5.3: Pulse Oximetry
- Page ID
- 10014
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Oxygen saturation, sometimes referred to as ‘‘the fifth vital sign,” should be checked by pulse oximetry in all breathless and acutely ill patients (British Thoracic Society, 2008). SpO2 and the inspired oxygen concentration should be recorded on the observation chart together with the oximetry result. The other vital signs of pulse, blood pressure, temperature, and respiratory rate should also be recorded in situations where supplemental oxygen is required.
Pulse oximetry is a painless, non-invasive method to monitor SpO2 intermittently and continuously. The use of a pulse oximeter (see Figure 5.1) is indicated in patients who have, or are at risk for, impaired gaseous exchange or an unstable oxygen status.
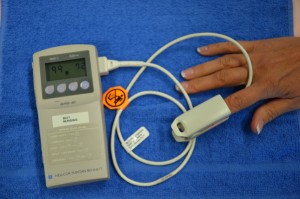
Figure 5.1 Pulse oximeter
The pulse oximeter is a probe with a light-emitting diode (LED) that is attached to the patient’s finger, forehead, or ear. Beams of red and infrared light are emitted from the LED, and the light wavelengths are absorbed differently by the oxygenated and the deoxygenated hemoglobin (HgB) molecules. The receiving sensor measures the amount of light absorbed by the oxygenated and deoxygenated Hgb in the arterial (pulsatile) blood. The more HgB that is saturated with oxygen, the higher the SpO2, which should normally measure above 95%.
Pulse oximeters have an indicator of signal strength (such as a bar graph, audible tone, waveform, or flashing light) to show how strong the receiving signal is. Measurements should be considered inaccurate if the signal strength is poor.
Pulse oximeters will also indicate heart rate by counting the number of pulsatile signals. To ensure accuracy, count the patient’s pulse rate by taking the pulse and comparing it to the pulse rate shown on the pulse oximeter.
Limitations
The most common cause of inaccuracy with pulse oximeters is motion artifact. Patient movement can cause pulsatile venous flow to be incorrectly measured as arterial pulsations, thus producing an inaccurate oximetry and pulse-rate reading.
Another common cause of inaccuracy is poor peripheral perfusion. Poor peripheral perfusion can be caused by conditions such as hypothermia, peripheral vascular disease, vasoconstriction, hypotension, or peripheral edema (Perry, Potter, & Ostendorf, 2014). A forehead probe can be used for patients with decreased peripheral perfusion.
Conditions such as jaundice, as well as intravascular dyes and carbon monoxide in the blood, can also influence oximetry readings. Anemic patients with low Hgb may have a normal SpO2 reading, even though the available oxygen is not enough to meet the metabolic demands of the body. Patients with elevated bilirubin concentrations may also have falsely low SpO2 readings (Howell, 2002).
Application of Pulse Oximetry
If measuring SpO2 by attaching the probe to a finger or toe, check the radial or pedal pulse and capillary refill of the finger or toe you plan to use. If the patient’s extremities are cold, you could try to warm his or her hands in yours, or apply warm towels to improve perfusion.
The patient’s finger or toe should be clean and dry. Check that the patient does not have artificial nails or nail polish, as both will influence the light transmission and should, therefore, be removed before applying pulse oximetry.
Check that the probe is positioned properly so that optical shunting (when light from the transmitter passes directly into the receiver without going through the finger) does not occur.
Bright ambient light may also affect the accuracy of pulse oximetry readings.
Hazards of Pulse Oximetry
Pulse oximetry is generally considered to be a safe procedure. However, tissue injury may occur at the measuring site as a result of probe misuse. Pressure sores or burns are possible effects of prolonged application (>2 hours).
Critical Thinking Exercises
- You are checking your patient’s SpO2 but the signal strength on the pulse oximeter is poor. What would be your next steps?
- Your patient has been admitted with a diagnosis of carbon monoxide poisoning with an SpO2 of 98%. What does this reading tell you?


