1.13: Management of Salvage Laryngectomy Defects and Related Wound Complications
- Page ID
- 17622
OPEN ACCESS ATLAS OF OTOLARYNGOLOGY, HEAD & NECK OPERATIVE SURGERY
MANAGEMENT OF SALVAGE LARYNGECTOMY DEFECTS AND RELATED WOUND COMPLICATIONS
Ryan Orosco and Eben Rosenthal
The goal of this chapter is to provide a working framework for the management of fistulae and complex wounds related to salvage total laryngectomy (sTL).
sTL is an effective oncologic intervention for recurrence following (chemo)radiation therapy, but is frequently associated with postoperative wound problems including salivary fistulae, infection, stomal dehiscence, and strictures etc. Overarching therapeutic goals in the acute postoperative period of sTL are optimization of wound healing, prevention of pharyngocutaneous fistulae, avoidance of prolonged hospitalization, and initiation of oral intake. Wound breakdown and fistula formation are associated with functional problems such as dietary restrictions, limited communication, and stricture formation
Steps to avoid and/or manage wound healing complications in the radiated neck are a critical element of achieving these objectives. However how to avoid wound healing complications in these high risk patients remains controversial and there is no consensus as to optimal preventative strategies. A stepwise approach can be summarized as follows:
- Prevention
- Conservative Management
- Negative Pressure Dressings
- Reconstruction
- Regional flaps
- Free tissue
There is however evidence that use of vascularized tissue for pharyngeal reconstruction at the time of sTL can help prevent and diminish complications1. When complications do occur, management is sometimes escalated in a conservative manner. A more aggressive approach with tissue transfer may be required if conservative interventions fail or may be instituted early on in “unsafe” neck wounds.
Prevention
Although primary mucosal closure is technically possible in some sTL cases, several factors are known to predispose to fistulae and wound breakdown. These include hypothyroidism, bilateral neck dissection, and previous chemotherapy2. Once a fistula occurs, it is associated with significantly longer hospital stay and stricture rates3,4
Contrast (barium) swallow radiographs are commonly employed postoperatively to exclude a leak and has a high specificity (approx. 94%), although the sensitivity is lower (approx. 26%). If a swallow test is negative, it is still possible that a leak will manifest later3. Therefore swallow studies should be done based on clinical suspicion rather than routinely.
Many tertiary institutions consider sTL an indication for vascularized tissue placement, usually a radial free forearm flap, but pectoralis major muscle overlay flaps or anterolateral free thigh flaps are also commonly employed. In a study of over 350 patients undergoing sTL at 7 high-volume academic centers, patients were grouped as either primary closure, pectoralis onlay flap, or closure with a free flap. Notably, most vascularized tissue flaps were interposed free flaps. Multivariate analysis demonstrated a significantly lower fistula rate with either flap technique. Furthermore, those patients who underwent closure using vascularized tissue could be managed without surgical intervention after occurrence of a fistula, and healed faster1. In other series, the use of onlay myofascial pectoralis major flaps were not superior to inlay myocutaneous flaps5. A recent meta-analysis of 33 studies from 2000 to 2013 reported a fistula rate of 14.3% (N=326 of 2177) for primary total laryngectomy and 28% in sTL. If a flap-reinforced closure was utilized, the fistula rate was much lower in the salvage setting (10%)6. A flap may be used even when there is sufficient mucosa to close the defect primarily.
Though no consensus exists as to the optimal choice of vascularized tissue with sTL, free tissue transfer is often preferred. Although a range of vascularized tissues have been used, including free anterolateral thigh and rectus abdominis flaps, interposition radial free forearm flaps are most commonly used (Figure 1).
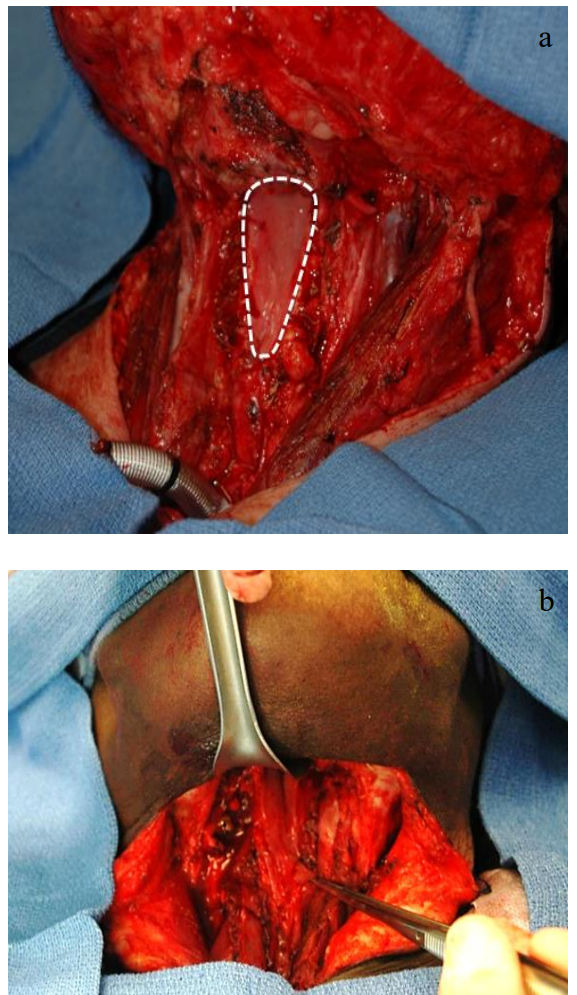
Figures 1a-d: Typical pharyngeal defects; RFFF; RFFF in place
After total laryngectomy with neck dissection(s), a wound is ideal for reconstruction, and the donor vessels are accessible for microvascular anastomosis. If one has a good reconstructive team, the authors favor using a free flap, reserving the pectoralis major flap for potential salvage repair should the flap fail, to cover exposed major vessels, and for emergent procedures.
Intraoperative fluorescence angiography may offer improvements in the surgeon’s ability to identify poor vascularity of the soft tissues that may indicate patients at-risk for impaired wound healing. Intraoperative fluorescence angiography uses systemically injected indocyanine green (5 - 10 mg, IV push) to measure the relative perfusion in tissues and has been shown to have value in breast reconstructive surgery, and may similarly have applications in head and neck reconstruction.
Use of antibiotics remains controversial, as there is no data to support antibiotics given for longer than 24 hours after surgery.
Conservative management
When wound complications occur following sTL, the wound management strategy evolves over time based on the response to medical or surgical interventions (Figure 2).

Figure 2: Management of wound breakdown after total laryngectomy (NPD = Negative pressure dressings)
Antibiotics are generally not indicated for uncomplicated fistulae. Once a cervical infection does occur, diagnosis (fistula, cellulitis, abcess, etc) should be determined before antibiotics are administered as it could mask a significant issue that will delay definitive management.
Surgical interventions for diagnostic and therapeutic reasons are based on bedside evaluation; CT scans are generally not needed. Contraindications to conservative wound management generally include a significant amount of infected tissue, and exposed large caliber vessels at risk of hemorrhage.
Initial danger signs usually manifest on Days 5-8 and may include lethargy, sweats, and cervical erythema (usually above the suture line).
The following days to weeks usually involve aggressive debridement and removal of necrotic tissue, which is often the source of infection.
Following initial wound debridement, conservative wound management is instituted as an inpatient with wound dressings applied until the wound can be considered “safe” for discharge (no risk of great vessel rupture). Conservative wound therapies in radiated wounds usually require 5-8 days of inpatient care to see the wound improve, and several months of home care to achieve maximal healing. Wound dressings frequently consist of wet-to-dry saline dressing changes or negative pressure wound care (Table 1).
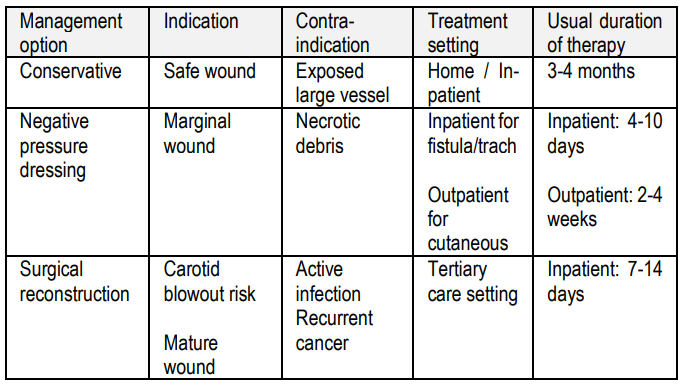
Table 1: Management options of wound breakdown after sTL
If the wound does not show improvement within several days, then definitive management should be planned to protect the great vessels using a pectoralis major muscle onlay flap and maintaining a patent pharyngocutaneous fistula.
Negative pressure dressings
Negative pressure dressings promote wound healing by applying a vacuum through a special sealed dressing. The continuous vacuum draws out fluid from the wound and increases blood flow to the area. (Figure 3). Although the negative pressure dressings will not achieve anything that conservative management will not do over time, they do accelerate wound healing by 20-40%.

Figure 3: Negative pressure dressing application of subatmospheric pressure using an electrical pump
The mechanism of action for negative pressure dressings is not well understood, but it is thought reduce bacterial content, improve blood supply, and promote contraction of the wound. Possible mechanisms of negative pressure dressings include:
- Removing secretions
- Debridement
- Reducing bacterial content
- Improving blood flow
General indications for negative pressure dressings include stomal seeding from a salivary fistula, non-healing wounds, exposure of great vessels, donor site skin grafts, and to support reconstructive tissues in a compromised wound. Negative pressure dressings should not be used if there is active infection or necrotic tissue.
Negative pressure dressings can be used to manage complex wounds. They can be directly applied to active pharyngocutaneous fistulae to accelerate healing in softtissue7, as intraluminal dressings during secondary pharyngeal repairs8, and can be placed directly over major vessels or a vascular pedicle.
Negative pressure dressings have 4 basic components:
- Dressing or filler material that is shaped to the wound contours and directly applied to the wound. It may consist of reticulated sponge (Figure 4) gauze, or honeycombed textiles. It can be safely placed over large, exposed vessels, free flap pedicles, and other “at risk” areas7, 8.
- Occlusive plastic tape dressing is applied over the sponge and taped to the surrounding skin to create an airtight sealed cavity over the wound (Figure 4)
- Plastic tubing that connects the sealed airtight cavity over the wound with the vacuum pump (Figure 5)
- Vacuum pump or wall suction to generate a constant negative pressure varying between −125 and −75 mmHg depending on the material used and patient tolerance (Figure 6)
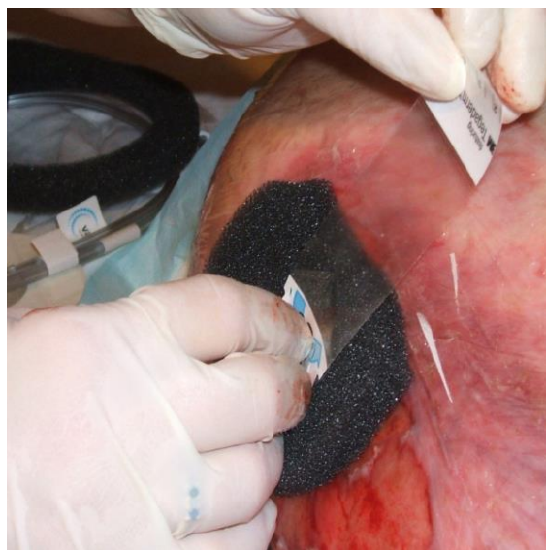
Figure 4: Application of adherent plastic sheeting over a foam filler (Wikipedia)
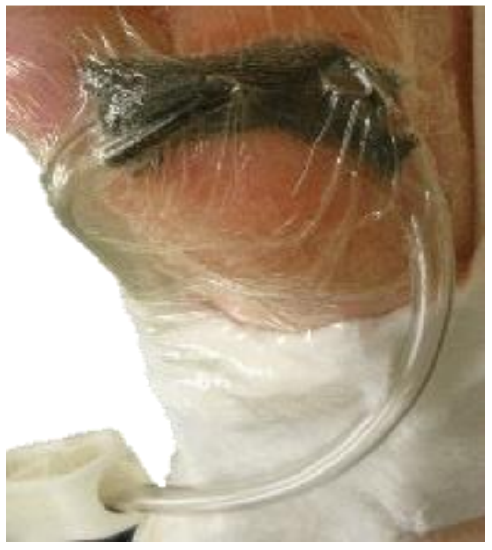
Figure 5: Example of tubing arrangement
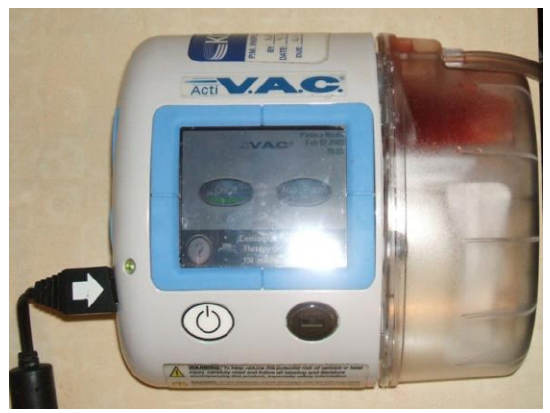
Figure 6: Example of a vacuum pump (Wikipedia)
Using negative pressure dressings on the neck presents significant challenges due to the presence of a tracheostomy or tracheostoma, pharyngocutaneous fistula, hair, poor skin quality, and contamination with saliva, all of which make it difficult to achieve an occlusive seal.
Negative pressure dressings are changed every 3-6 days. Generally, 4-10 days of inpatient therapy is required, followed by 2- 4 weeks’ outpatient wound care.
Negative pressure dressings are sometimes not used due to cost (sponges are often cheap, but pump rentals in the USA can cost hundreds of dollars/day). When considering use of negative pressure dressings, the associated costs should be balanced against the cost savings and quality of life of a potentially shorter inpatient hospital stay.
Aggressive management
Escalation to reconstruction is the next level of complexity in the wound management strategy (Table 1). Indications include imminent risk of carotid or internal jugular bleeding, or a very mature wound ready for definitive repair. Relative contraindications to flap repair include active infection or recurrent / persistent cancer.
Because of the simplicity of the pectoralis flap in postoperative sTL wound situations, free tissue transfer is less commonly used.
In a vessel-depleted neck, the pectoralis major flap or less commonly the latissimus dorsi flap may be used. The pectoralis major flap is particularly well suited for emergent and urgent postoperative wound complications as it done using standard equipment with the patient in a supine position, and without specialized nursing at any time of the day or night, and it is a very robust flap. Therefore the authors use the pectoralis major flap as a backup flap to manage sTL complications rather than a primary reconstructive strategy.
There are situations in which free tissue transfer is ideal for the management of sTL complications. Dissection of quality donor vessels is however paramount, and preparation of donor vessels suitable for microvascular anastomosis may be challenging in an unfavorable postoperative wound in a previously irradiated neck, particularly in the presence of sepsis, and may lead to exposure of additional segments of the carotid artery.
In a vessel-depleted neck it is worth considering using the internal mammary vessels as they are generally well protected from cardiovascular disease. Access is achieved by removing a short section of the 2nd rib as it joins the sternum. It is usually better to use the right side in case there is future need for cardiac revascularization.
Summary and conclusions
- Delayed reconstruction following sTL wound complications is challenging and emphasises the importance of implementing preventative strategies in this high risk patient population
- Use vascularized tissue reconstruction as a preventative strategy to manage sTL defects
- Reconstruction at the time of tumor extirpation decreases salivary fistula rates and limits the duration of complications when they do occur
- Given availability of the required expertise and facilities, free flap reconstruction is the preferred technique for upfront reconstruction
- The pectoralis major flap is however a reliable, simple flap that can be used in emergent or unanticipated circumstances
- Management of complex wounds following sTL progresses through a stepwise algorithm aimed at the creation of a safe wound and promotion of subsequent healing
- Wound debridement and management of active infection should be followed by conservative wound dressings and sometimes negative pressure dressings
References
- Patel, U.A., et al., Impact of pharyngeal closure technique on fistula after salvage laryngectomy. JAMA Otolaryngol Head Neck Surg, 2013. 139(11): p. 1156-62
- Andrades, P., et al., Fistula analysis after radial forearm free flap reconstruction of hypopharyngeal defects. Laryngoscope, 2008. 118(7):1157-63
- Sweeny, L., et al., Incidence and outcomes of stricture formation postlaryngectomy. Otolaryngol Head Neck Surg, 2012. 146(3):395-402
- Withrow, K.P., et al., Free tissue transfer to manage salvage laryngectomy defects after organ preservation failure. Laryngoscope, 2007. 117(5): 781-4
- Khan, N.A., et al., Fistula rates after salvage laryngectomy: comparing pectoralis myofascial and myocutaneous flaps. Laryngoscope, 2014. 124(7):1615-7
- Sayles, M. and D.G. Grant, Preventing pharyngo-cutaneous fistula in total laryngectomy: a systematic review and meta-analysis. Laryngoscope, 2014. 124(5):1150-63
- Asher, S.A. et al., Negative pressure wound therapy in head and neck surgery. JAMA Facial Plast Surg, 2014. 16(2):120-6
- Asher, S.A., et al., Intraluminal negative pressure wound therapy for optimizing pharyngeal reconstruction. JAMA Otolaryngol Head Neck Surg, 2014. 140(2):143-9
- Rosenthal, E.L., et al., Use of negative pressure dressings in head and neck reconstruction. Head Neck, 2005. 27 (11):970-5
- Shreenivas, S., J.S. Magnuson, and E.L. Rosenthal, Use of negative-pressure dressings to manage a difficult surgical neck wound. Ear Nose Throat J, 2006. 85(6):390-1
Authors
Ryan K Orosco, MD
Instructor of Otolaryngology
Division of Head and Neck Surgery
Department of Otolaryngology
Stanford University, CA, USA
orosco@stanford.edu
Eben L. Rosenthal, MD
Ann and John Doerr Medical Director
Stanford Cancer Center
Professor of Otolaryngology, Stanford
University School of Medicine
875 Blake Wilbur Drive
C-2213 Stanford, CA 94305
elr@stanford.edu
Editor
Johan Fagan MBChB, FCS(ORL), MMed
Professor and Chairman
Division of Otolaryngology
University of Cape Town
Cape Town, South Africa
johannes.fagan@uct.ac.za

The Open Access Atlas of Otolaryngology, Head & Neck Operative Surgery by Johan Fagan (Editor) johannes.fagan@uct.ac.za is licensed under a Creative Commons Attribution - Non-Commercial 3.0 Unported License



