3.6: Nucleic Acids
- Page ID
- 11111
Learning objectiveS
- Describe the general chemical structure of nucleic acids, identify monomers and polymers, and list the functions of RNA and DNA
- Describe the structure and function of ATP in the cell
Nucleic acids contain the same elements as proteins: carbon, hydrogen, oxygen, nitrogen; plus phosphorous (C, H, O, N, and P). Nucleic acids are very large macromolecules composed of repetitive units of the same building blocks, nucleotides, similar to a pearl necklace made of many pearls. We can also define nucleic acids as polymers assembled from many smaller covalently bonded monomers.
Nucleic acids are the molecules that function in encoding, transmitting and expressing genetic information in our cells.
All nucleotides are made of three subunits: one or more phosphate groups, a pentose sugar (five-carbon sugar, either deoxyribose or ribose), and a nitrogen-containing base (either adenine, cytosine, guanine, thymine, or uracil). See figure \(\PageIndex{1}\) below.
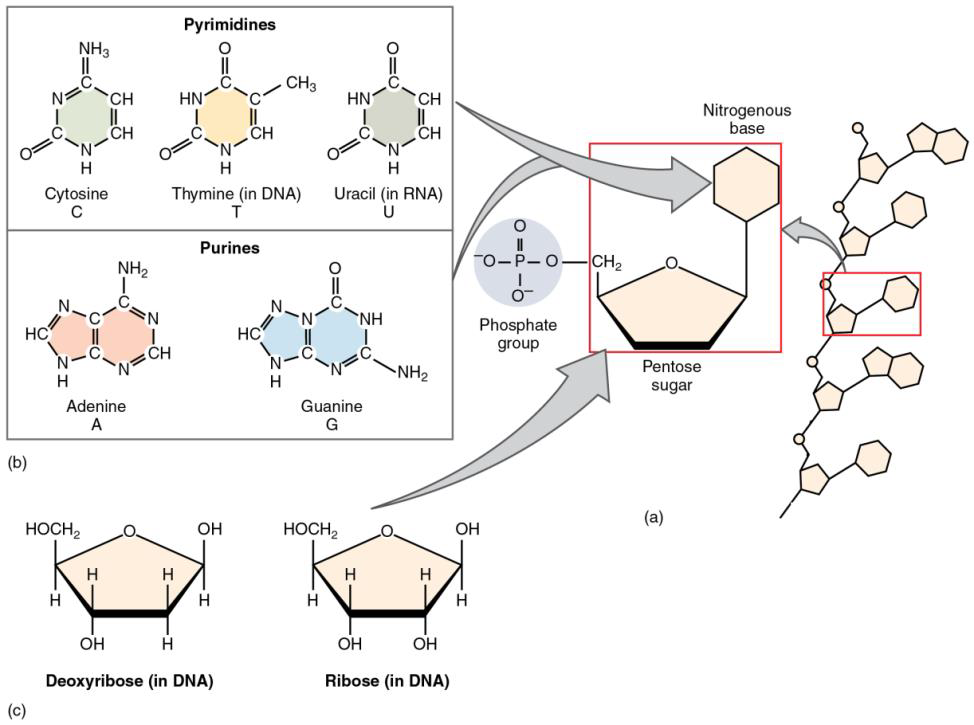
Figure \(\PageIndex{1}\) A nucleic acid short fragment made of five nucleotides is shown on the right; one nucleotide is enclosed in a red rectangle. Each nucleotide is made of one of the five nitrogenous bases, a pentose sugar (ribose or deoxyribose) and a phosphate group. Ribonucleic acid (RNA) has ribose for a pentose, whereas deoxyribonucleic acid (DNA) has deoxyribose. The five nitrogenous bases are classified as pyrimidines (cytosine, thymine, and uracil), which have a ring structure; and purines (adenine and guanine), which have a double-ring structure. RNA molecules may have up to few-thousand nucleotides and are singlestranded, whereas DNA molecules have billions of nucleotides organized in two strings of nucleotides forming a helix. DNA, RNA, and proteins are related to each other as shown in table \(\PageIndex{1}\) below.
| DNA \(\rightarrow\) | is used to synthesize | RNA \(\rightarrow\) | which is used to synthesize | Proteins |
| Polymers of nucleotides | Polymer of nucleotides | Polymer of amino acids | ||
| Encodes amino acid sequence of proteins | Transmits and expresses information in DNA | Perform most cellular functions |
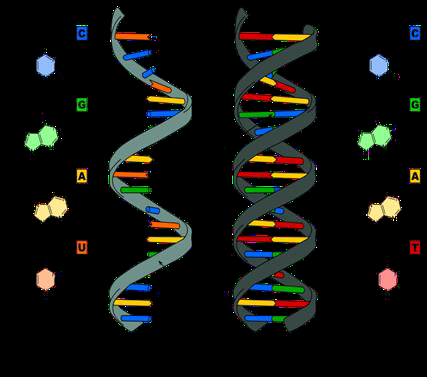
Figure \(\PageIndex{2}\) DNA and RNA share three nucleotides in their composition (cytosine, guanine, and adenine), and they differ in uracil (found only in RNA) and thymine (found only in DNA). RNA is single strand, whereas DNA in double strand
| Type of Nucleic Acid | Function |
|---|---|
| DNA | Encodes and transmits inherited genetic information from one generation to the next |
| RNA | Translates the information encoded in DNA for the production of proteins and help in their synthesis |
Concepts, terms, and facts check
Study Questions Write your answer in a sentence form (do not answer using loose words)
1. What is a nucleic acid?
2. What elements are nucleic acids made of?
3. What are the monomers that make the building blocks of nucleic acids?
4. What are the three components of a nucleotide?
5. List the types of nucleic acids described in the module
6. What are the functions of nucleic acid listed in the module?
Nucleotides are the monomers that make up the nucleic acid polymers. Adenosine triphosphate (ATP) is a nucleotide that has an important function by itself. ATP is a direct and rapid energy source for most cellular activities. ATP consists of a single adenosine (the nitrogen-containing base adenine and the sugar ribose), linked to three phosphate ions.
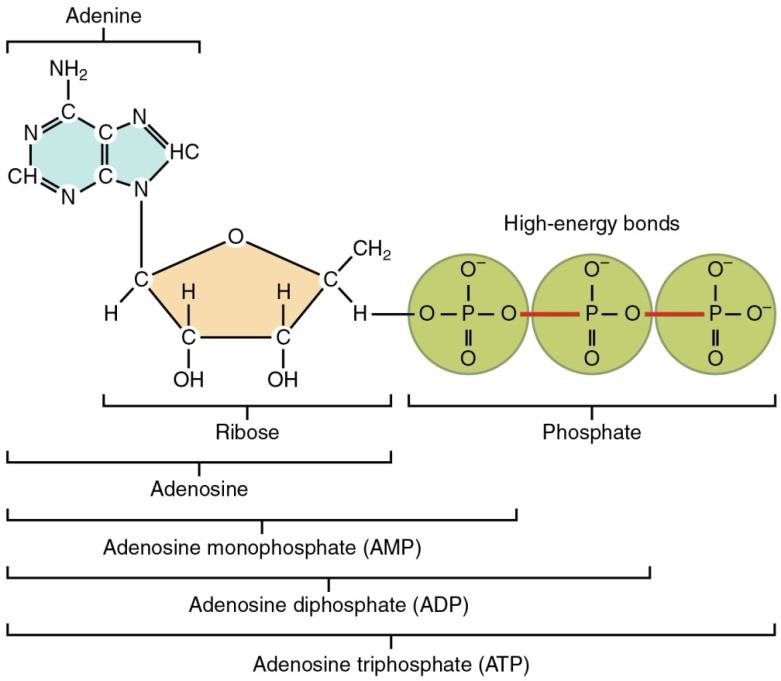
Figure \(\PageIndex{3}\) The two covalent bonds on the right of the molecule (shown in red) are high energy bonds. When an enzymatic reaction breaks them down, a large amount of energy is released. This energy is ready to be used by a cell. On the other hand, when molecules (like the ones we incorporate in our diet) are broken down by enzymes they release energy. This energy can be temporarily held on ATP molecules in the covalent bonds formed between free phosphate groups and adenosine diphosphate (ADP)
ATP is regularly referred to as the primary energy currency for the cell. ATP serves as an intermediary molecule between chemical reactions that release energy, and chemical reactions that require energy. It does so by temporarily “holding” the energy released by an enzymatic reaction in the covalent bonds that attach phosphates to ADP (the red ones in the figure above). Then, the molecule of ATP can give up that energy where it is needed.
The chemical formula summarizing this process, is
\[ATP \leftrightarrow ADP + P_{i} \textsf{(inorganic phosphate)}\]
Since the reaction can go in either direction (from ADP to ATP, or from ATP to ADP), this is an example of a reversible reaction, and it is represented with an double arrow pointing in both directions.
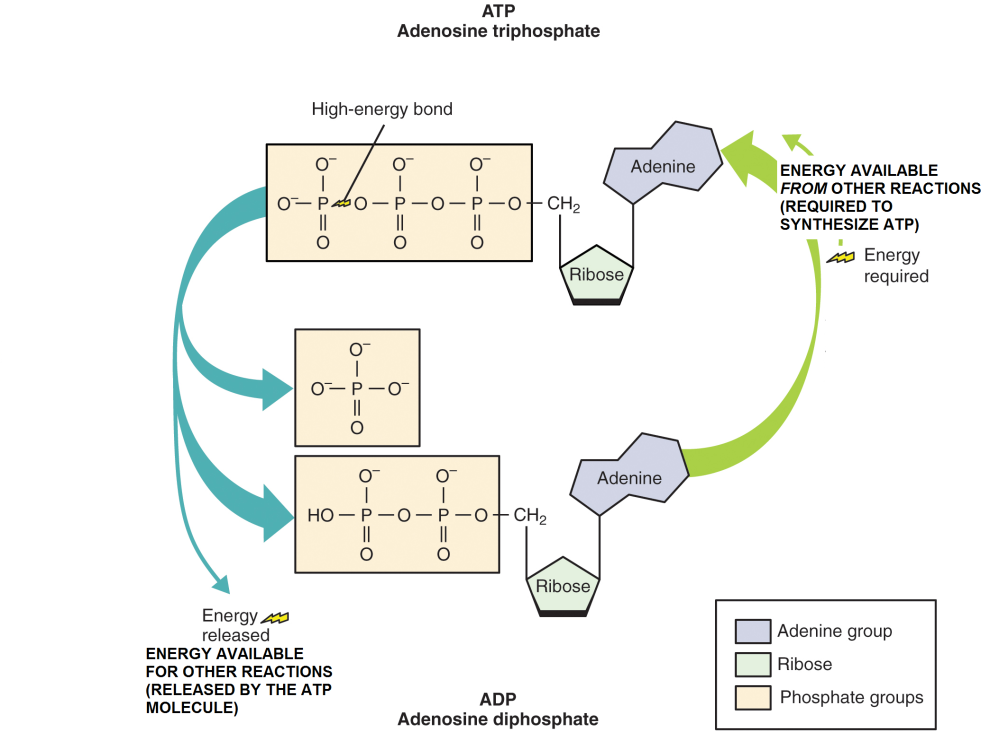
Figure \(\PageIndex{4}\) Adenosine triphosphate (ATP) is the energy molecule in a cell. Energy released by decomposition reactions can be used to make a high energy covalent bond in ATP as shown in the figure. Then, ATP can give up this energy to be used for synthesis reactions.
Concepts, terms, and facts check
Study Questions Write your answer in a sentence form (do not answer using loose words)
1. What type of organic molecule is ATP?
2. What is the function of ATP?

