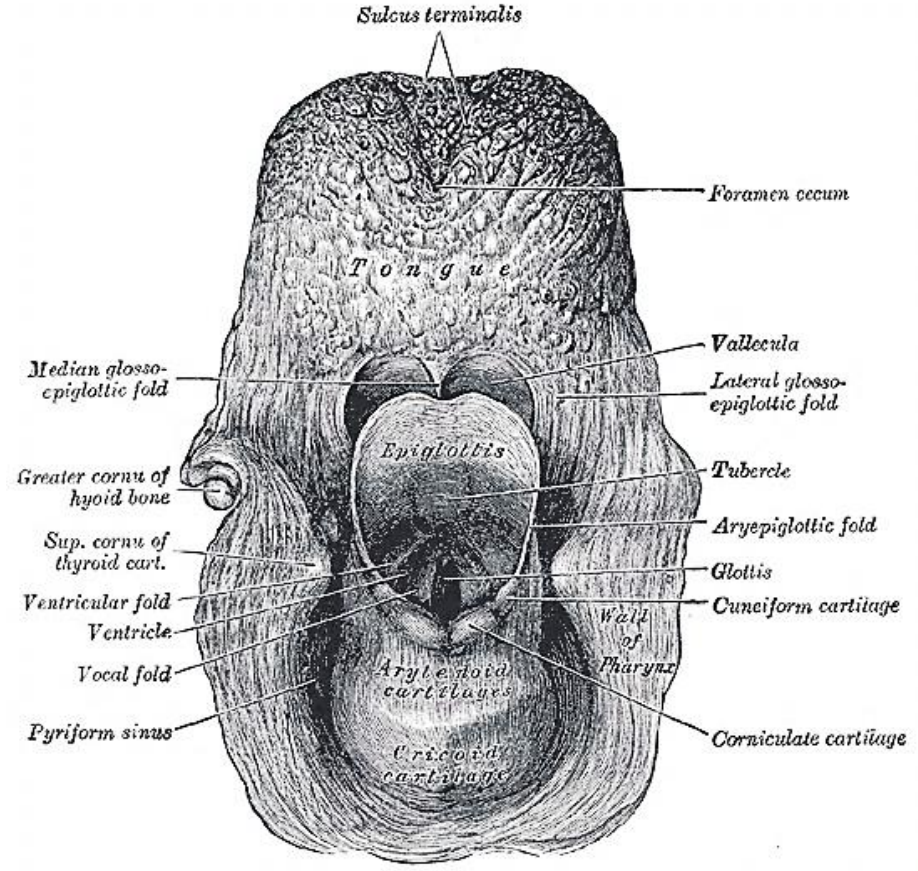
Figure 2: Posterior topographical view of the larynx and BOT
The BOT comprises the posterior 1/3 of the tongue behind the foramen caecum and sulcus terminalis (Figures 1, 2). The mucosa is rough, thick and fixed to the underlying muscle and contains lymphoid follicles (lingual tonsil); this makes it difficult to identify the edges of a BOT tumor; hence frozen section is especially useful to assess resection margins. Posterolaterally the tonsillolingual sulcus separates the tongue from the tonsil fossa. The valleculae separate the base of tongue from the lingual surface of the epiglottis and are divided in the midline by the median glossoepiglottic fold. (Figures 1, 2)
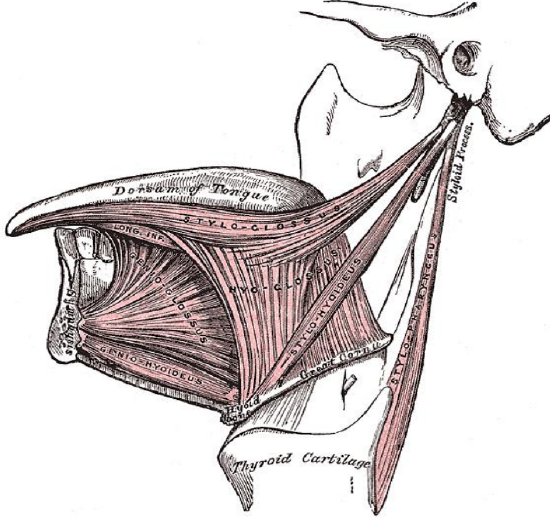
Figure 3: Extrinsic tongue muscles (palatoglossus not shown)
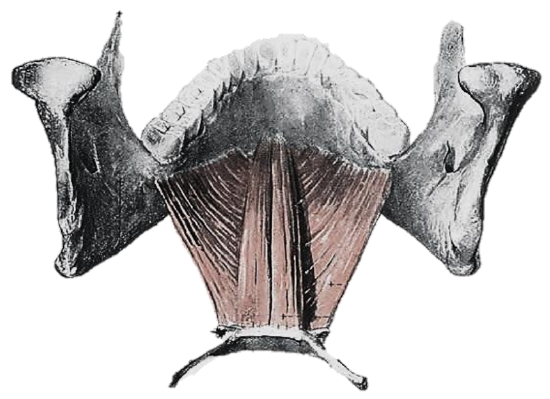
Figure 4: Geniohyoid and mylohyoid muscles
The tongue has eight muscles; four extrinsic muscles (genioglossus, hyoglossus, styloglossus, palatoglossus) control the position of the tongue and are attached to bone (Figure 3); four intrinsic muscles modulate the shape of the tongue and are not attached to bone. Below the tongue are the geniohyoid and mylohoid muscles; the mylohyoid muscle serves as the diaphragm of the mouth and separates the tongue and FOM from the submental and submandibular triangles of the neck (Figures 3, 4).
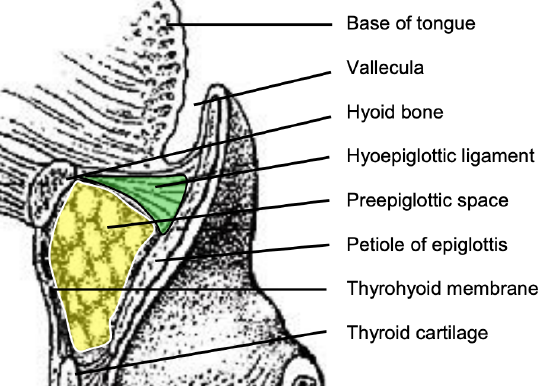
Figure 5: Sagittal view of posterior relations of BOT and preepiglottic space (yellow)
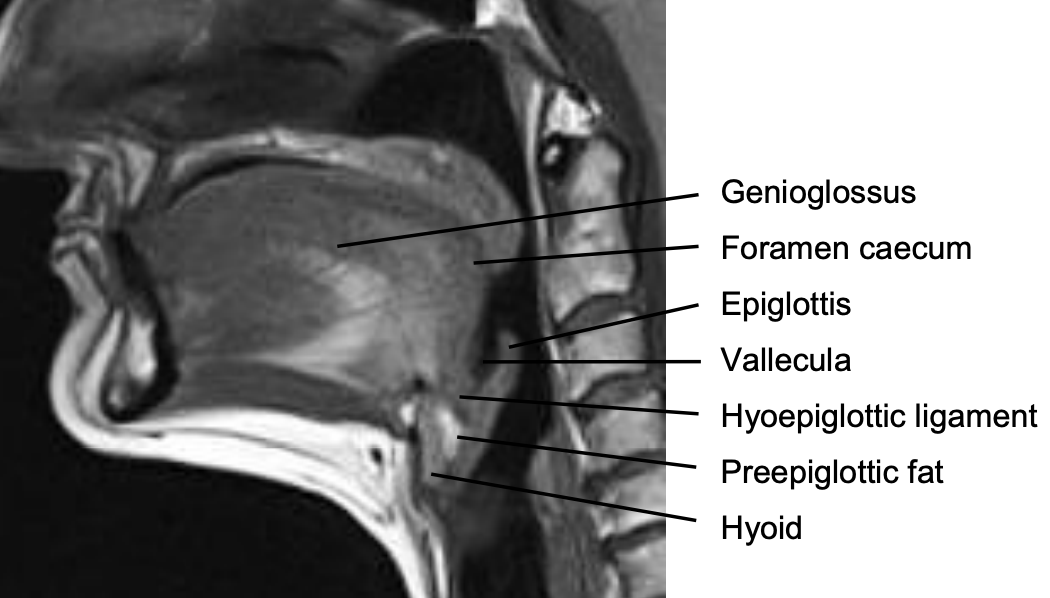
Figure 6: Midline sagittal view of tongue
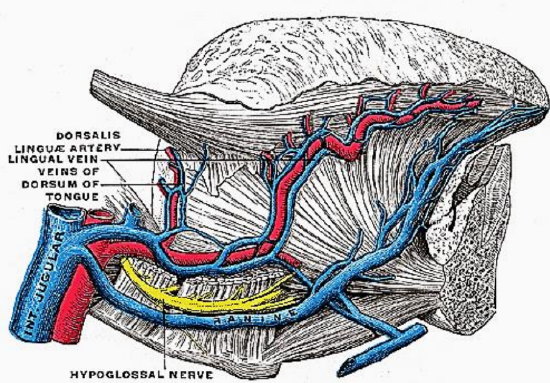
Figure 7: XIIn accompanied by the ranine veins
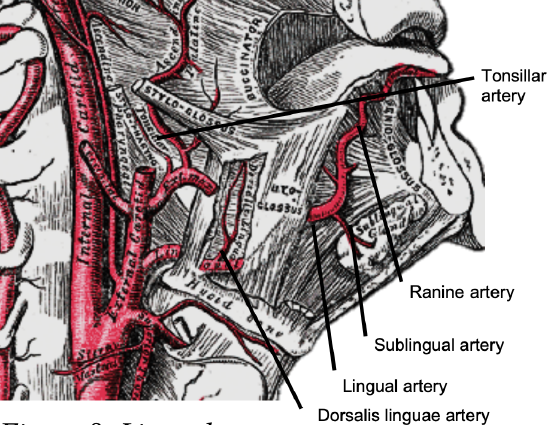
Figure 8: Lingual artery
Posteriorly, the hyoid bone and preepiglottic space are important anatomical structures, an understanding of which is key to assessing whether the larynx can be preserved when using a suprahyoid surgical approach (Figures 5, 6). Most of the tongue muscles attach to the hyoid bone (Figures 3, 4); the lingual artery and hypoglossal nerve (XIIn) pass medial to the greater cornu of the hyoid (Figures 7, 8). The hyoid bone forms the anterior boundary of the preepiglottic space; the superior boundary is the hyoepiglottic ligament (floor of vallecula), and the posterior border is formed by the petiole of the epiglottis (Figures 5, 6).
The arterial supply of the BOT is derived from the paired lingual arteries and its posterior (dorsalis linguae) branches (Figures 7, 8). Additional supply to the BOT emanates from the tonsillar branch of the facial artery and the ascending pharyngeal artery. In practice the lingual arteries are the only vessels that have to be looked for during BOT resection as they are easily injured; it is important to preserve at least one lingual artery to avoid infarction of the tongue. (The author has had one case where sacrifice of a single lingual artery led to necrosis of half the tongue; this is however unusual as there is usually adequate crossflow).
The lingual artery originates from the external carotid artery between the superior thyroid and facial arteries and courses obliquely forwards and medial to the greater cornu of the hyoid (Figures 7, 8). It then loops downward and anteriorly and passes medial to XIIn and the stylohyoid muscle. It then courses directly anteriorly and deep to the hyoglossus and finally ascends submucosally on the undersurface of the tongue as far as its tip as the ranine artery (profunda linguae); it lies to either side of the genioglossus and is accompanied by the lingual nerve. Two or three small dorsalis linguæ arteries arise beneath the hyoglossus and ascend to the posterior part of the dorsum of the tongue and also supply the mucous membrane of the posterior floor of mouth (FOM) and oropharynx (Figures 7, 8).
Venous drainage is via lingual and ranine veins (Figure 7). The lingual veins originate on the dorsum, sides, and undersurface of the tongue and accompany the lingual artery and join the internal jugular vein. The ranine veins originate below the tip of the tongue and are visible on its ventral surface; they accompany the XIIn as venae comitantes and either join the lingual vein or pass lateral to hyoglossus to join the common facial vein.
An understanding of the nerve supply is important to preserve oral function. All intrinsic and extrinsic muscles are innerveted by the XIIn except for palatoglossus which is innervated by Xn. The IXn provides somatic afferent and taste sensation to the posterior 1/3 of the tongue. The lingual nerve provides general somatic sensation to the anterior 2/3 of the mouth and FOM; taste is provided by the chorda tympani branch of VIIn via the lingual nerve.
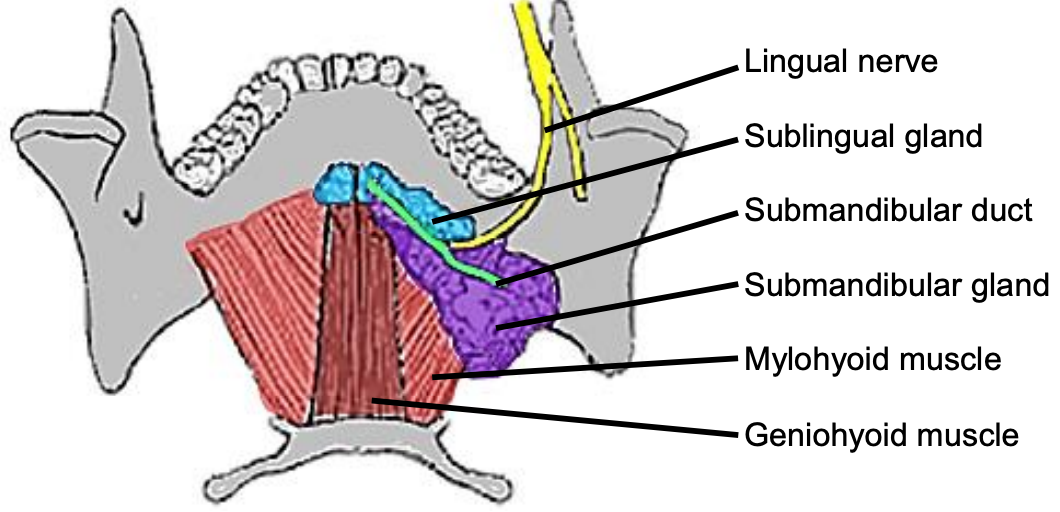
Figure 9: Superior view of FOM, submandibular gland and duct, lingual nerve and mylohyoid and geniohyoid muscles
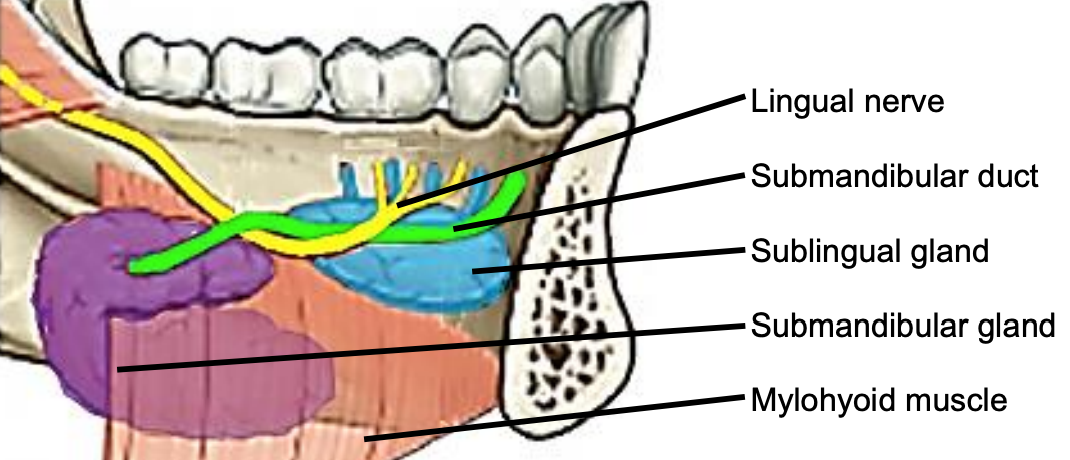
Figure 10: Intraoral view of left sublingual gland with ducts of Rivinus, submandibular gland and duct, lingual nerve and mylohyoid muscles
The lingual nerve is at risk when using a mandibular swing approach to resect tumors of the BOT. The nerve crosses deep to the submandibular duct in the lateral FOM; in the anterior FOM it is located posterior to the duct (Figures 9, 10).
The remainder of this chapter focuses on surgical resection of cancers of the BOT.
Surgical Objectives
- Adequate tumor resection margins; this requires a surgical approach that permits good exposure
- Surgical morbidity should be kept to a minimum relating to swallowing, aspiration, speech, mandibulotomy, lingual nerve function and voice (laryngectomy is required in selected cases to avoid aspiration)
- Control occult cervical metastasis; both sides of the neck are electively treated because of a significant risk of having bilateral occult cervical metastases; the author generally does bilateral elective neck dissection (END) levels I-IV for squamous cell carcinoma of the BOT
Key surgical points
- Oral or transnasal intubation may be difficult or impossible
- Select a surgical approach(es) that ensures adequate exposure; transoral access is often inadequate
- The surface of the BOT is firm and irregular making it difficult to determine margins; employ frozen section to control resection margins
- Preserve tongue function
- Preserve at least one XIIn; sacrificing both nerves is crippling from speech and swallowing perspectives
- Preserve at least one lingual artery to avoid infarction of the tongue
- Preserve sensation with the mandibular swing approach by preserving the lingual nerve
- Avoid/minimize aspiration; this may necessitate total laryngectomy in selected cases
Preoperative evaluation
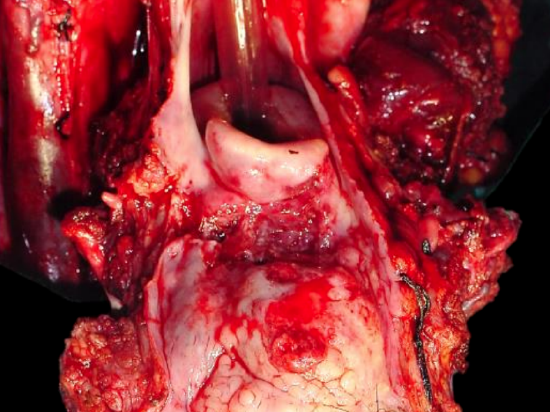
Figure 11: BOT cancer invading vallecula, pre-epiglottic space and epiglottis and requiring total laryngectomy
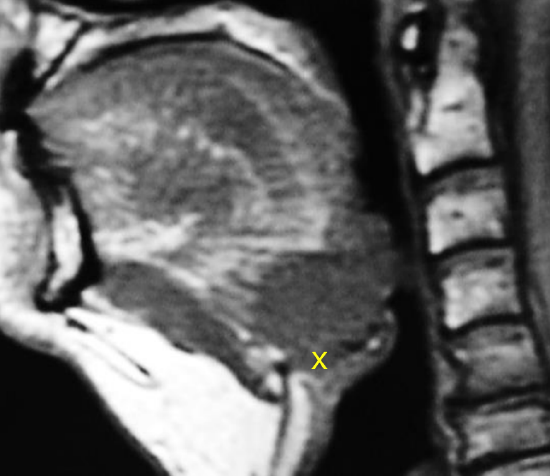
Figure 12: BOT cancer invading and replacing the pre-epiglottic space (x)
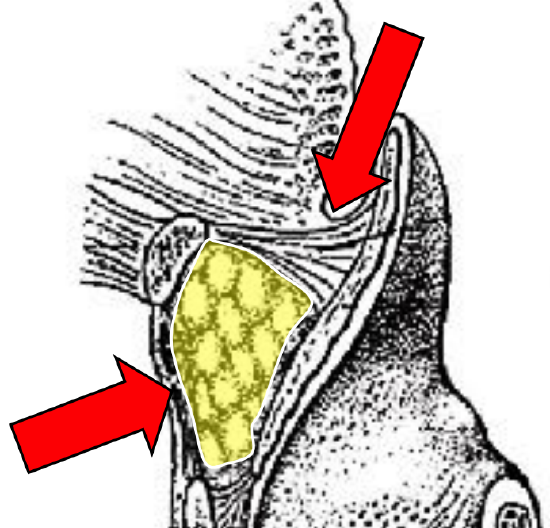
Figure 13: Digital palpation for involvement of pre-epiglottic space
- Are there synchronous primaries, cervical or distant metastases? CXR or CT chest, and panendoscopy
- Is the tumor resectable? BOT cancer may be irresectable if it invades the parapharyngeal space and carotid sheath or extends anteriorly to necessitate total glossectomy in patients not prepared to undergo such surgery. It may be difficult to assess the extent of the primary due to pain, tenderness or trismus. If in doubt, a patient requires imaging such as CT/MRI, or examination under anesthesia.
- Is laryngectomy required? Invasion of the pre-epiglottic space or epiglottis generally indicates a need for laryngectomy to permit swallowing without aspiration (Figure 11). Invasion of the pre-epiglottic space is best assessed with MRI / CT scan in a sagittal plane (Figure 12). Invasion of the pre-epiglottic space can also be assessed intraoperatively by palpating the pre-epiglottic space between a finger placed in the vallecula and a finger placed on the skin of the neck just above/below the hyoid bone; fullness is indicative of tumor in the pre-epiglottic space (Figure 13).
- Is reconstruction required? Restoring BOT bulk with soft tissue flaps may improve speech; however, having an insensate flap may impair swallowing. Therefore, filling the defect with a flap should only be done in selected cases.
- Can the patient tolerate aspiration? Physical fitness, pulmonary reserve, and cognitive function should be considered when selecting patients for major resections and reconstructions as such surgery may be complicated by a variable measure dysphagia and aspiration, especially if followed by chemoradiation.
- Status of dentition? Carious teeth should be removed at the time of the surgery to avoid osteoradionecrosis.
- Is a tracheostomy required? Almost all patients require temporary tracheostomy.
Anesthesia
The surgeon always stands by the patient during induction of anesthesia as it may be difficult or impossible to intubate a patient with a bulky BOT tumor that precludes elevation of the tongue to visualize the larynx. Should the anesthetist be unable to intubate the larynx, the surgeon may be able to intubate with a laryngoscope or do an emergency tracheostomy or cricothyroidotomy; it is prudent to inject the tracheostomy or cricothyroidotomy site with local anesthetic with adrenaline prior to induction. Nasal intubation facilitates resection of BOT tumors and is converted to a tracheostomy during the course of the operation.
Perioperative antibiotics are prescribed for 24 hours.
Surgical access
Good surgical access is essential in order to attain adequate resection margins, to control bleeding, and for reconstruction. A combination of open surgical approaches can be used and will now be discussed. Level 1 of the neck should first be dissected if neck dissection is indicated before proceeding to address the primary tumor.
Transoral access with electrocautery (Bovie) resection
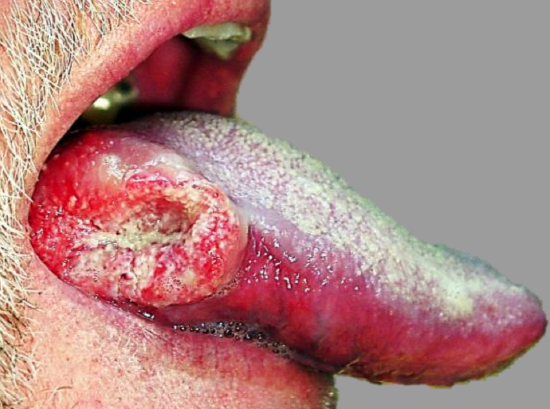
Figure 14: Laterally located BOT cancer suited to transoral resection
The adequacy of transoral access to the BOT varies considerably. A useful way to predict whether transoral resection is likely to be possible is to pull on the anterior tongue with a cotton swab during preoperative clinical evaluation and to then see how accessible the tumor is. Note that tumors become more visible and accessible as the resection proceeds, especially once the thick BOT lining has been incised around the tumor. Laterally placed BOT cancers, especially in edentulous patients, are more amenable to trans-oral access (Figure 14).
Figure 15: Dental bite block is interposed between lateral teeth to keep mouth open
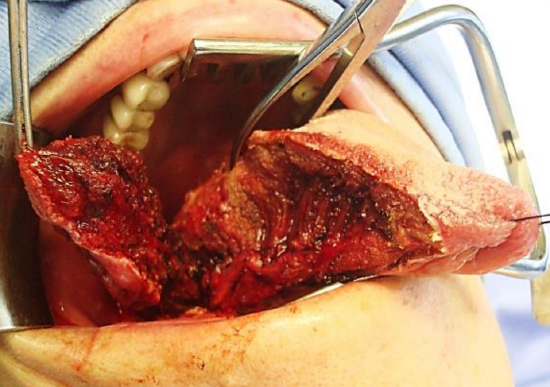
Figure 16: Self-retaining retractor; lateral oral and BOT tumor being resected
The mouth is kept wide open either with a dental bite block (Figure 15) or with a selfretaining retractor, taking care to protect the teeth from injury (Figure 16). Apply traction to tongue and tumor with silk traction sutures or with tissue forceps (Figure 16).
Resect the tumor with at least a 1 cm margin of normal tissue using electrocautery (Bovie). As the resection proceeds posteriorly, insert new silk traction sutures or move the tissue forceps more posteriorly to facilitate delivery of the tumor into the mouth.
Transoral access with median glossotomy
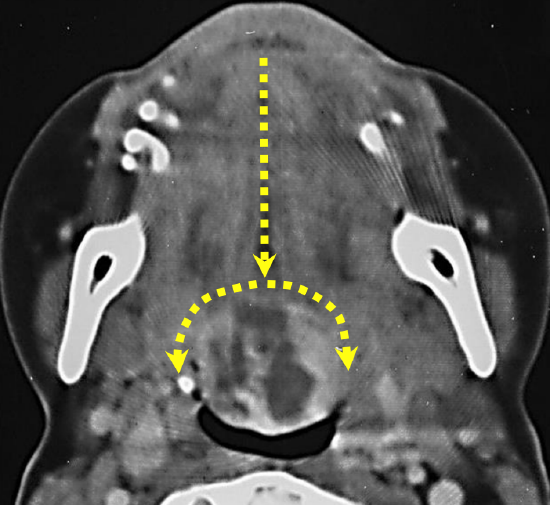
Figure 17: Median glossotomy for lingual thyroid
Midline BOT tumors may be exposed by bisecting the tongue along the median raphe with electrocautery (Figure 17); it is a relatively avascular plane as the vessels and nerves all course laterally, and results in little, if any, functional deficits. The incision can be carried posteriorly up to the hyoid bone if needed.
Mandibulotomy with mandibular swing
This affords excellent access to the BOT. It is especially well suited to cancers that extend anteriorly to involve the oral tongue (Figure 14), or onto the soft palate. It does however leave a facial scar; it may cause deformity of the lower lip; there is a risk of complications relating to the mandibulotomy and dental malocclusion; and the lingual nerve is at risk of injury.
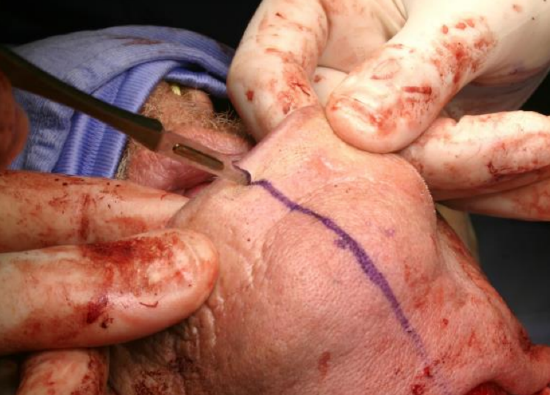
Figure 18: Scoring vermillion border for accurate closure
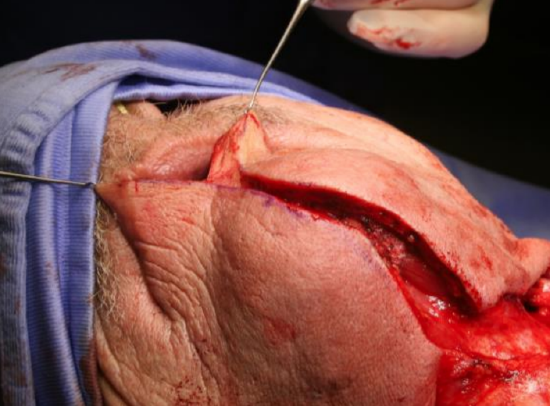
Figure 19: Lip-split incision down to bone
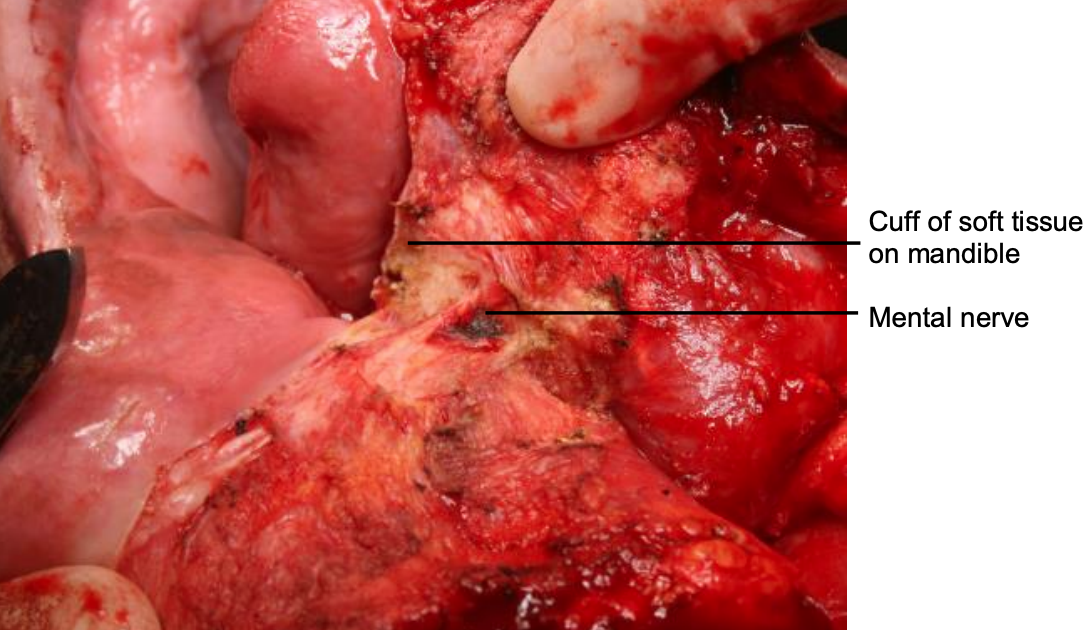
Figure 20: Note preserved mental nerve and cuff of soft tissue
The vermillion border is scored or marked to ensure an accurate repair (Figure 18). The lower lip is divided vertically in the midline (Figure 19). Bleeding from the labial artery is controlled with cautery. Incise the gingivolabial and gingivobuccal mucosae >0.5 cm from the bone leaving a cuff of soft tissue on the bone to facilitate subsequent soft tissue closure (Figure 20). Strip soft tissue off the mandible with monopolar cautery or with a periosteal elevator up to the mental foramen, taking care not to injure the mental nerve where it exits the foramen (Figure 20).

Figure 21: Gigli saw
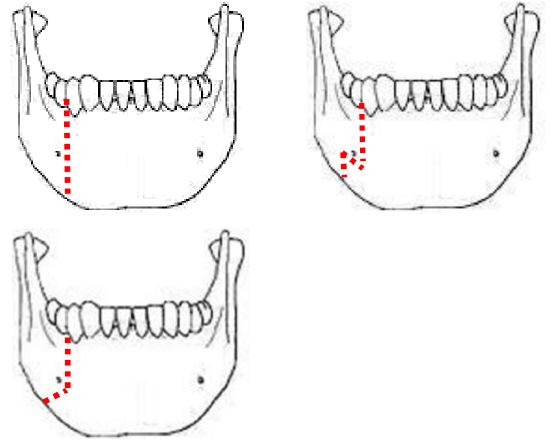
Figure 22: Examples of osteotomies
The mandible is divided just anterior to the mental foramen with a Gigli or a powered saw (Figure 21). The osteotomy may be made vertically or alternatively in a stepor V-shaped fashion to achieve a more stable repair (Figure 22). It is advisable to extract a tooth and make the osteotomy through the dental socket so as avoid devitalizing adjacent teeth. In dentate patients the mandible is preplated with titanium miniplates contoured to the mandible so as to ensure perfect dental alignment. Two 4- holed, non-compression, 2 mm mandibular plates, one placed along the inferior border of the mandible, and the other placed more superiorly are used. Once the plates have been contoured and the holes drilled, they are removed and the bone cut is made.
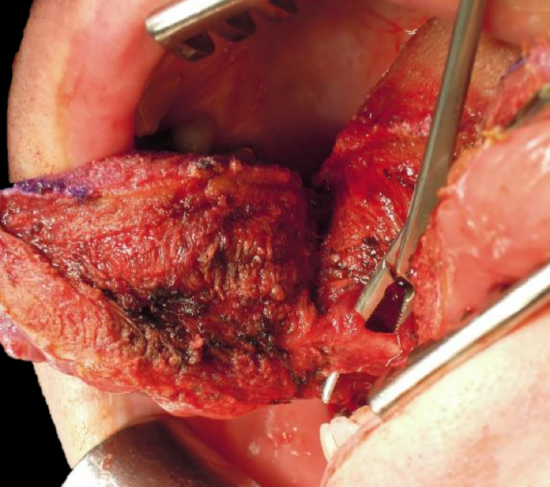
Figure 23: Lingual artery
The surgeon then distracts the cut ends of the mandible to gain access to the oral cavity and proceeds to divide the floor of mouth (FOM) mucosa and mylohyoid muscle about 1 cm from, and parallel to, the mandible, so as to leave soft tissue on the mandible for the subsequent FOM repair at conclusion of surgery. Continue the incision posteriorly along the FOM until the tumor comes into view; the lingual artery (medial to the hyoglossus muscle) and the XIIn course medial to the FOM incision are not at risk of injury at this stage of the dissection. The tumor is resected using electrocautery. The lingual artery may have to be ligated (Figure 23).
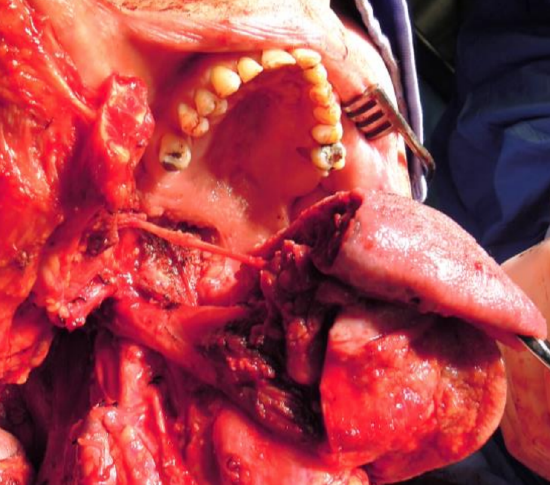
Figure 24: Note wide exposure afforded by mandibulotomy approach to the base of tongue and oropharynx, and the preserved lingual nerve crossing the surgical field
Posteriorly the lingual nerve extends from the skull base and crosses the line of the incision from lateral to medial to course along the lateral FOM; preserve the nerve if possible (Figure 24).
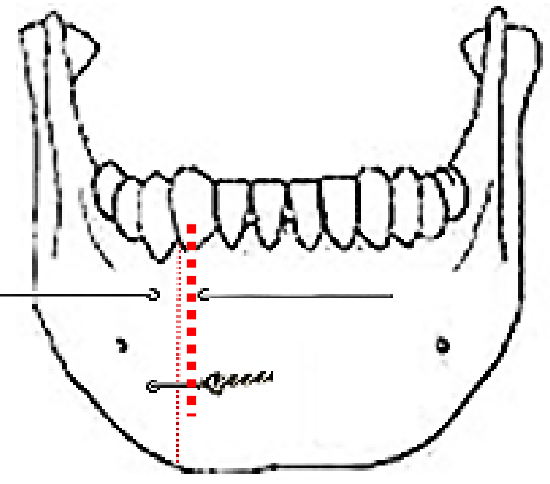
Figure 25: Mandible wired together with stainless steel wire
At conclusion of surgery the FOM is closed with a running vicryl suture, and the osteotomy is plated; when plating sets are not available, opposing holes are drilled on either side of the osteotomy and the mandible is wired together with stainless steel wiring (Figure 25). The lip is carefully repaired in layers to approximate the muscles as well as mucosa and skin.
Suprahyoid approach
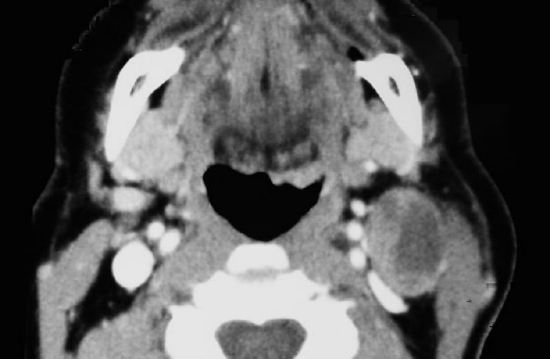
Figure 26: BOT cancer with cystic metastasis suited to suprahyoid approach
This is one of the external approaches favored by the author. It is similar to the suprahyoid dissection step during total laryngectomy. It is well suited to most BOT cancers (Figure 26), although access is limited with cancers that extend far anteriorly into the tongue.
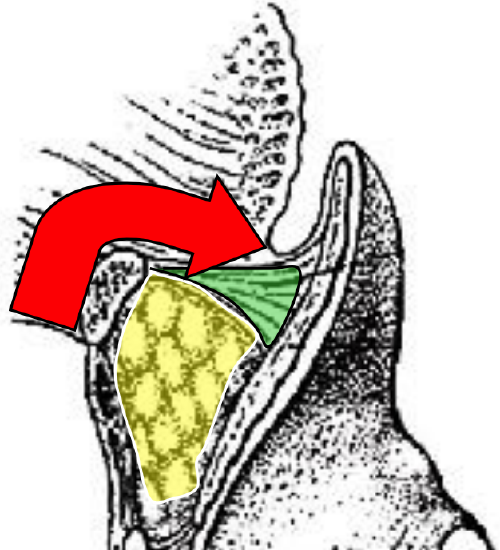
Figure 27: Red arrow indicates suprahyoid access to BOT (hyoepiglottic ligament colored green)
Because the pharynx is entered through the vallecula, it is not suited to cancers that involve the apex of the vallecula, preepiglottic space or epiglottis (Figure 11). Following completion of Levels 1a and b of the neck dissection(s), the superior aspect of the body of the hyoid bone is skeletonized with electrocautery, keeping the dissection between the lesser cornua of the hyoid bone. The dissection is carried posteriorly above the hyoepiglottic ligament, which forms the roof of the preepiglottic fat (Figure 27). The vallecula is entered at its apex (Figure 27).
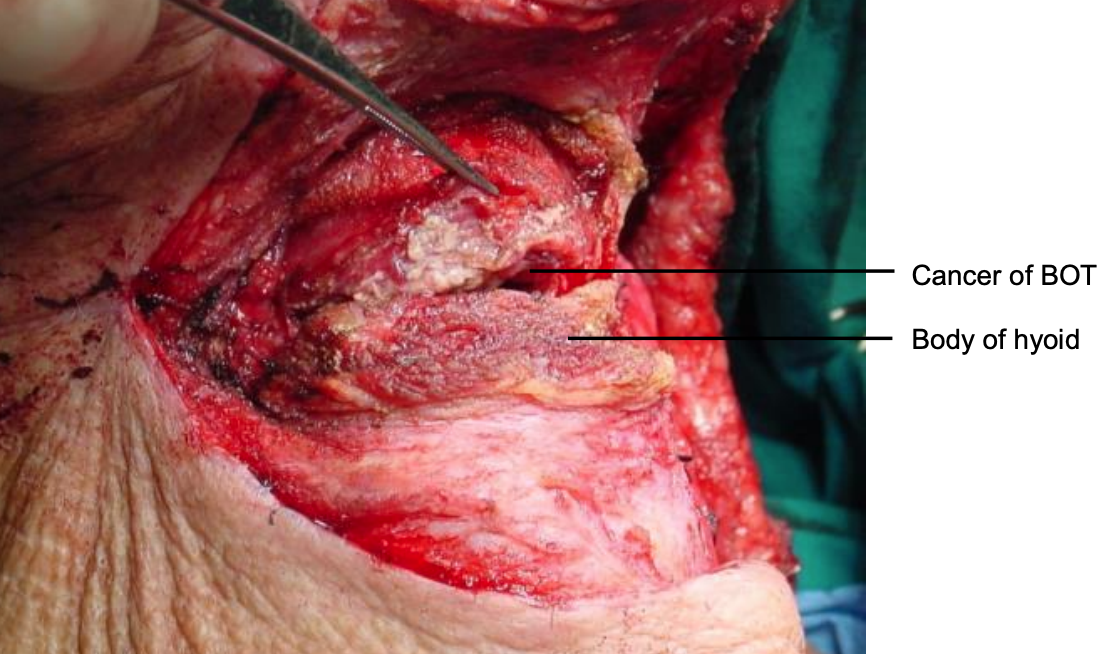
Figure 28: Initial entry into vallecula
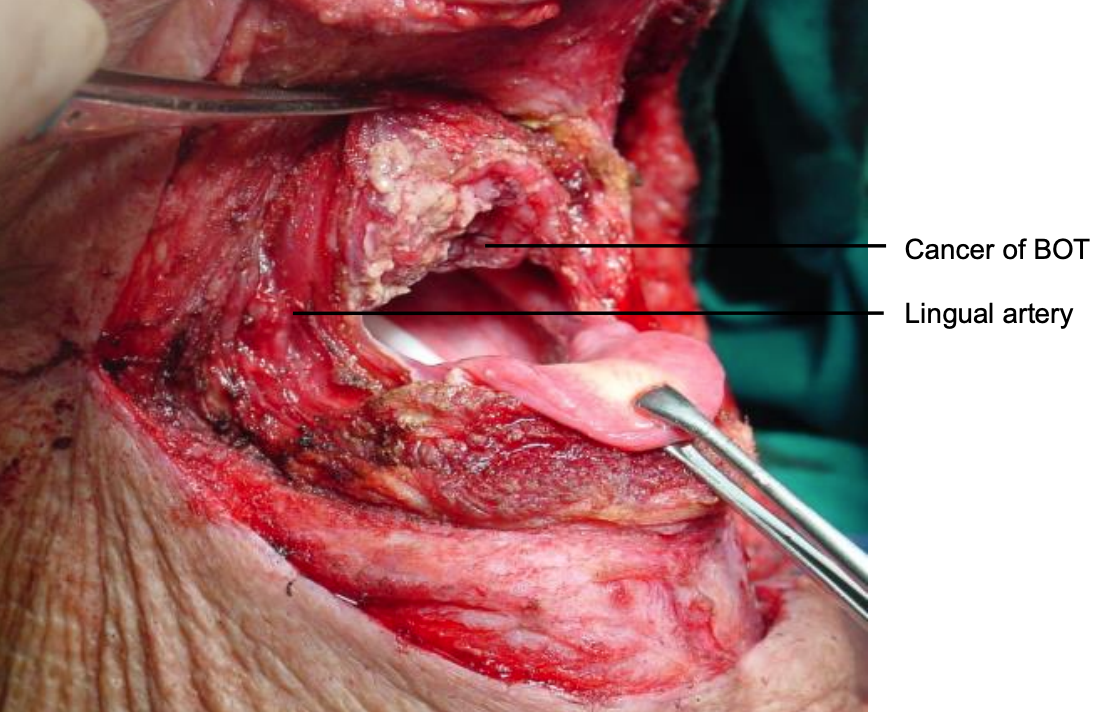
Figure 29: Note wide access to epiglottis, BOT and posterior wall of pharynx
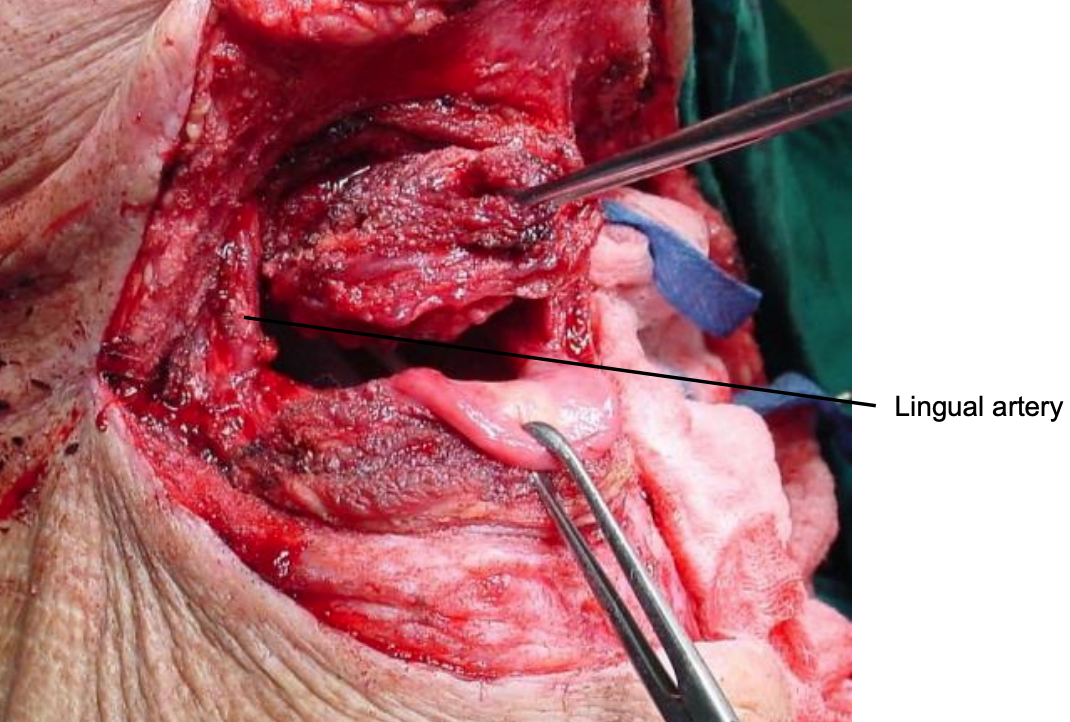
Figure 30: BOT cancer resection
The tumor now comes into view. The cancer is resected with electrocautery (Figures 28-30). Take care to identify and preserve the lingual arteries and, if dissecting even more laterally, the XIIn (Figures 29, 30). Tumor exposure improves as the thick BOT lining is incised to either side of the tumor making the tumor more mobile (Figure 30).
Orientate the specimen for the pathologist with a suture before removing the specimen so as not to lose orientation. The adequacy of the resection margins is checked with frozen section if available.
Primary closure can almost always be done by approximating the BOT resection margin to the vallecula with a running vicryl suture. The supra- and infrahyoid muscles are then sutured together and the neck is closed in the usual fashion.
A temporary tracheostomy is done, and a nasogastric feeding tube is inserted. Once the airway appears adequate the tracheostomy tube is corked for 24 hours before being removed.
Lateral pharyngotomy combined with suprahyoid approach
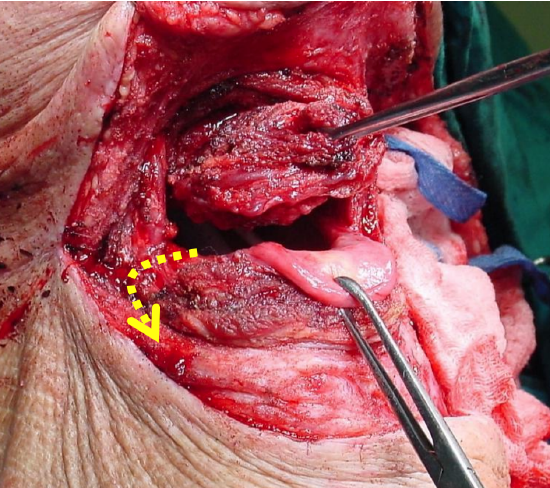
Figure 31: Additional access by lateral pharyngotomy extension (Yellow arrow)
Additional exposure can be obtained by extending the incision along the greater cornu of the hyoid bone and along the posterior margin of the thyroid cartilage; take special care to identify and not to injure the XIIn, the lingual artery or the superior laryngeal nerve (Figure 31).
Pull-through approach
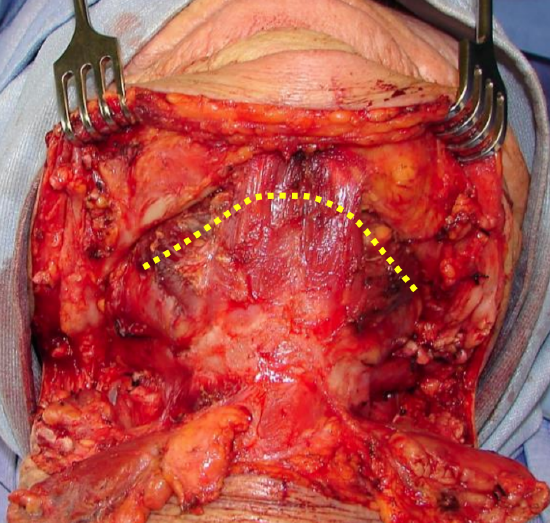
Figure 32: Incision for pull-through approach (Yellow line)
This may be employed when tumor stops some distance from the inner aspect of the mandible. The mucosa of the lateral and anterior FOM is divided >0.5 cm from the inner aspect of the mandible (to facilitate later repair), taking care not to injure the lingual and XIIns, or the submandibular ducts. Following bilateral neck dissections of Levels 1a and 1b, the mandibular attachments of the anterior bellies of digastric, mylohyoid, geniohyoids and genioglossus are divided with electrocautery working from inferiorly (Figure 32). This permits the surgeon to drop the FOM and tongue into the neck and to proceed with the resection. At conclusion of surgery the FOM mucosa and muscle are reapproximated with vicryl sutures.
Repair / Reconstruction
Obtain meticulous haemostasis using ties, monopolar and bipolar cautery before closing the defect. Primary closure of BOT defects gives the best swallowing function although inadequate bulk impairs speech if the BOT cannot approximate the soft palate. Simply shaping a flap to match the resected tissue may well restore form but may have a poor functional result. Therefore one must carefully assess the defect to determine how best to optimize swallowing and speech.
Radial free forearm flap: This is a thin and pliable flap and preserves mobility of the tongue but has limited bulk.
Anterolateral free thigh flap: Muscle harvested with the flap can be tailored to conform to the volume of the defect to be filled. However, it is less pliable than the radial free forearm flap and is only suitable for oral reconstruction in patients with thin thighs.
Pectoralis major flap: This is a good option to use
Buccinator myomucosal flap: A posteriorly based buccinator flap can be used to close a small orocervical communication following resection of a lateral BOT cancer.
Submental artery island flap: The bulk of the flap can be increased by including mylohyoid and digastric if needed.
Supraclavicular flap: This is a thin and pliable flap and preserves mobility of the tongue but has limited bulk.
Final comments
Resecting tumors of the BOT is challenging particularly in terms of access and maintaining swallowing function. One should not compromise resection margins for function. The surgical team must master an array of surgical approaches and reconstructive techniques to ensure the best oncological and functional outcomes.
Author & Editor
Johan Fagan
MBChB, FCS (ORL), MMed
Professor and Chairman
Division of Otolaryngology
University of Cape Town
Cape Town, South Africa
johannes.fagan@uct.ac.za


