2.10: Ossiculoplasty
- Page ID
- 17704
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)OPEN ACCESS ATLAS OF OTOLARYNGOLOGY, HEAD & NECK OPERATIVE SURGERY
OSSICULOPLASTY
Tashneem Harris, Thomas Linder
Ossiculoplasty may be defined as restoring the hearing mechanism between the tympanic membrane and the oval window by re-establishing a functioning ossicular chain. It is indicated with ossicular discontinuity following erosion, trauma or ossicular fixation due to tympanosclerosis, adhesions etc. Operating on an only hearing ear is an absolute contraindication for ossiculoplasty.
Chronic otitis media may also cause ossicular disruption due to erosion of the ossicular chain. Ugo Fisch classified ossicular defects seen in chronic otitis media in accordance with the expected functional hearing outcomes (Table 1).1
| ABG | Preop status of ossicles and mastoid |
| <10dB |
Malleus: Handle intact Incus: Absent Stapes: Intact |
| <20dB |
Malleus: Handle intact Incus: Absent Stapes: Fixed /no superstructure |
| <30dB |
Open/closed cavity mastoidectomy Malleus: Absent Incus: Absent Stapes: Mobile/fixed/mobile footplate |
Table 1: Anticipated postoperative airbone gap (ABG) according to preoperative status of ossicular chain and type of mastoidectomy (Fisch U, May J. Tympanoplasty, Mastoidectomy and Stapes Surgery)
Patient selection
Patient selection is based on the preoperative audiogram AND the potential to regain serviceable hearing. A patient will benefit if the hearing level of the poorer hearing ear is raised to within 15 dB of the better hearing ear. Second stage ossiculoplasty is usually less invasive and can be done by an endaural approach. One can anticipate the likely course of the ossiculoplasty from otoscopy and audiogram findings.
Methods of ossicular reconstruction
The principal determinants of the method of reconstruction and the expected hearing results are
- Mobility of the stapes footplate
- Integrity of the stapes superstructure
- Presence/absence of a malleus handle
Mobility of the footplate is assessed by placing a few drops of saline in the round window niche. The saline is carefully observed while gently touching the stapes head. Mobility of the footplate is verified when the water moves as the stapes head is touched. If the mobility of the stapes is restricted due to adhesions or granulation tissue, then the tissue may be carefully removed before again assessing stapes mobility using the above technique before proceeding with ossiculoplasty.
Ossiculoplasty using a Partial Ossicular Reconstruction Prosthesis (PORP) is done when the stapes superstructure is intact. Ossiculoplasty with Total Ossicular Reconstruction Prosthesis (TORP) is required when the stapes superstructure is absent, and the footplate is mobile.
The malleus handle is an important clinical audiological prognostic factor for ossiculoplasty.12,13 Anchoring the prosthesis to the manubrium of the malleus provides better stabilization and reduces extrusion. The angle of the prosthesis relative to the stapes is also important in order to achieve good acoustic results; placing the prosthesis at >45° causes loss of acoustic transmission and carries the risk of subluxation onto the promontory resulting in a complete conductive block (A-B gap >60 dB).9 If the manubrium of the malleus is very anterior to the stapes or there is a large distance between malleus and stapes, the prosthesis would lie at >45° angle. In such cases better hearing results are obtained by placing the prosthesis in direct contact with the tympanic membrane with an interposed cartilage graft.
Staging the surgery
In some cases, it may be preferable to do ossiculoplasty as a staged procedure i.e. not at the same time as doing a tympanoplasty or mastoidectomy. The possibility of a 2nd stage operation should always be discussed with the patient at the time of primary surgery. Staging improves the outcomes of ossicular reconstruction for the following reasons:
- Better assessment of the position of the tympanic membrane and eustachian tube function 6-12 months after primary surgery
- Should the mucosa around the oval window have been traumatized, there is a risk of scarring of the oval window if ossiculoplasty is performed in as single stage procedure; staging allows damaged mucosa of the middle ear to first to heal
- With cholesteatoma surgery, to verify that no residual cholesteatoma is present; the 2nd stage (ossiculoplasty) surgery is delayed for about a year after the primary surgery
- It may be preferable to stage ossiculoplasty if only the stapes is present in the setting of a tympanic membrane perforation
If ossiculoplasty is staged and the middle ear mucosa has been traumatized and/or eustachian tube function is poor, then thin silastic sheeting is placed in the protympanum of the middle ear to allow the middle ear mucosa to heal and to reduce adhesions that may form between the middle ear mucosa and the tympanic membrane.
Before undertaking 2nd stage or revision surgery the surgeon needs to know the following:
- What was the status of the ossicular chain at the time of primary surgery?
- Was reconstruction attempted and what was used in the reconstruction?
- Is the chorda tympani nerve intact?
- Was silastic sheeting placed in the ear?
- Was temporalis fascia harvested? If so, then the postauricular incision may need to be extended superiorly to access temporalis fascia. This must be anticipated when draping and preparing the surgical site
Materials used for ossiculoplasty
Autologous ossicle: This is the gold standard of ossicular reconstruction. Whether it can be employed depends on the presence of ossicular remnants. Autologous ossicles are easily shaped to the required size and shape with diamond burrs. Disadvantages include prolonged operating time and the possibility of them harboring microscopic remnants of cholesteatoma. Advantages include low extrusion rates, low cost, biocompatibility and zero risk of transmitting disease. Failure of ossicular reconstruction relate to lateralization, bony fixation, atrophy and displacement.1
Autologous bone graft: Cortical bone grafts may be used for ossicular reconstruction, although it is fairly controversial when one examines the literature. While some studies report the functional results of cortical bone grafts to be as good as with ossicular bones2,3 others quote less favorable outcomes.4,5 The most common causes of deterioration of hearing are atrophy and bony fixation.4,5
Cartilage: The use of cartilage is controversial. Some studies show evidence of cartilage resorption over the long term.4,5 Yet other studies report long term stability with good functional results.6,7 Advantages include versatility, availability, cost, biocompatibility, and low extrusion rates; however operative time may be prolonged as cartilage has to be harvested and molded. Tragal cartilage is most commonly used; conchal and chondral cartilage has been used.
Homologous ossicle: Some years ago, it used to be common practice for otolaryngology departments to have their own “ossicle banks”. Healthy ossicles that were removed from patients for various reasons during middle ear and mastoid surgery or from cadavers were stored in alcohol and used for ossiculoplasty in other patients. Concerns about transferring prions of Creutzfeldt Jacob Disease as well as the advent and ready availability of ossicular prostheses have minimized this practice. However, some countries still permit allogeneic ossicles to be used based on donor selection criteria and special processing methods. Lubbe reviewed the risks associated with homologous ossicles and provides detailed guidelines about preparation and storage of homologous ossicles.8 (Homograft ossicles). “Ossicle banks” may therefore be appropriate in centers that cannot afford middle ear prostheses.
Alloplastic prostheses: A variety of synthetic materials have been used to manufacture prostheses. Advantages of such presculpted prostheses are reduced operating time, and no risks of residual cholesteatoma or transmitting disease. Disadvantages are higher extrusion rates and expense.
Alloplastic grafts may be divided into three categories based on the body’s reaction i.e. bio-inert, biotolerant, and bio-active.9

Figure 1: Titanium prostheses
• Bio-inert materials: The body does not react to bio-inert materials and they are not degraded by inflammatory processes. Bio-inert alloplastic implants include stainless steel, gold, platinum, and titanium (Figure 1).
Titanium is an excellent biocompatible material, is light but strong, and allows for many different prosthetic designs; it has good long-term success rates.10 Cartilage needs to be interposed between the titanium prostheses and the tympanic membrane to prevent extrusion.

Figure 2: Examples of biotolerant prostheses
- Biotolerant materials: Biotolerant materials are initially recognized by the body as foreign. However they soon develop a strong capsule to which endogenous proteins attach themselves; this shields the implant from the immune system. Examples of bio-tolerant materials include Plastipore® (high density polyethylene sponge), and Teflon (polytetrafluorethylene) (Figure 2). Teflon has been in longest use and is most frequently used. Excellent long term audiological results have been reported particularly in stapes surgery.11 Flex H/A and Hapex are both composite allografts, the former consisting of hydroxyapatite and silastic; the latter consists of hydroxyapatite and polyethylene.
- Bio-active materials: Bio-active materials have favorable biocompatibility but high biodegradability. Resorption rates are increased with inflammatory processes.9 Examples of bio-active materials are glass ceramics and hydroxyapatite (HA). The biocompatible nature of HA allows the prosthesis to be placed directly in contact with the tympanic membrane.
Ossiculoplasty for partial or complete absence of incus
When the long process of the incus is eroded, continuity of the ossicular chain may be re-established by interposing autologous incus, head of malleus, a cortical bone chip, cartilage, or a PORP between the stapes head and malleus handle. These techniques are next discussed in greater detail.
Autologous incus interposition
Autologous incus is used whenever possible as it does not cost anything, is situated in the surgical field, and has a low extrusion rate through the tympanic membrane. There are however times when this technique it is unsuitable: the incus may be absent, eroded, or covered by cholesteatoma. Alternative interpositions are also indicated when the incus does not fit well e.g. when the stapes superstructure and/or the malleus handle are absent. Incus interposition is performed at the time of primary surgery other that when the anterior half of the tympanic membrane is absent, or if there is a large perforation. In these two situations it is necessary to stage the procedure. The techniques for open and closed cavities are similar. Where the malleus head has been removed e.g. in an open cavity or with a medialized malleus, incus interposition can still be done without compromising the hearing result. It is vital to preserve the tensor tympani tendon as it stabilizes and prevents anterior migration the malleus handle which is important for a good functional outcome.
Incus interposition: Surgical steps
Point of departure: Endaural approach and inspection of ossicular chain reveals an eroded long process of incus
- The incus is removed by rotating it laterally with a 45° hook
- The Fisch microraspatory is a useful means to assess the size and inclination of the modified incus. The working end of the microraspatory is 2.5 mm in length and usually corresponds to the distance between stapes head and the malleus handle

Figure 3: Incus held by its body
- A small curved clamp is used to hold and stabilize the body of the incus while shaping it with a diamond drill (Figure 3)

Figure 4: Removing long process
- Looking through the microscope, the surgeon removes the long process with a diamond drill (Figure 4)
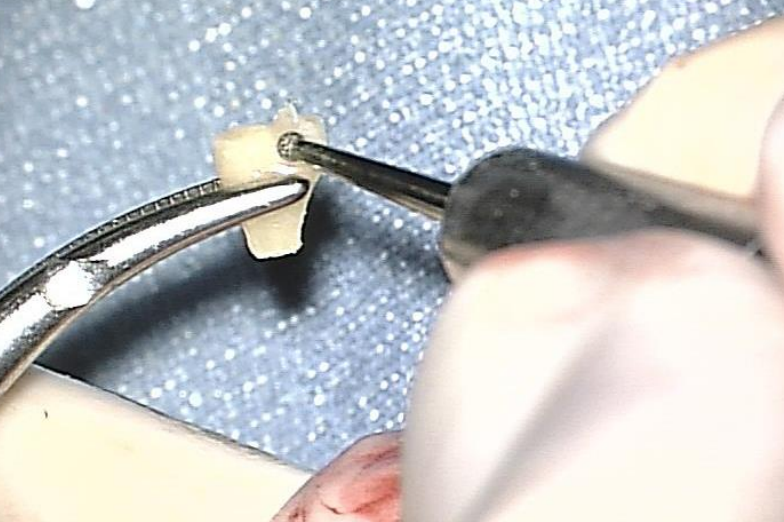
Figure 5: Shaping the articular surface to accommodate malleus handle
- The articular surface of the incus is shaped to accommodate the malleus handle (Figure 5)
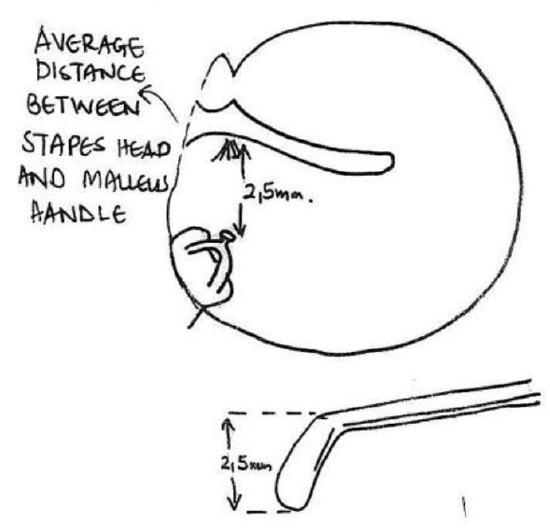
Figure 6: Fisch microraspatory used to determine the distance between malleus and stapes
- The modified incus should fit snugly between the stapes head and malleus handle. A notch is drilled in the body of the incus to accommodate the stapes head using 0.6 mm and 0.8 mm diamond burrs. The required depth of the notch is estimated by using the Fisch microraspatory to measure the distance between the malleus handle and stapes superstructure (Figure 6)
- Use the largest microsuction tube to pick up the prosthesis and place it in the middle ear, lateral to the preserved chorda tympani
- The groove drilled on the articular surface of the modified incus is maneuvered into contact with the malleus handle using a 1.5 mm 45° hook
- While maintaining the incus against the proximal malleus handle just below the lateral process, use the 1.5 mm 45° hook to position the notch drilled in the body of incus over the head of stapes
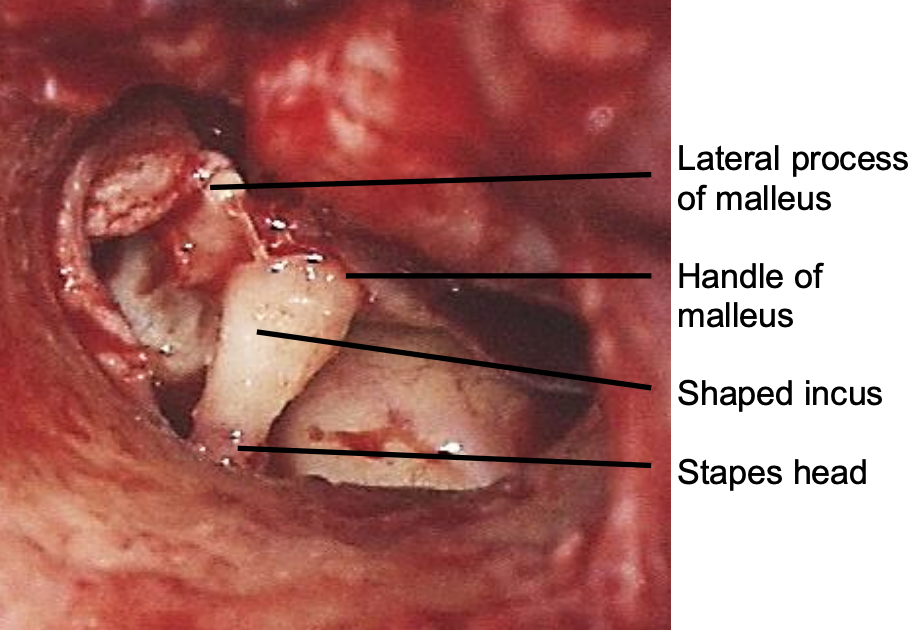
Figure 7: Incus interposition
- Figure 7 shows an autologous incus interposed between malleus handle and stapes head. (Note: malleus head has been removed)
Head of malleus interposition
The head of the malleus may be used when the incus is either missing or has been eroded such that the remaining incus is too small to use. The head of the malleus is amputated with a malleus nipper. Holding the malleus with an artery forceps, it is shaped with a burr to create a notch which fits the malleus handle and a hole on the opposing side to accommodate the stapes head.
Cortical bone interposition
Cortical bone can be obtained from the temporal squama or from the mastoid cortex along the edge of the mastoid cavity. For small defects such as when only the distal part of the long process of the incus has been eroded, a square-shaped bone graft is be harvested. An acetabulum is drilled for the head of the stapes on one surface; on the opposing surface a groove is drilled for the remaining long process of the incus. For larger defects the bone can be sculpted in a shape similar to an incus interposition with a groove for the malleus and an acetabulum drilled for the head of the stapes. To harvest bone from the temporal squama, elevate the temporalis muscle and expose the temporal squama above the temporal line. Using a small diamond drill, only the outer table of the cortex is drilled in the shape of a small square. This can be lifted off the underlying bone using a straight chisel. Bone chips can be also harvested from the mastoid cortex using a straight chisel along the free edge of the mastoid cavity.
Cartilage interposition
Cartilage may be harvested from tragal cartilage (conchal cartilage is thinner and tends to be more curved) and modeled into the shape of an autogenous incus interposition.
PORP interposition
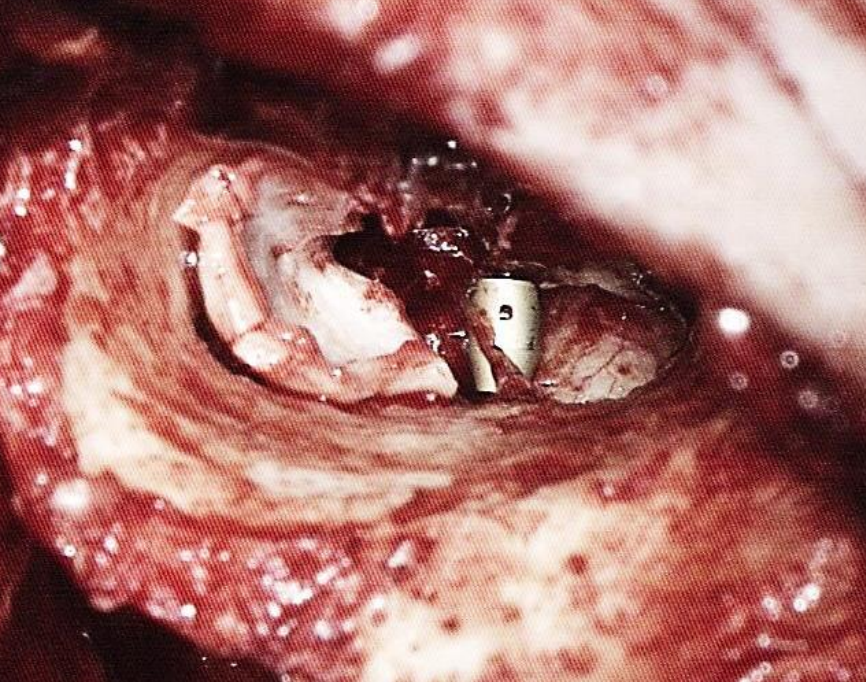
Figure 8: Titanium incus interposition with reconstruction of posterosuperior canal wall with cartilage
Prostheses may be manufactured from hydroxyl-apatite, plastic or titanium (Figures 1, 2, 8).
The PORP is placed on the stapes head. To prevent extrusion, a piece of cartilage is interposed between the prosthesis and tympanic membrane. If only a small distal portion of the lenticular process is eroded, then a prosthesis can be used which fits over the head of the stapes and has an arm which embraces the remaining long process of the incus.14
Ossiculoplasty when only stapes is intact
- Stapes superstructure intact, mobile footplate
- A stapes elevation technique can be performed by placing cartilage over the capitulum of the stapes (Type III tympanoplasty). A notch may be drilled on the surface of the cartilage with a small drill to accommodate the stapes head
- Alternatively, a PORP may be placed over the mobile stapes. If the malleus handle is intact the prosthesis is placed between the stapes and the handle of malleus
- A TORP can be placed between the crura of the intact stapes; this allows for a more stable reconstruction. All alloplastic implants must be covered by cartilage to prevent extrusion
- Stapes superstructure eroded, mobile footplate
- Tragal cartilage may be harvested and trimmed into the shape of a “T“ and interposed between the tympanic membrane and the footplate to create a columella effect
- A TORP may be placed onto the stapes footplate.
Techniques used to stabilize the foot of the prosthesis on the footplate include:
- Perforation of the center of the footplate with a wire spike placed in the foot of the prosthesis1, 15
- Placing gelfoam at the footplate around the prosthesis
- Cutting small wedges of cartilage and placing them on the footplate on either side of the prosthesis
- Fisch devised a cartilage shoe with a central perforation to accommodate the shaft of the Fisch titanium total prosthesis to stabilize the prosthesis on the footplate.1,15 Tragal cartilage is harvested and the perichondrium is removed. A hole is made in the cartilage with a 0.6 mm diamond burr. A disc is cut around the perforation in the cartilage to the dimensions of 1.5x3mm; these dimensions are slightly larger than the oval window. Using a microscope the final sizing is done to fit the oval window niche. The prosthesis shaft is placed through the central perforation of the cartilage disc before placing it in the oval window niche.
- Along a similar concept, Hüttenbrink et al developed a cartilage guide for the oval window niche. An oval 2.5 x 3.5 mm cartilage with a central hole is cut out of a thin (0.2-0.3 mm) cartilage plate with a cartilage punch. The cartilage is placed into the oval niche and the shaft of the prosthesis is guided through the hole of the cartilage shoe onto the center of the foot-plate.16,17
References
- Fisch U, May J. Tympanoplasty, Mastoidectomy and Stapes Surgery. New York: Thieme; 1994
- Romanet P, Duvillard C, Delouane M. Mastoid cortical bone grafts in ossiculoplasty. Ann Otolaryngol Chir Cervicofac. 2000;117(2):105-9
- Mudhol RS, Naragund AI, Shruthi VS. Ossiculoplasty Revisited. Indian J Otolaryngol Head and Neck Surg. doi:10.1007/s12070-011-0472-7
- Hüttenbrink KB. Die Chirurgische Therapie der chronischen Otitis media. III: Mittelohrrekonstruktion. HNO. 1994;42(11):701-18
- Kley W, Draf W. Histologische Untersuchungen über autotransplan-tierte Gehörknöchelchen und Knochenstückchen im Mittelohr beim Menschen. Acta Otolaryngol. 1965;59:593- 603
- Belal A, Linthicum FH, Odnert S. Fate of cartilage autografts for ossiculoplasty: an electron microscopic study. Clin Otolaryngol. 1981;6:231-6
- Quaranta N, Fernandez-Vega Feljoo S, Piazza F, et.al. Closed tympanoplasty in cholesteatoma surgery, long term (10 years) hearing results using cartilage ossiculoplasty. Eur Arch Otorhinolaryngol. 2001;258:20-4
- Lubbe D, Fagan JJ. Revisiting the risks involved in using homograft ossicles in otological surgery. J Laryngol Otol. 2008;122(2):111-5
- Beutner D, Huettenbrink KB. Passive and active middle ear implants. GMS Curr Top Otorhinolaryngol Head Neck Surg.doi:10.3205/cto000061
- Fong JC, Michael P, Raut V. Titanium vs autograft ossiculoplasty. Acta Otolaryngol. 2010 ;130(5):554-8
- Vincent R, Sperling NM, Oates J, Jindal M. Surgical findings and longterm hearing results in 3,050 stapedotomies for primary otosclerosis: a prospective study with the otologyneurotology database. Otol Neurotol. 2006;27(8 Suppl 2):S25-47
- Bared A, Angeli SI. Malleus handle: determinant of success in ossiculoplasty. Am J Otolaryngol. 2010;31(4): 235-40
- De Vos C, Gersdorff M, Gerard JM. Prognostic factors in ossiculoplasty. Otol Neurotol. 2007; 28(1):61-7
- Hornung JA, Brase C, Bozzato A, Zenk J, Iro H. First experience with a new titanium clip stapes prosthesis and a comparison with the earlier model used in stapes surgery. Laryngoscope. 2009;119(12):2421-7
- Fisch U, May J, Linder T, Naumann IC. A new L-shaped titanium prosthesis for total reconstruction of the ossicular chain. Otol Neurotol. 2004;25(6): 891-902. DOI: 10.1097/00129492- 200411000-00007
- Beutner D, Luers JC, Huttenbrink KB. Cartilage "shoe": a new technique for stabilization of titanium total ossicular replacement prosthesis at centre of stapes footplate. J Laryngol Otol. 2008;1-5 DOI:10.1017/S0022215108002545
- Hüttenbrink KB, Zahnert T, Beutner D, Hofmann G. Der Knorpelschuh zur Stabilisierung einer Columella-Prothese auf der Fußplatte. Laryngo-RhinoOtol. 2004;83(7):450-6. DOI: 10.1055/ s-2004-814447
Author
Tashneem Harris MBChB, FCORL, MMed (Otol), Fisch Instrument Microsurgical Fellow
ENT Specialist
Division of Otolaryngology
University of Cape Town
Cape Town, South Africa
harristasneem@yahoo.com
Senior Author
Prof. Thomas Linder, M.D.
Chairman and Head of Department of Otorhinolaryngology, Head, Neck and Facial Plastic Surgery
Lucerne Canton Hospital, Switzerland
thomas.linder@ksl.ch
Editor
Johan Fagan MBChB, FCS(ORL), MMed
Professor and Chairman
Division of Otolaryngology
University of Cape Town
Cape Town, South Africa
johannes.fagan@uct.ac.za


