3.5: Proteins
- Page ID
- 11110
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Learning objectiveS
- Describe the general chemical structure of proteins and identify monomers and polymers; and list and explain functions proteins perform
- Define conjugated protein, lipoprotein, glycosylated molecule, glycoprotein, and glycolipid
- Define enzyme, enzymatic reaction, substrate, product, and active site
Proteins contain the same elements as carbohydrates and lipids: carbon, hydrogen and oxygen; plus nitrogen (C, H, O, and N). Proteins are usually much larger than polysaccharides and triglycerides. Proteins are macromolecules composed of repetitive units of the same building block, amino acids, similar to a pearl necklace that is composed of many pearls. We can also define proteins as polymers assembled from many smaller covalently bonded monomers. The type of covalent bond linking neighboring amino acids is called peptide bond, as shown below in figure \(\PageIndex{3}\).
Proteins carry out most of the jobs in a cell and serve the most diverse range of functions of all the organic macromolecules in our cells and body. Different proteins form parts of cell and tissue structures; regulate physiological processes; contract to move cellular or body parts; or protect cells and the whole body; they may serve in transporting gases or other substances, or as important part of membranes; or they may be working as enzymes, which perform all chemical reactions happening in our cells and body.
Protein structures, like their functions, vary greatly. However, they are all made of combinations of twenty different amino acids available in nature, and all amino acids share the same basic structure as shown in figure \(\PageIndex{1}\) below.
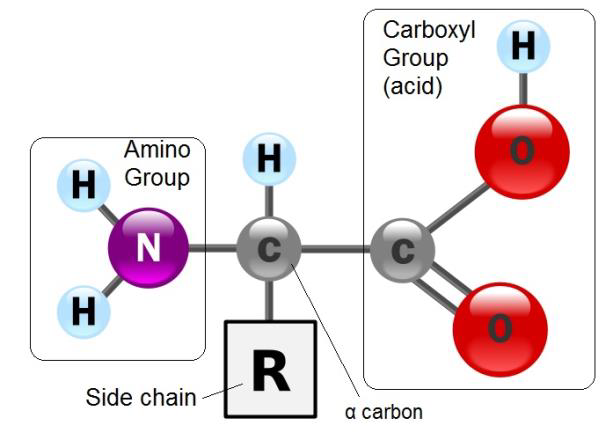
Figure \(\PageIndex{1}\) All amino acids have a central α carbon covalently bound to a hydrogen, an amino functional group (-NH2), a carboxyl (acid) functional group (-COOH), and a side chain R. R represents any of the twenty different functional groups found in amino acids.
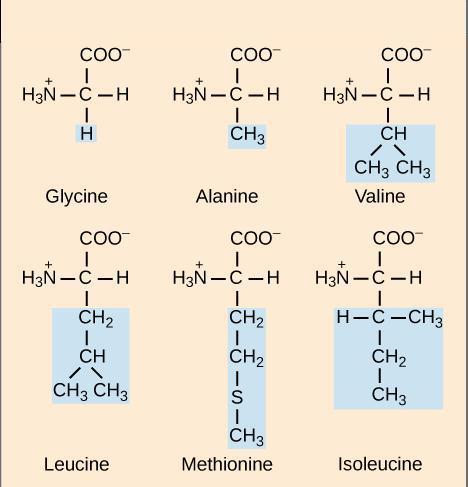
Figure \(\PageIndex{2}\) Six different amino acids are shown. All amino acids have in common a central α carbon covalently bound to a hydrogen (shown here on the right), an amino functional group (represented as –NH3+), here, and shown here on the left), a carboxyl (acid) functional group (shown on the top as -COO-,). Different amino acids have a different side chain R (shown on the bottom in blue)
Depending on the amino acid side chain (R) different amino acids may be polar or nonpolar. The properties of the resulting protein, including its shape and function, are determined by the sequence and types of amino acids found in the polypeptide. Two amino acids join to form a dipeptide, three amino acids form a tripeptide, four a tetrapeptide, and many amino acids form a polypeptide.
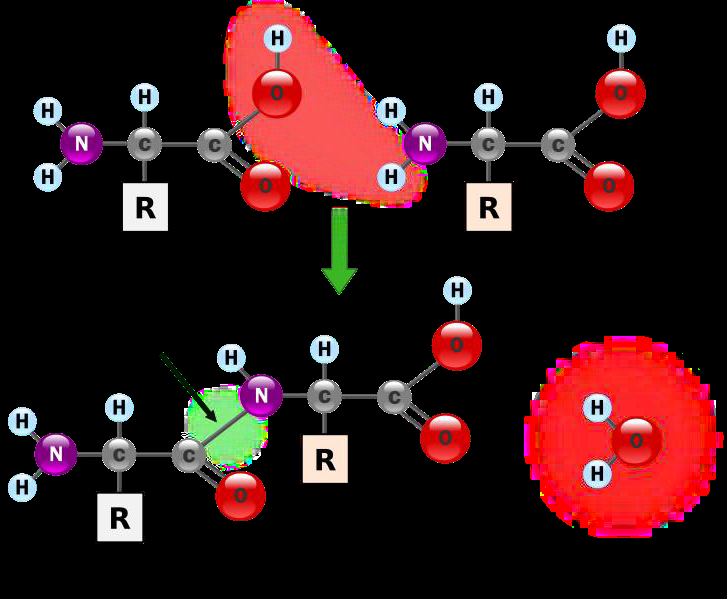
Figure \(\PageIndex{3}\) A dipeptide is formed when amino acid 1 and amino acid 2 are joined in a dehydration synthesis reaction to form a peptide bond. -OH from amino acid 1 and -H from amino acid 2, both shown in the red cloud, join to form a water molecule. This allows carbon of amino acid 1 on the left and nitrogen of amino acid 2 on the right to form a peptide bond
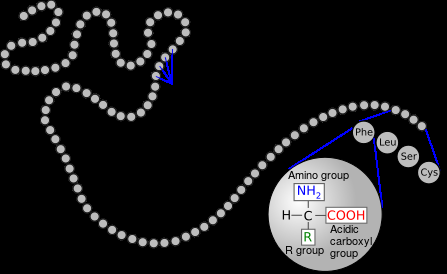
Figure \(\PageIndex{4}\) Each pearl represents an amino acid, and the whole pearl necklace represents a polypeptide. Polypeptides may have up to thousands of amino acids
The terms protein and polypeptide are often used exchangeable. However, a protein usually refers to the functioning macromolecule, whereas polypeptide refers to the sequence of amino acids linked by peptide bonds not necessarily functional. A polypeptide folded into a specific three-dimensional shape, determined by the interactions among its amino acids, is what we usually call a protein.
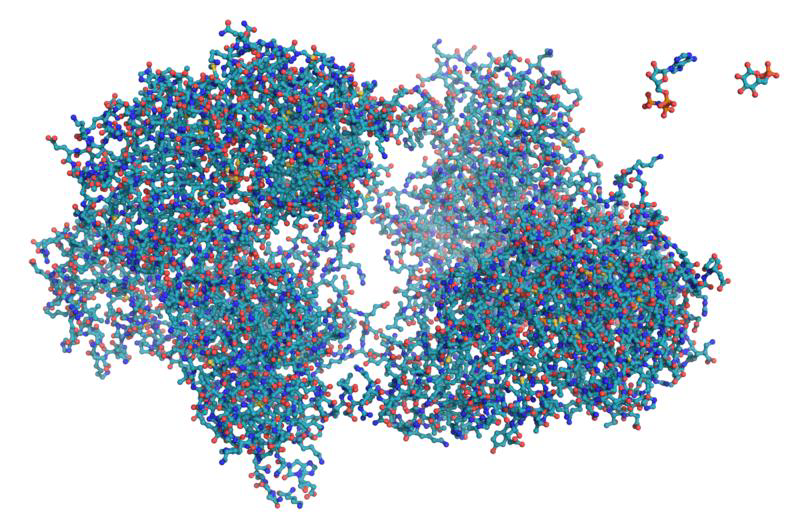
Figure \(\PageIndex{5}\) A computer generated three-dimensional representation of a protein (hexokinase) made of more than 400 amino acids. Each individual amino acids cannot be differentiated from the rest here. Each tiny ball represents an element (e.g. carbon is blue, oxygen is red, etc.). The folding of a polypeptide in the space gives shape to it, and its shape determines its function.
| Type | Examples | Functions |
|---|---|---|
| Enzymes |
Peptidase Lipase |
Increase speed of chemical reactions in the body (all metabolic reactions) |
| Transport |
Hemoglobin Albumin |
Carry substances in the blood or lymph throughout the body |
| Structural |
Collagen Tubulin |
Provide structural support Construct different structures, like the cytoskeleton (inner skeleton in a cell) |
| Hormones |
Insulin Oxytocin |
Chemical messengers that coordinate the activity of different body systems |
| Defense |
Immunoglobulins Complement |
Protect the body from foreign pathogens |
| Contractile |
Actin Myosin |
Participate in muscle contraction |
| Storage |
Myoglobin Ferritin |
Store oxygen in muscle cells Stores iron in the liver |
Concepts, Terms, and Facts Check
Study Questions Write your answer in a sentence form (do not answer using loose words)
1. What is a protein?
2. What elements are proteins made of?
3. What are the monomers (building blocks) of proteins?
4. What type of bond links neighboring amino acids in a protein?
5. What is the difference between the terms “polypeptide” and “protein”?
6. What are the functions of proteins listed in the module?
Not all biomolecules are pure protein, pure carbohydrate, or pure lipid. Conjugated proteins are protein molecules combined with another kind of biomolecule. For example, proteins combine with lipids to form lipoproteins. Lipoproteins are found in in the blood, where they act as carriers for less soluble molecules, such as cholesterol. Glycosylated molecules are molecules to which a carbohydrate has been attached. Proteins combined with carbohydrates form glycoproteins. Lipids bound to carbohydrates become glycolipids. Glycoproteins and glycolipids, are important components of cell membranes. The second part of the name is always the largest part. For example proteoglycans have more carbohydrate, while glycoproteins have more protein.
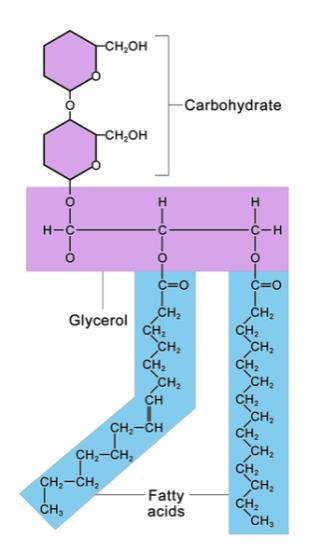
Figure \(\PageIndex{6}\) Lecithin, a glycolipid, is composed of a carbohydrate portion and a lipid portion (fatty acids plus glycerol part)
Concepts, terms, and facts check
Study Questions Write your answer in a sentence form (do not answer using loose words)
1. What is a conjugated protein?
2. What is a glycosylated molecule?
While proteins have a diversity of important functions, many proteins work as enzymes. Enzymes speed up chemical reactions. We can define enzymes as organic catalysts (organic because proteins are organic compounds; catalyst = “substance which speeds a chemical reaction but itself remains unchanged”). Chemical reactions catalyzed by enzymes are called enzymatic reactions. All synthesis, decomposition, and exchange reactions occurring in our cells and body tissues are enzymatic reactions.
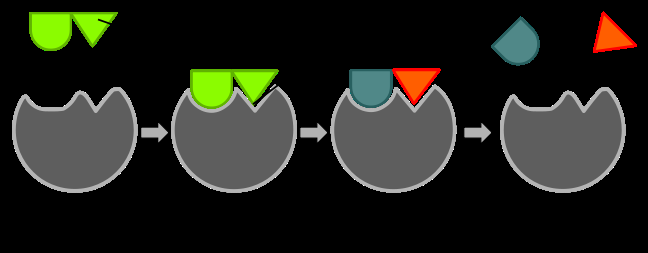
Figure \(\PageIndex{1}\) Mechanism of enzyme action. In this example, the green substrate is catalyzed by the gray pacman-like enzyme into blue and red products. This is a decomposition reaction, which would also require water (not shown).
All chemical reactions begin with one or more substances that enter into the reaction. Substances in our cells and body tissues that enter into the reaction are called substrates. The one or more substances produced by a chemical reaction are called products. Substrate and enzyme bind in the active site of the enzyme (a space that matches the shape of the substrate). There is a very brief moment when an enzyme/substrate complex forms and then this complex transitions into an enzyme/product complex. The product is then released. The enzyme is not changed by the reaction and once the products are released is ready to bind more substrate. See the mechanism of enzyme action in figure \(\PageIndex{1}\) above.
Enzymes are commonly named using the suffix –ase. The prefix (or the root of the word) provides information about the type of reaction a particular enzyme catalyzes. For example, lactase is an enzyme that breaks down the disaccharide lactose; protease is an enzyme that breaks down proteins, DNA polymerase is an enzyme involved in making DNA. Generally speaking, a “somethingase” is an enzyme that does something.
Concepts, terms, and facts check
Study Questions Write your answer in a sentence form (do not answer using loose words)
1. What is an enzyme?
2. What is an enzymatic reaction?
3. What is an “active site” in an enzyme?
4. What is the difference between substrate and product?
5. What is the suffix used to name enzymes?

