3.5: Energy and Calories
- Page ID
- 1620
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Skills to Develop
- Estimate your caloric contribution of each type of macronutrient in foods you commonly consume.
Energy is essential to life. You must eat to have energy. You must go to bed at a decent time, so that when you wake up in the morning, you will not be too tired and you will have sufficient energy for the next day’s activities. Energy is also everywhere in our environment: sunlight, wind, water, plants, and animals. All living things use energy every day. Energy can be defined as the quantity of work a particular system can perform, whether it be a growing child’s body or a train transporting passengers from one place to another. Energy also helps us perform daily functions and tasks such as breathing, walking up a flight of steps, and studying for a test.
Energy is classified as either potential or kinetic. Potential energy is stored energy, or energy waiting to happen. Kinetic energy is energy in motion. To illustrate this, think of an Olympic swimmer standing at the pool’s edge awaiting the sound of the whistle to begin the race. While he waits for the signal, he has potential energy. When the whistle sounds and he dives into the pool and begins to swim, his energy is kinetic (in motion).
Some basic forms of energy are:
- Thermal (heat) energy. We can say that a cup of hot tea has thermal energy. Thermal energy is defined as the collective, microscopic, kinetic, and potential energy of the molecules within matter. In a cup of tea, the molecules have kinetic energy because they are moving and oscillating, but they also possess potential energy due to their shared attraction to each other.
- Chemical energy. In your body, blood sugar (glucose) possesses chemical energy. When glucose reacts chemically with oxgen, energy is released from the glucose. Once the energy is released, your muscles will utilize it to produce mechanical force and heat.
- Electrochemical energy. In the body, electrical impulses travel to and from the brain encoded as nerve impulses. Once the brain receives an electrical impulse it causes the release of a messenger chemical (glutamate, for example). This in turn facilitates electrical impulses as they move from one neuron to another.
The Calorie Is a Unit of Energy
The amount of energy in nutrients can be quantified into specific units that can be measured. The unit of measurement that defines the energy contained in a energy-yielding nutrient is called a calorie. A calorie is the amount of energy in the form of heat that is required to heat one gram of water one degree Celsius. To measure the number of calories in a particular food substance, a certain amount of food is burned in a device called a calorimeter. As the food burns, heat is created. The heat dissipates to the surrounding water while a thermometer detects the change in temperature of the water. You can even perform calorimetry at home with a more basic device. However, it is not likely that you will use this device to measure calorie content in the foods that you eat since mathematical formulas have been developed to estimate caloric content.
Estimating Caloric Content
The energy contained in energy-yielding nutrients differs because the energy-yielding nutrients are composed of different types of chemical bonds. A carbohydrate or a protein yields 4 kilocalories per gram, whereas a lipid yields 9 kilocalories per gram. A kilocalorie (Calorie) is the amount of heat generated by a particular macronutrient that raises the temperature of 1 kilogram of water 1 degree Celsius. A kilocalorie of energy performs one thousand times more work than a calorie. On the Nutrition Facts panel, the calories within a particular food are expressed as kilocalories, which is commonly denoted as “Calories” with a capital “C” (1 kcal = 1 Calorie = 1,000 calories).
Calculating the number of Calories in commercially prepared food is made fairly easy since the total number of Calories in a serving of a particular food is listed on the Nutrition Facts panel. If you wanted to know the number of Calories in the breakfast you consumed this morning just add up the number of Calories in each food. For example, if you ate one serving of yogurt that contained 150 Calories, on which you sprinkled one half of a cup of low-fat granola cereal that contained 209 Calories, and drank a glass of orange juice that contained 100 Calories, the total number of Calories you consumed at breakfast is 150 + 209 + 100 = 459 Calories. If you do not have a Nutrition Facts panel for a certain food, such as a half cup of blueberries, and want to find out the amount of Calories it contains, go to MyFood-a-pedia, a website maintained by the USDA.
Interactive 3.2
Try using "Fooducate" which will give each food a letter grade (A, B, C, D or F) for how healthy it is! Just type any food into the search box at the top. For example, Fig Newton Cookies get a C+.
Estimating the Amount of Energy from Each Macronutrient
Also listed on the Nutrition Facts panel are the amounts of total fat, total carbohydrate, and protein in grams. To calculate the contribution of each macronutrient to the total kilocalories in a serving, multiply the number of grams by the number of kilocalories yielded per gram of nutrient. For instance, from the Nutrition Facts panel for yogurt given in Figure 3.7, the protein content in one serving is 7 grams. Protein yields 4 kilocalories per gram. The number of kilocalories from protein is calculated by using the following equation:
# of grams of protein × 4 kilocalories/gram of protein 7 grams × 4 kilocalories/gram = 28 kilocalories
(# kilocalories from protein ÷ total kilocalories per serving) × 100 (28 ÷ 150) × 100 = 18.7 percent
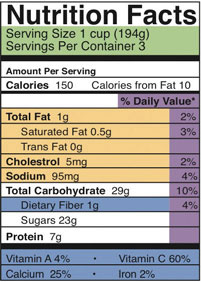
Figure 3.5.1: A Nutrition Facts Panel for Yogurt: Because the total number of kilocalories in each serving of yogurt is 150, the percent of energy obtained from protein is calculated by using the following equation:
Key Takeaways
- Energy is vital to life and is categorized into two types—kinetic and potential. There are also different forms of energy such as thermal, chemical, and electrochemical.
- Calories are a measurement of a specific quantity of energy contained in foods. The number of calories contained in a commercially prepared food is listed on the Nutrition Facts panel.
Discussion Starter
- Estimate the total number of kilocalories that you have eaten so far today. What percentage of the kilocalories you consumed was obtained from carbohydrates? Protein? Fat?

