1.3: Sentinel Lymph Node Biopsy (SLNB) Techniques for Cutaneous and Mucosal Malignancy of the Head and Neck
- Page ID
- 15428
OPEN ACCESS ATLAS OF OTOLARYNGOLOGY, HEAD & NECK OPERATIVE SURGERY
SENTINEL LYMPH NODE BIOPSY (SLNB) TECHNIQUES FOR CUTANEOUS & MUCOSAL MALIGNANCY OF THE HEAD & NECK
Cecelia E. Schmalbach, Carol R. Bradford
Sentinel lymph node biopsy (SLNB) is a minimally invasive procedure used to localize and sample the first echelon individual draining nodes of a malignant tumor and to identify occult (micro-metastatic) disease. In doing so, the technique provides accurate regional staging and identifies patients who may benefit from additional surgery and/or adjuvant therapy. Even though sentinel nodes are by definition, confined to the first echelon draining nodes, technological advances now permit multiple echelons to be detected.
The advantage of SLNB is that patients are saved the morbidity associated with traditional selective neck dissection (SND). In addition, the pathologist is provided a limited number of lymph nodes for comprehensive evaluation that includes microsectioning and cancer-specific immunohistochemical staining. This comprehensive evaluation is not feasible from time and cost standpoints for all nodes harvested in a traditional SND which can exceed twenty nodes.
The combination of auditory cues from the radioactive nucleotide identified with the handheld gamma probe and visual cues from the blue dye injection provides the highest success rate for SLN identification. For this reason, the combined modalities are considered standard of care in many countries. However, when nuclear medicine resources are unavailable, one can attempt to perform the SLNB technique as outlined below using intradermal blue dye alone and employing a wider surgical approach.
Indications
Indications for SLNB are cancer-type specific. The technique should be considered in the workup of patients with cutaneous melanoma1, Merkel cell carcinoma2, early oral cavity squamous cell carcinoma3 and aggressive, recalcitrant cutaneous squamous cell carcinoma4.
Cutaneous Melanoma
All patients presenting with clinically and radiographically localized Stage I and II disease and a tumor depth of invasion >1 mm should be offered SLNB. In addition, select patients with thinner melanomas measuring a depth of 0.75 - 1.0mm in the setting of poor prognostic features to include young age, tumor regression, ulceration, mitotic rate >1 feature/mm2, angiolympathic invasion, and a positive deep margin can also be offered the procedure.
Merkel Cell Carcinoma (MCC)
MCC is a rare neuroendocrine cutaneous cancer common in the head and neck region of elderly patients. Due to the high rate of regional metastasis, patients with localized MCC can be offered SLNB.
Cutaneous Squamous Cell Carcinomas (cSCC)
It is also recognized that a subset of aggressive, recalcitrant cSCCs such as those seen in the immunosuppressed patient population, may benefit from regional restaging with SLNB.
Mucosal SCC
Oral cavity SLNB studies demonstrate a 90-100% detection rate for occult nodal metastasis when conducted in the setting of experienced institutions5. SLNB has been adopted for early stage mucosal SCC of the oral cavity. Patients with early stage T1 and T2 tumors can be considered for this staging modality if they are clinically and radiographically node negative. Other mucosal subsites within the head and neck region have not been included due to the challenge of anatomic access for preoperative injections described below.
Regardless of cancer type, all patients require a punch biopsy or narrow margin (1-3 mm) excisional biopsy of the primary tumor for diagnosis (Figure 1). Wide local excision (WLE) with larger 0.5-2 cm margins is discouraged because the surrounding tissue which contains the true draining lymphatics of the cancer is removed in the process and thereby can compromise sentinel lymph node (SLN) mapping and SLNB accuracy.

Figure 1: Melanoma of left temple following diagnostic punch biopsy with 1 cm margin marked for definitive wide local excision following SLNB. Note the surrounding solar changes
Preoperative work-up
SLNB is traditionally considered a staging modality. Therefore, thorough clinical physical examination of all draining nodal basins is required. If available, radiographic evaluation with contrasted CT scanning or ultrasound should be considered. Any patients with suspicious nodal disease clinically or radiographically should undergo fine needle aspiration. If negative, SLNB can be considered.
Collaborative Effort
SLNB is a collaborative team effort. The multidisciplinary oncology team should ideally include dermatopathology, radiology, dermatology, otolaryngology, surgical oncology, radiation oncology and medical oncology to review the final pathology and appropriate use of adjuvant therapy.
There is a significant surgical learning curve. The surgeon should ideally meet with the nuclear medicine team during the preoperative stage. At some institutions the surgeon is responsible for injection of the radioactive colloid. Data from the first melanoma Multicenter Selective Lymphadenectomy Trial (MSLT-1) demonstrated a 50-case learning curve to reduce the rate of false omission.6
The nuclear medicine team must be well versed in injection of the radioactive nucleotide, because if the material is spilled or splattered during the initial injection, the gamma probe will be rendered useless and the case will need to be rescheduled. For cutaneous injections, clear communication with the nuclear medicine team is paramount to ensure that the correct lesion is injected because most skin cancer patients have significant solar changes (Figure 1).
Equipment for SLNB
- Radioactive colloid
- Lymphoscintigraphy or Single-photon emission computed tomography/computed tomography (SPECT/CT)
- Gamma probe/monitor: a hand-held device containing a scintillation counter and is used intraoperatively following injection of the radioactive colloid to locate the sentinel lymph nodes by their radioactivity (Figure 2)
- Blue dye (methylene blue preferred)
- Local anesthesia
- Scalpel
- Small retractors
- Forceps
- Scissors
- Electric cautery/bipolar
- Facial nerve monitor for tumors draining to parotid nodal basin
- Nerve stimulator
- Needle driver and closing suture
- Pathology cups with formalin

Figure 2: Example of gamma probe (front) and monitor. The number on the screen depicts the radioactivity detected by the probe
SLNB Technique
- 2 to 4 hours prior to surgery patients undergo an intradermal or mucosal 4-quadrant injection of radioactive colloid tracer into the tissue immediately surrounding the malignant lesion.
- Delayed imaging provides the surgeon with a ‘road map’ of the anticipated number and location of sentinel nodes.
- Traditionally, 2-dimenstional planar imaging (lymphoscintigraphy) was used (Figure 3)

Figure 3: Traditional lymphoscintigraphy with 2-dimensional planar imaging. This left temple melanoma drained to the parotid basin and level IIA
- Recently, fused single-photon emission computed tomography/computed tomography (SPECT-CT) has emerged as a superior modality allowing for increased nodal harvest and improved melanoma disease free survival. The added anatomic information of SPECT-CT is especially helpful in the parotid region and Level IB (Figures 4a-c)
- The superior detail provided by SPECTCT allows for smaller incisions and often alters the surgical approach compared to traditional planar imaging. Regardless of imaging technique, the radioactive injection is especially helpful for midline lesions which have a propensity to drain bilaterally
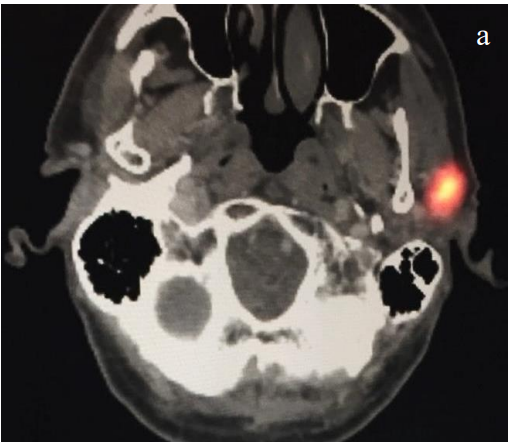

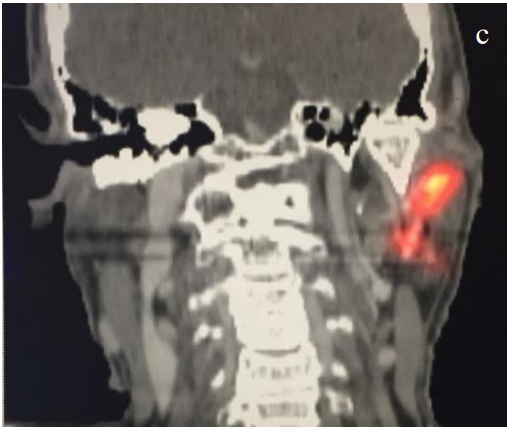
Figures 4ab (axial) and 4c (coronal): Same temple melanoma imaged with SPECT/CT following radioactive colloid injection. Note the increased anatomic information with a clear sentinel node in the left parotid basin and the anatomic CT correlate (white arrow)
- Following radioactive injection, patients are brought to the operating room and placed under general anesthesia
- A second intradermal injection of blue dye (methylene blue is preferred) into the surrounding cutaneous or mucosal tissue is completed (Figure 5). Typically 1-2ml is adequate. Several minutes must pass prior to injection of local anesthesia with vasoconstrictors in order to allow for adequate dissemination throughout the immediate lymphatic system
- The patient is prepped and draped in the standard surgical fashion

Figure 5: Methylene blue dye injection into the subdermal region immediately surrounding the left temple melanoma lesion. Note the facial nerve monitor given drainage to the parotid nodal basin
- Due to the close proximity of head and neck primary tumours and surrounding draining nodal basins, WLE of the primary cancer with the standard recommended margins is typically performed first to avoid masking the surrounding nodal basins; this is known as radioactive “shine through” and renders the handheld gamma probe useless (Figure 6)

Figure 6: Resection of the primary temple melanoma to avoid radioactive shine-through. Note the circular demarcations identified with transcutaneous use of the gamma probe. These increased areas of radioactive correspond to the preoperative imaging depicted in Figures 3 and 4
- With cSCC, oral cavity SCC, and MCC, the primary site can be closed at the time of SLNB, ideally following pathologic evaluation of margins using frozen section. However, because frozen section carries a 10% false negative rate for melanoma, most surgeons will elect to delay definitive repair and closure of melanomas until negative permanent margins have been achieved
- With cSCC, oral cavity SCC, and MCC, the primary site can be closed at the time of SLNB, ideally following pathologic evaluation of margins using frozen section. However, because frozen section carries a 10% false negative rate for melanoma, most surgeons will elect to delay definitive repair and closure of melanomas until negative permanent margins have been achieved
- Following WLE, the handheld gamma probe is used to identify areas of increased radioactivity representing the location of the SLN
- Once identified, a small 1-3 cm incision is made directly over the area of activity
- Make the incision wide enough to allow for introduction of the entire circumference of the head of the gamma probe into the subcutaneous tissues (Figure 7)
- Using careful blunt dissection and minimal cautery, identify the SLN through the combined auditory cues of the radioactive nucleotide/gamma probe and visual clues from the blue dye (Figures 8, 9)
- Send each harvested SLN individually to the pathologist for permanent histologic evaluation and immunohistochemical staining if required
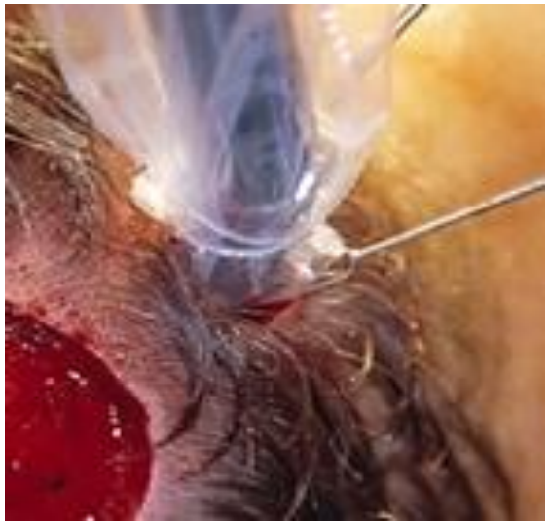
Figure 7: A 1-2 cm incision is made over the area of increased radioactivity. The incision should be large enough to introduce the entire head of the gamma probe

Figure 8: Sentinel node dyed by methylene blue

Figure 9: Methylene blue dye in lymphatic vessel (arrow) leading to sentinel node (*)
- Many sentinel lymph nodes are in close proximity to cranial nerves e.g. level IB lymph nodes are in close proximity to the marginal mandibular branch of the facial nerve; and nodes in level V are in close proximity to the accessory nerve. Detailed anatomical knowledge, use of careful blunt dissection, bipolar cautery and a nerve stimulator +/ monitor help to minimize the risk nerve injury
- In the parotid region, SLNB in experienced hands has proven to be safe and effective. Use of a facial nerve monitor is recommended (Figure 5)
- SLNB in the parotid region is done by placing the incision somewhere along the cosmetically favorable modified Blair incision as opposed to dissecting directly over the sentinel node as this provides for a more acceptable cosmetic result of a camouflaged scar which can easily be incorporated into the surgical approach in the event that a formal parotidectomy is required following a positive SLNB (Figure 10)

Figure 10: Preauricular approach to a parotid sentinel node. This small incision can easily be incorporated into a modified Blair incision should a formal parotidectomy be required following positive sentinel node biopsy
- The SLNB procedure is considered complete when the surgical background radioactivity drops to 10% of the hottest ex vivo SLN
- The SLNB harvested sites are irrigated
- Hemostasis is achieved and wounds sutured
Post-SLNB management
Positive SLNB
- Patients with a positive SLNB should ideally be returned to the operating room within 1-2 weeks for completion lymphadenectomy (CLND) of all atrisk nodal basins
- The surgeon should now have a clear understanding of the drainage pattern identified on radiographic imaging. However ultimately the nodal echelons to be dissected are based on the anatomic location of the primary tumor
- Cutaneous cancers of the lower face and neck should include Levels 1-4
- Because cancers higher on the cheek, scalp, temple, or forehead drain via parotid nodes en route to the cervical nodes, a parotidectomy with facial nerve preservation should be done
- Cutaneous lesions posterior to an imaginary coronal plane through external auditory canal, e.g. the posterior neck or scalp, require a posterolateral neck dissection up to the posterior midline of the neck, as well as the suboccipital and retroauricular nodes
- Patients with a positive oral cavity SLNB should have Levels 1-4 included
MCC warrants special consideration given that these elderly patients often have significant medical comorbidities. For MCC patients with a positive SLNB, adjuvant radiation to the at-risk draining nodal basins is a reasonable alternative2.
Negative SLNB
Patients with a negative SLNB are traditionally followed closely with routine examinations of the primary tumor site and draining lymph nodes.
MCC is an exception due to the high rate of regional metastasis that exceeds that of melanoma. MCC patients with a negative SLNB can be considered for prophylactic irradiation to the nodal basins2. The SLN mapping serves as a helpful guide to radiation planning for midline lesions which may drain in multiple directions.
SLNB in cutaneous melanoma
The role of SLNB in cutaneous melanoma has recently shifted from a staging and diagnostic modality to a potentially therapeutic intervention7. Results of the long-awaited MSLT-II have now been published8. This international, multi-institutional prospective trial randomized patients with a positive SLNB to CLND (n=824) versus observation with ultrasound (n=931); 14% of enrolled patients had primary head & neck melanoma. At 3-years’ follow-up the primary outcome measure of melanoma specific survival was identical between the immediate CLND and observation arms (86 +/- 1.3% versus 86 +/- 1.2%; p=0.42). For this reason, melanoma patients with a positive SLN are now being offered observation with serial ultrasound instead of CLND at some institutions. It should be noted that immediate CLND did demonstrate an improved overall disease-free survival (68 +/- 1.7% vs. 63 +/- 1.7%; p=0.05). Specifically, the disease control rate in the regional nodes was 92 +/- 1.0% for the CLND arm versus 77 +/1 1.5% in the observation arm (p<0.001). Ultimately, a candid conversation is required to allow patients to understand the risks and benefits associated with CLND versus observation so that they can make an informed decision.
SLNB Complications
- Normal risks of surgery that include that of general anesthesia, bleeding, scarring, loss of skin sensation, and infection
- Damage to cranial nerves
- Hematoma/seroma/sialocele
- Failure to identify the sentinel node
- Anaphylaxis related to methylene blue (rare)
SLNB in resource constrained settings
Lymphoscintigraphy and nuclear medicine facilities are unavailable in most of the developing world. Hence reliance must be placed on methylene blue dye for SLNB. Methylene blue is cheap, relatively safe, and promising results have been reported in SLN studies in breast cancer when used alone9. Methylene blue dye alone was also used for SLN identification in early oral cancers with good accuracy and sensitivity in a small study by Ramamurthy et al9. However, without lymphoscintigraphy one loses the benefit of doing targeted biopsies through small incisions, as all the nodes in the drainage area must be exposed to inspect them for blue discoloration. Another drawback is the inability to stage the contralateral N0 neck without contralateral wide surgical exposure. Consequently, the value of SLNB using only blue dye, as compared to an elective SND, is questionable.
References
- National Comprehensive Cancer Network Clinical Practice Guidelines in Oncology: Melanoma (Cutaneous). V2.2018. www.nccn.org. March 8, 2018
- National Comprehensive Cancer Network Clinical Practice Guidelines in Oncology: Merkel Cell Carcinoma. V1.2018. www.nccn.org. March 7, 2018
- National Comprehensive Cancer Network Clinical Practice Guidelines in Oncology: Head and Neck Cancers. V1.2018. www.nccn.org. March 18, 2018
- National Comprehensive Cancer Network Clinical Practice Guidelines in Oncology: Squamous Cell Carcinoma. V2.2018. www.nccn.org. March 8, 2018
- Civantos FJ, Stoeckli SJ, Takes RP, Woolgar JA, et al. What is the role of sentinel lymph node biopsy in the management of oral cancer in 2010? Eur Archive Otolaryngol. 2010;367: 839-44
- Morton DL, Thompson JF, Essner Ret al. Validation of the accuracy of intraoperative lymphatic mapping and sentinel lymphadenectomy for earlystage melanoma: a multicenter trial. Multicenter Selective Lymphadenectomy Trial Group. Annals Surg 1999; 230:453-463; discussion 463-55
- Schmalbach CE, Bradford CR. Completion lymph node dissection for sentinel node positive cutaneous head and neck melanoma. Laryngoscope Investig Otolaryngol. 2018 Feb 5;3(1): 43-Faris MB, Thomspon JF, Cochran AJ, et al. Completion dissection or observation for sentinel-node metastasis in melanoma. N Engl J Med 2017;376: 2211-22
- Ramamurthy R, Kottayasamy Seenivasagam R, Shanmugam S, Palanivelu K. A Prospective Study on Sentinel Lymph Node Biopsy in Early Oral Cancers Using Methylene Blue Dye Alone. Indian Journal of Surgical Oncology. 2014;5(3):178-83
Authors
Cecelia E. Schmalbach MD, MS, FACS
Professor of Otolaryngology
Department of Otolaryngology-HNS
Indiana University School of Medicine
Roudebush VA Medical Center
Indianapolis, IN, USA
cschmalb@iu.edu
Carol R. Bradford MD, MS
Executive Vice Dean for Academic Affairs
University of Michigan Medical School
Ann Arbor, Michigan, USA
Cbradfor@med.umich.edu
Editor
Johan Fagan MBChB, FCS (ORL), MMed
Professor and Chairman Division of Otolaryngology
University of Cape Town
Cape Town, South Africa
johannes.fagan@uct.ac.za

The Open Access Atlas of Otolaryngology, Head & Neck Operative Surgery by Johan Fagan (Editor) johannes.fagan@uct.ac.za is licensed under a Creative Commons Attribution - Non-Commercial 3.0 Unported License



