2.11: Myringoplasty and Tympanoplasty
- Page ID
- 17706
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)OPEN ACCESS ATLAS OF OTOLARYNGOLOGY, HEAD & NECK OPERATIVE SURGERY
MYRINGOPLASTY & TYMPANOPLASTY
Tashneem Harris & Thomas Linder
The goals myringoplasty and tympanoplasty are to achieve a dry, self-cleansing ear while preserving or restoring hearing.
Myringoplasty refers to grafting of the tympanic membrane without inspection of the ossicular chain.
Tympanoplasty entails grafting of the tympanic membrane with inspection of ossicular chain with/without reconstruction of the middle ear hearing mechanism.
Ossiculoplasty is reconstruction of the hearing mechanism using either an autologous graft or prosthesis.
Meatoplasty involves enlargement of the lateral cartilaginous portion of the external auditory canal. A narrow entrance to the ear canal within the cartilaginous portion of the canal prevents proper ventilation and self-cleaning of the ear canal, and may compromise hearing aid fitting.
Canalplasty is partial or total widening of the bony portion of the external ear canal. In order to visualize the tympanic annulus, particularly in anterior or subtotal perforations, canalplasty is essential and may be an integral part of myringoplasty or tympanoplasty.
Preoperative assessment
Otomicroscopy: Both the size and site of the perforation determine the surgical approach
- Size of perforation: Is the perforation limited or subtotal? Microperforations may have a higher failure rate than larger perforations
- Site of perforation: Does the perforation extend far into the anterosuperior quadrant? Does it only involve the posterior quadrant?
- What is the status of the middle ear mucosa? Is it normal or granular/ polypoidal? Is there airflow through the perforation with a Valsalva maneuver?
- Is the handle of the malleus medialized? This may necessitate an ossiculoplasty even in the presence of an intact ossicular chain
- Is an ossiculoplasty required?
- Does the ossiculoplasty need to be staged?
- What is the status of the contralateral ear?
Audiometry: This should have been done recently i.e. within the preceding 3 months. Correlate the size of the perforation with the audiogram, particularly the air-bone gap. Lerut, Pfammater & Linder investigated the correlation between air-bone gap and perforation size. There was a strong correlation between air-bone gap and increasing perforation size; however, the location (anterior/posterior) had no impact on hearing. The greatest changes in airbone gap were at 0.5 and 4 kHz, and the smallest changes at 2 kHz. The audiograms thus revealed a consistent “V”-shaped pattern with the turning point at 2 kHz. This can be explained by the fact that 2 kHz is the resonance frequency of the middle ear; thus, hearing is better preserved at this frequency. The clinical significance is that one can predict the expected air-bone gap by looking at the size of the perforation. If the audiogram does not correspond with the expected findings, then additional middle ear pathology must be expected. If the air-bone gap is greater than 30 dB, then an ossiculoplasty may be required.
Tympanometry: Tympanometry provides additional information regarding the patient’s middle ear function; it can also be used to assess eustachian tube dysfunction (see below). The curve is always flat in the presence of a tympanic membrane perforation, but the volume measurements are of interest.
Eustachian tube function: Successful myringoplasty depends on ventilation of the middle ear and mastoid which in turn affects the final position of the reconstructed tympanic membrane.
- CT scan: The best assessment of eustachian tube function is a CT scan of the mastoid; favorable findings are a well-aerated middle ear and a well pneumatized mastoid
- Tympanometric volume: This is a good indicator of eustachian tube function and ventilation of the middle ear in the presence of a tympanic membrane perforation when CT scanning is not available. As a general rule, values for ear canal volume in the presence of an intact tympanic membrane should be approximately 1.5-2.0 mL (adults). With a perforation of the tympanic membrane, the ear canal volume measurement should be high, because the instrument will measure the volume of the entire middle ear space and mastoid in addition to the volume of the ear canal (4-5 mL in adults). If the volume is less than this (e.g. 2.5-3 mL) then ventilation of the middle ear cleft and mastoid must be poor.
- Valsalva maneuver: Absence of a positive Valsalva test i.e. absence of airflow through the perforation on performing a Valsalva maneuver, identifies patients at risk of poorer outcome
Surgical Approaches
Optimal graft take depends on securing the graft; this in turn depends on having sufficient surgical exposure in order to stabilize the graft. Three different approaches may be used i.e. transcanal, endaural or retroauricular.
Transcanal approach: The ear operation is performed through an ear speculum placed in the external ear canal. Because exposure is one of the limiting factors of the transcanal approach, its use is limited to repairing traumatic perforations or in cases where there is a wide ear canal with a posterior perforation. The ear canal must be wide enough, and one should be able to visualize the entire margins of the perforation; most often this would apply to posterior perforations. The anterior margin of an anterior perforation may be obscured by an overhanging canal wall.
Endaural and retroauricular approaches: These afford better exposure. These two approaches will next be discussed in more detail.
Endaural Approach
The endaural approach entails making an incision between the tragus and helix; the entrance to the ear canal is then stretched open with endaural retractors. It is a good approach to use for posterior perforations. While the view is better than with a transcanal approach, it is not suited to anterior perforations.
Surgical Steps
- Infiltrate the site of the endaural skin incision as well as the ear canal in 4 quadrants with local anesthetic (lidocaine 1% and adrenaline diluted to 1:200 000), using a nasal speculum to expose the ear canal
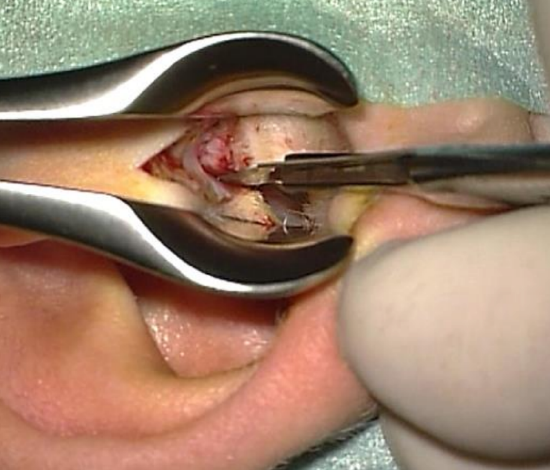
Figure 1: Endaural incision at 12 o’clock in right ear canal
- A skin incision is made in the bony external canal with a #15 blade from the 12 o’clock position, spiraling upwards between the cartilages of the helix of the pinna and tragus (Figure 1). The incision is 1.5 cm in length and extends down to the bone
- Hemostasis is achieved using bipolar coagulation
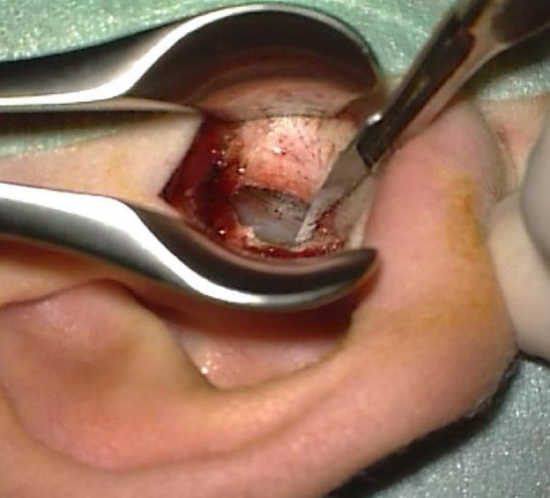
Figure 2: Posterior canal incision that meets endaural incision
- Using a #11 blade, a skin incision is made in the posterior aspect of the bony external canal parallel to the annulus, starting at 8 o’clock (for right ear) and ascending in a spiral fashion to meet the endaural incision at 12 o’clock (Figure 2)
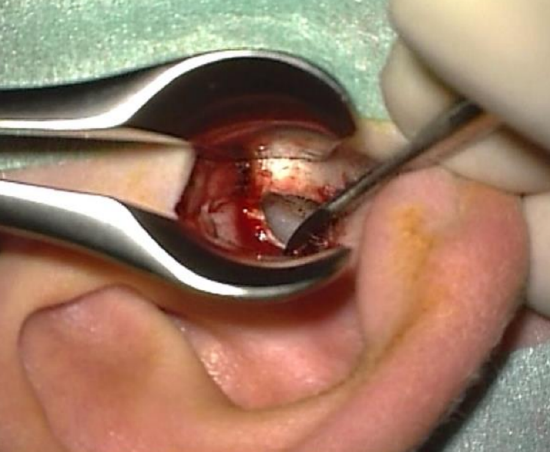
Figure 3: Reflecting canal skin and soft tissue laterally
- A Key raspatory is used to reflect the canal skin and soft tissue laterally and away from the edges of the tympanomeatal flap (Figure 3)
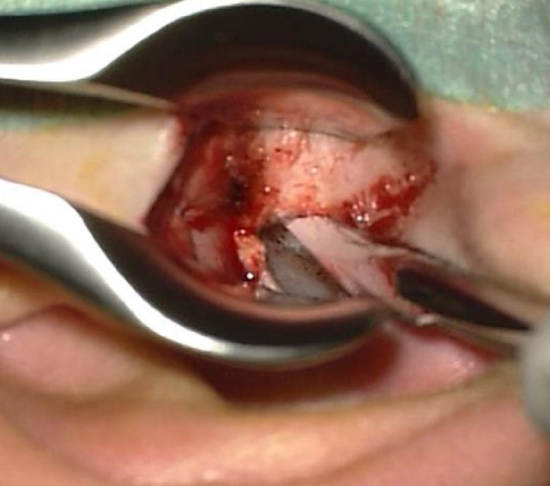
Figure 4: Anterior incision starting at 2 o’clock
- An incision is made anteriorly in the bony canal skin, parallel to the annulus and remaining medial to the cartilage of the ear canal. It starts at 2 o’clock and meets the endaural skin incision at 12 o’clock (Figure 4)
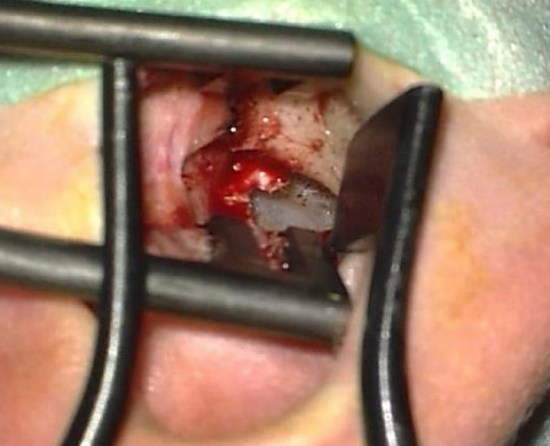
Figure 5: Placement of retractors
- Two endaural retractors are next placed in the ear canal to improve exposure (Figure 5)
- The edges of the perforation are freshened using a sickle knife before elevating the tympanomeatal flap
- A tympanomeatal flap is elevated using a Fisch microraspatory and adrenaline gauze
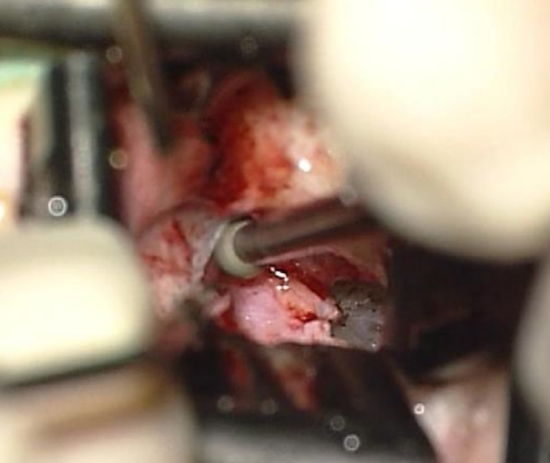
Figure 6: Tympanosquamous suture drilled away
- If a prominent tympanosquamous suture prevents good exposure, then it needs to be drilled away using a 2.7 mm diamond drill (Figure 6). To avoid bone dust from entering the ear, place gelfoam soaked in Ringer’s lactate into the perforation. Never leave gauze or cotton in the surgical field that can be caught up in the drill bit.
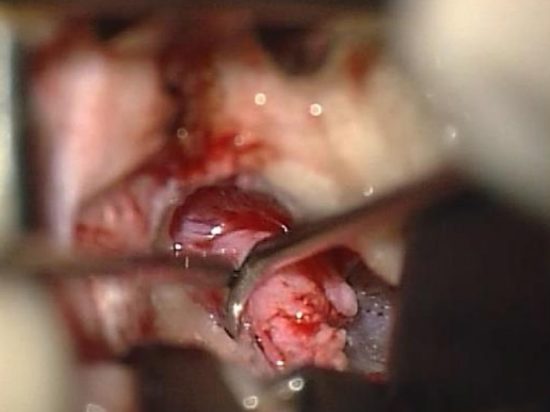
Figure 7: Elevating the tympanomeatal flap
- The middle ear is entered by elevating the tympanomeatal flap at the level of the posterior tympanic spine (Figure 7)
- The annulus starts at this level and can be easily dissected free of its sulcus using the microraspatory as a shovel
- Identify and preserve the chorda tympani immediately beneath the tympanic spine at this level
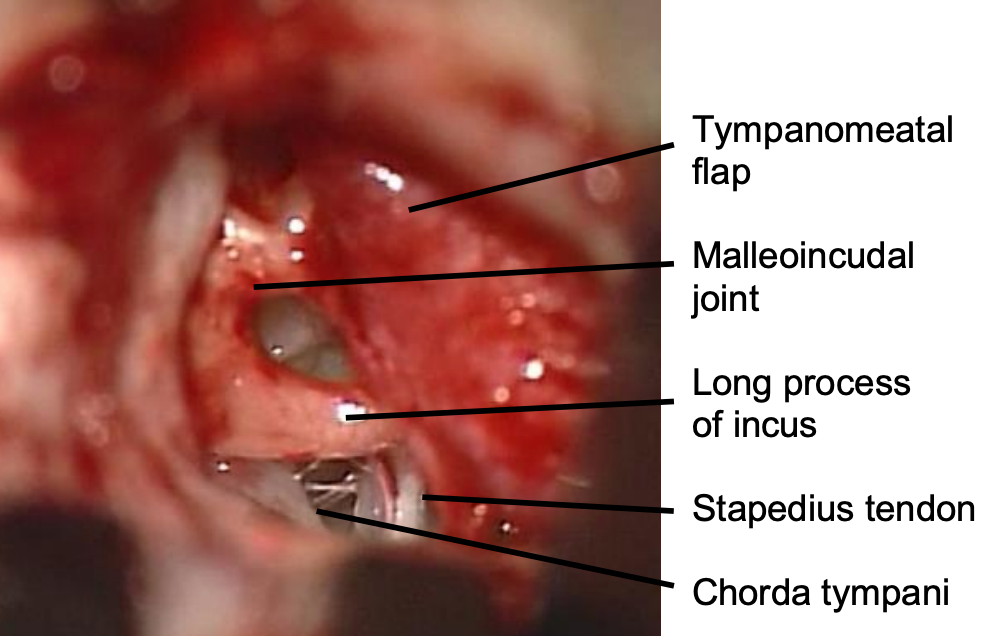
Figure 8: Middle ear structures after curetting posterior tympanic spine
- In order to assess ossicular chain mobility, use the small end of a curette to remove the posterior tympanic spine and bone of the posterior canal wall until the incus, incudostapedial joint, lateral process of the malleus and round window are visible (Figure 8)
- Use a 1.5 mm, 45° hook to palpate the malleus and incus. If the ossicular chain is intact, then proceed to graft the perforation
- Tragal perichondrium is generally harvested via the endaural incision and placed as an underlay graft beneath the edges of the perforation. Posteriorly and superiorly it is laid onto the bony canal lateral to the posterior tympanic sulcus. Temporalis fascia may also be harvested via the endaural incision or through a separate postauricular incision and used as a graft
- The tympanomeatal flap is returned to its original position and gelfoam pledgets are placed over the graft to secure it over the posterior tympanic sulcus
- The endaural skin incision is closed with 3/0 Nylon sutures
Retroauricular Approach
A retroauricular incision is made close to the hairline with soft tissue and pinna reflected anteriorly. It is favored for anterior perforations. It allows one to do a circumferential canalplasty in cases where there is a significant anterior bony overhang.
Surgical Steps
- Using local anesthetic (lidocaine 1% and adrenaline diluted to 1:200 000), infiltrate the post-auricular sulcus. Then advance the needle and infiltrate the tissues anteroinferiorly and anterosuperiorly
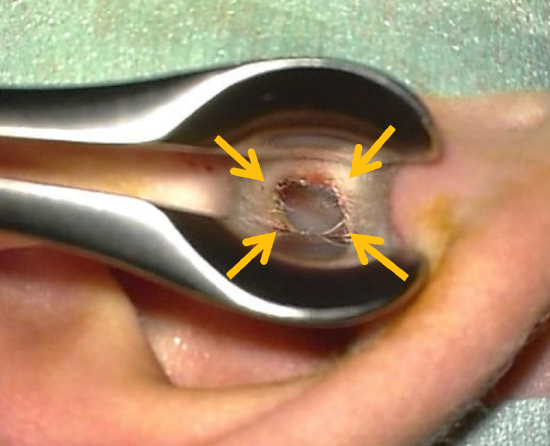
Figure 9: Points of injecting ear canal
- Using a Lempert’s speculum to visualize the bony-cartilaginous junction of the ear canal, inject the 4 quadrants of the skin of the ear canal (Figure 9)
- A postauricular incision is made about 2 cm behind the retroauricular sulcus extending from the upper border of the pinna to the level of the mastoid tip. Light crosshatchings are made with a scalpel before incising the skin, to facilitate aligning the skin when closing the wound
- Use a large rake retractor to reflect the pinna anteriorly with the left hand while developing a tissue plane anteriorly toward the external ear canal, using a scalpel with a #10 blade
- The scrub nurse or assistant uses a large suction tube to clear excess blood so that no time is lost trying to achieve hemostasis while elevating the skin
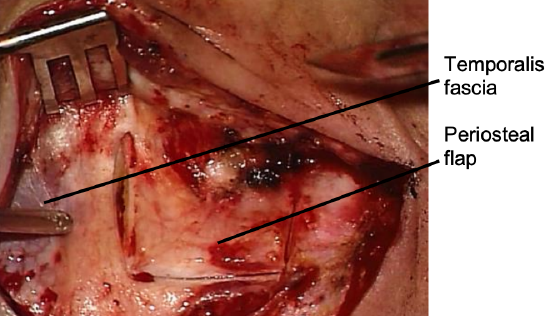
Figure 10: Temporalis fascia and retroauricular periosteal flap
- As the flap is developed the posterior auricular muscle is encountered. This is transected in order to get into the correct surgical plane. Superiorly, the temporalis fascia comes into view (Figure 10)
- A retroauricular periosteal flap is now developed which is anteriorly based. The vertical incision is made approximately 1.5 cm from the ear canal (Figure 10). Using a #10 blade, the superior incision is extended anteriorly along the linea temporalis up to 12 0’ clock relative to the bony ear canal. The inferior incision is extended to the inferior border of the ear canal
- The periosteal flap is elevated from the bone with a mastoid raspatory until both the Spine of Henlé and the bony external canal up to the 12 o’clock position, are exposed
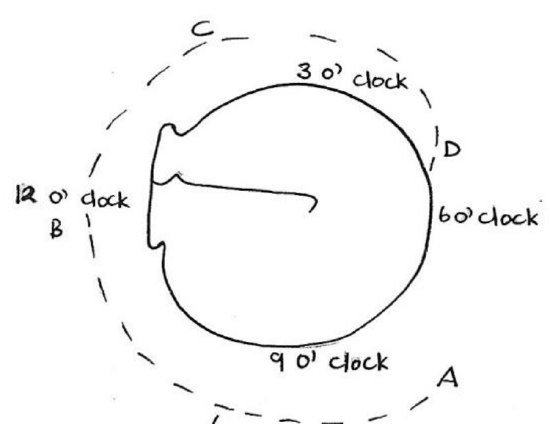
Figure 11: Incisions for spiral flap technique
The next step is to raise a meatal skin flap. The authors favor a spiral flap technique, which should be practiced in the temporal bone laboratory and will next be described (Figure 11).
- Elevate the periosteal flap up to about 2 mm deep to the lateral edge of the bony external ear canal
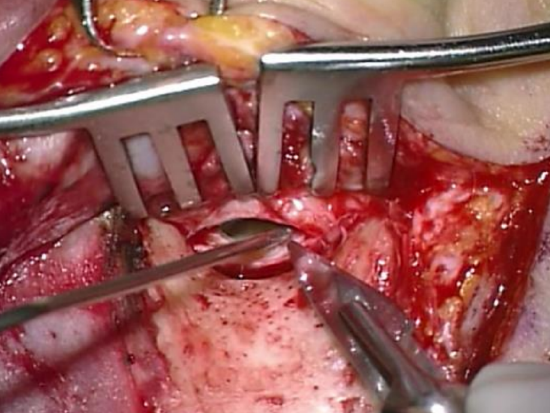
Figure 12: Incising posterior ear canal
- Using a #11 blade, enter the ear canal via a transverse incision in the posterior ear canal skin at about 8 o’clock (right ear). The lumen of the ear canal is then visible through the incision (Figure 12)
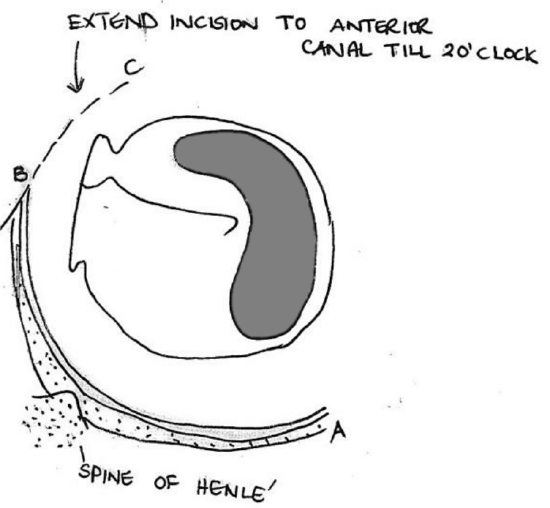
Figure 13: Posterior incision extended as spiral incision to anterior canal wall
- Cut with the scalpel in a cephalad direction and extend the incision superiorly up to 12 o’clock. It is important that the blade remains on the bone. The first incision now extends from A - B (Figures 11, 13)
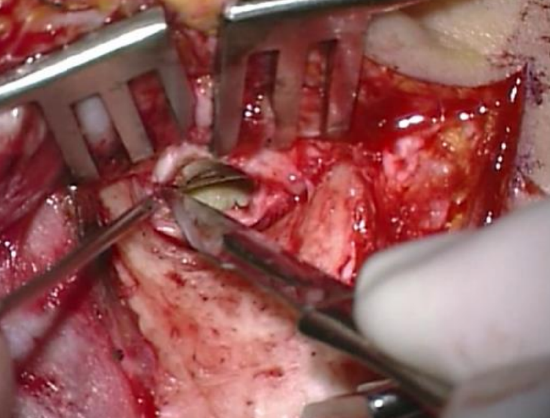
Figure 14: Incising anterior ear canal
- An incision is now made from C - B in the skin of the anterior wall of the ear canal starting at 2 o’clock and extended superiorly to meet the previous incision at 12 o’clock (Figure 13, 14). This incision runs lateral to the tympanosquamous suture line and also has to remain on bone and medial to tragal cartilage; if the incision is placed too laterally then the tragal cartilage will be injured
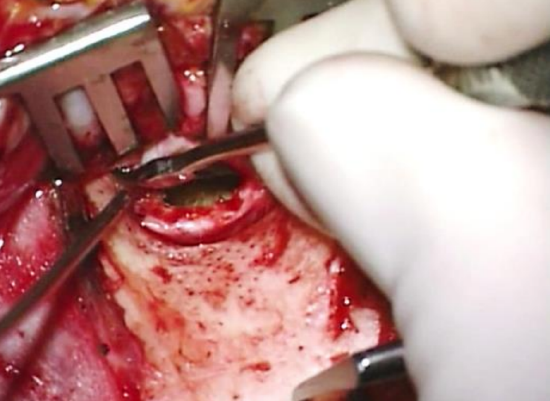
Figure 15: Elevating skin from bone
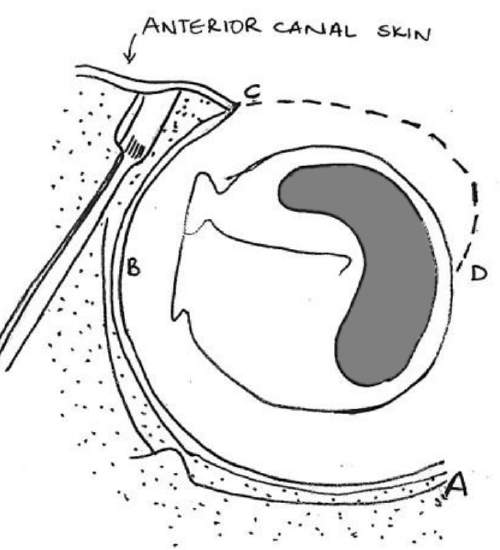
Figure 16: Completing incision in anterior canal wall
- Use a Key raspatory to elevate the lateral canal skin from the underlying bone (Figure 15, 16)
- Attach a sharp towel clip to the periosteal flap at the level of the canal to reflect the pinna and soft tissue forward
- Two self-retaining retractors are placed superiorly and inferiorly and used to reflect the soft tissue and improve exposure. In children, one retractor is sufficient
- The meatal skin is now further incised anteroinferiorly from D to C, starting close to the tympanic membrane at about 5 o’clock (Figure 16). The incision spirals laterally and superiorly along the anterior canal wall to meet the earlier incision made in the anterior canal at 2 o’clock (right ear)
- A common mistake when doing this flap for the first time is not incising the skin fully onto the bone and this causes the flap to tear when elevating it with the microraspatory; it is therefore important that the blade stays hard on bone when making the canal incisions

Figure 17: Fisch microraspatory used to elevate meatal skin off bone
- The meatal skin flap is circumferentially elevated from the underlying bone with a Fisch microraspatory (Figure 17)
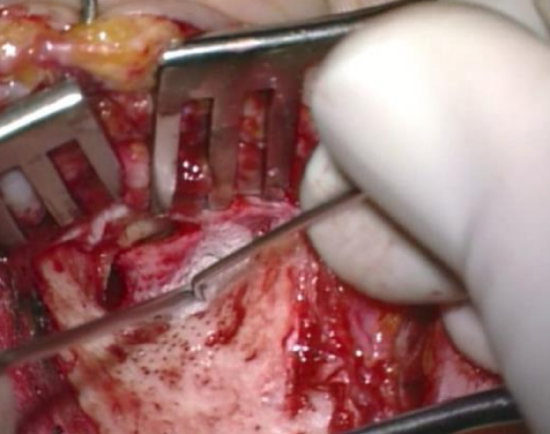
Figure 18: Elevating meatal skin
- To keep this dissection blood-free and to avoid injuring the skin flap, adrenaline-soaked gauze is placed between the Fisch microraspatory and the meatal skin. Suction is never directly applied to the meatal skin flap; rather the excess blood is suctioned through the adrenaline-soaked gauze. The blade of the Fisch microraspatory stays vertical to the bone and the shoulder of the instrument is used to push against the adrenaline gauze which then atraumatically elevates the very fragile meatal skin (Figures 17, 18)
- The meatal skin is elevated until the posterosuperior margin of the tympanic membrane and the anteroinferior overhang of bone are exposed
- At the tympanosquamous suture line (located posterosuperiorly) the skin flap is very tightly bound to the underlying bone and may be dissected free using the following technique: Using the raspatory like a shovel, free the skin flap posterior to the suture. Then elevate the flap anterior to the suture.
- After freeing the flap posteriorly and then anteriorly, there may still be a remaining bridge of soft tissue connections to the suture which is cut with Bellucci scissors
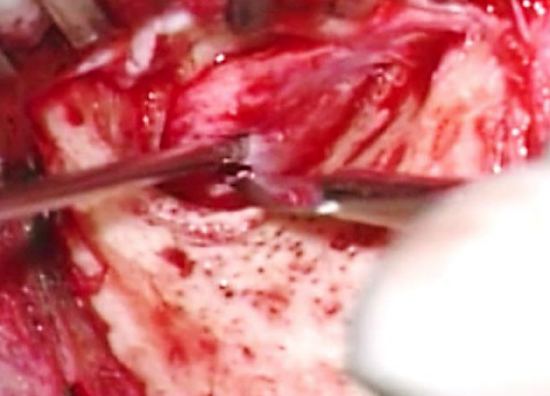
Figure 19: Incising posterior limb of meatal skin flap 2 mm lateral to annulus
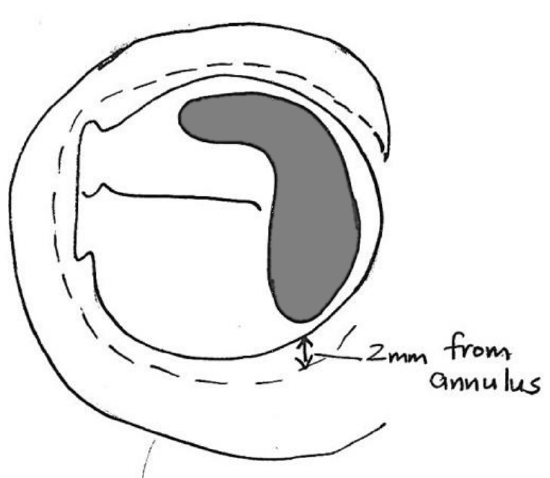
Figure 20: Incise posterior limb of meatal skin flap 2 mm lateral to and parallel to the annulus
- Using a #11 blade incise the posterior limb of the meatal skin flap 2 mm lateral and parallel to the annulus (Figures 19, 20)
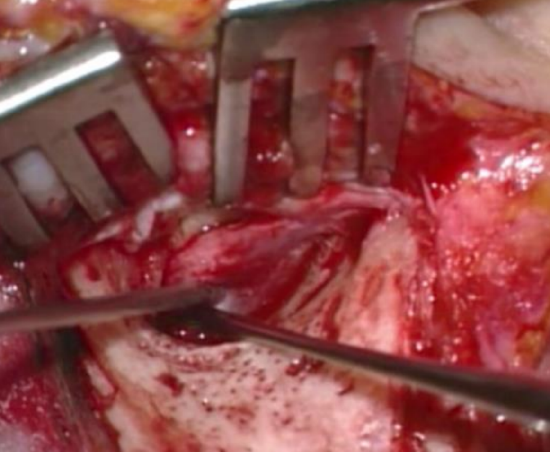
Figure 21: Extending incision anteriorly, remaining 2 mm lateral to and parallel to annulus
- Bellucci scissors are then used to continue incising the meatal skin flap 2 mm lateral to and parallel to the annulus until the anterior part of the flap is reached (Figures 20, 21)
- Transect the remaining meatal skin to 6 o’clock, 2 mm lateral to the annulus, using an Iowa raspatory or a round knife (Figure 21)
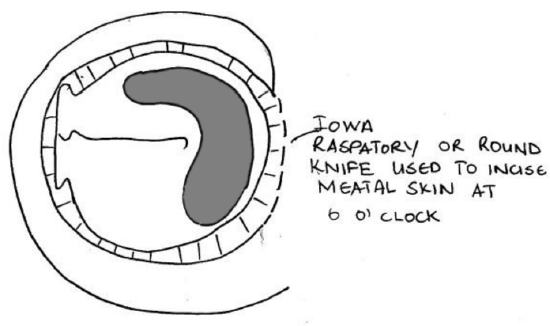
Figure 22: Transect remaining meatal skin to 6 o’clock, 2 mm lateral to the annulus
- Using the larger (Iowa) raspatory the meatal skin lateral to this circumferential incision is dissected free from the bony canal. Using a bigger instrument avoid injuring the inferiorly based pedicle (Figure 22)
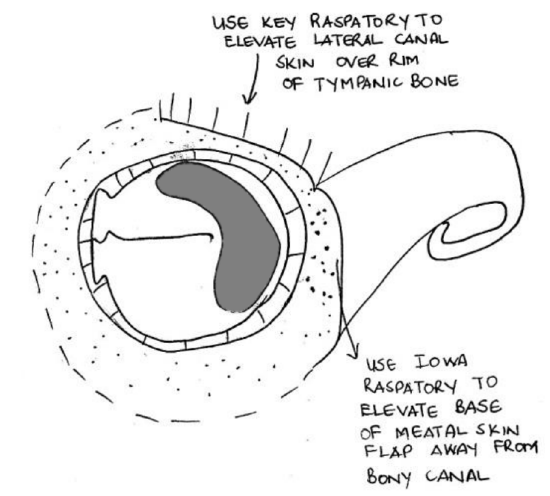
Figure 23: Flap pedicled inferiorly and elevated from canal wall
- Elevate the lateral aspect of the anterior meatal skin flap with a Key raspatory and using two hands, advance the raspatory over the lateral rim of the tympanic bone (Figure 23)
Figure 24: Soft tissue is elevated out of the bony external canal
- Once the lateral edge of the tympanic bone is reached, keep the tip of the instrument in contact with the bone and underlying tissue but swing the handle of the Key raspatory anteriorly so that the soft tissue is elevated out of the bony external canal, leaving it pedicled inferiorly (Figures 23, 24)
Figure 25: Ribbon passed through teeth of self-retaining retractor
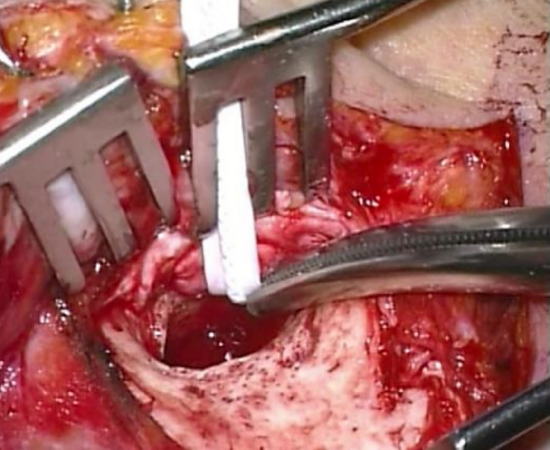
Figure 26: Ribbon passed around meatal spiral flap
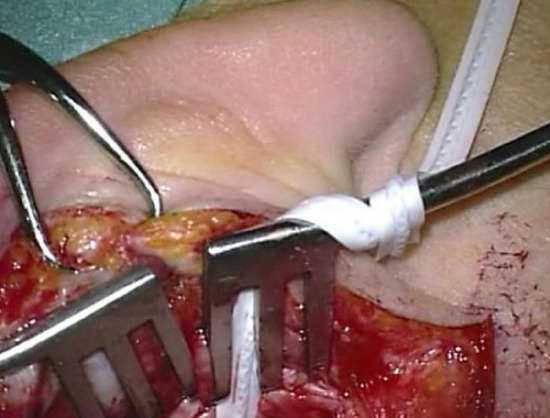
Figure 27: Ribbon secured to selfretaining retractor
- The spiral of elevated meatal skin is kept out of the surgical field by using the aluminum strip or ribbon of the surgeon’s mask which has been sterilized. First, the ribbon is passed through the teeth of the self-retaining retractor (Figure 25). Then artery forceps are used to place it against or pass it around the meatal skin flap (Figure 26). The ribbon is then folded back over the top of the retractor (Figure 27)
Canalplasty
It is imperative to have an adequate view of the annulus as well as all the edges of the perforation to do an adequate myringoplasty or tympanoplasty operation. With perforations only involving the posterior quadrant where the entire margin of the perforation is visible, performing a canalplasty is unnecessary. However, there is often a bony overhang anteriorly and/or posteriorly which restricts the surgeon’s view. If the perforation extends to involve the anterior quadrant then it is necessary to do a canalplasty.
Surgical steps
- Standard retroauricular approach
- Elevate meatal spiral flap
- If there is a tympanic membrane perforation, gelfoam soaked in Ringer’s Lactate is placed over the perforation to avoid bone dust entering the middle ear
- Starting with a 2.7 mm rough diamond burr, enlarge the ear canal by drilling away any bony overhang
- Commence drilling posteriorly, then move inferiorly and finally drill the anterior canal wall
- The technique of skeletonization is important. With the correct technique the bluish color of the temporomandibular joint (TMJ) will show through the irrigation; this should alert one to stop drilling before the joint is entered
- It is important to check that burr sizes are correct by first placing a new burr in the canal before using it
- Always drill under direct vision and never behind overhanging edges of bone. In this way one avoids opening mastoid air cells or injuring the facial nerve posteriorly and TMJ anteriorly
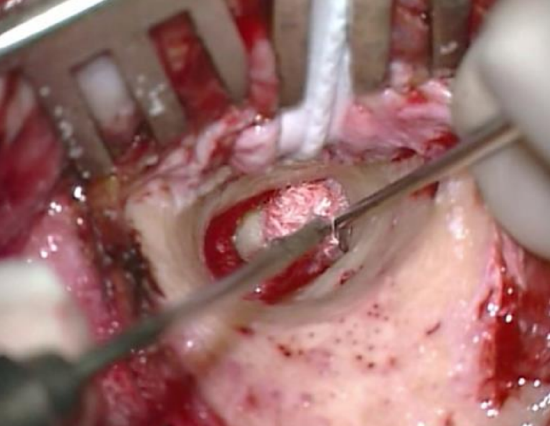
Figure 28: Microraspatory used to elevate meatal skin using adrenaline-soaked gauze
- Use the Fisch microraspatory (with adrenaline gauze) to elevate the meatal skin before drilling away bony overhangs (Figure 28)
- When drilling close to meatal skin, a diamond burr is used so that the meatal skin is not injured by the burr
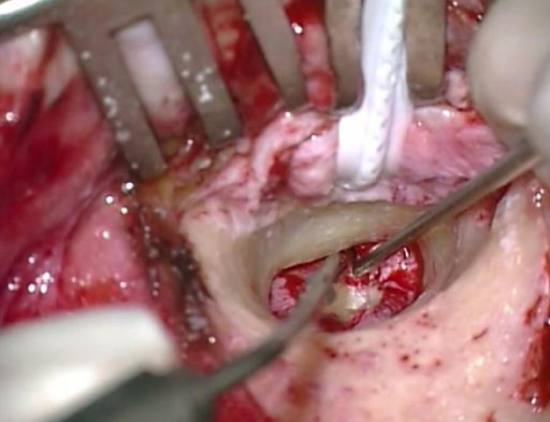
Figure 29: Tip of microraspatory used to determine how much bone needs to be removed lateral to annulus
- The microraspatory is held perpendicularly to the bone at the level of the annulus; the tip of the microraspatory is not visible because of the anterior bony overhang. Using this technique, one can determine how much bone needs to be removed before reaching the annulus (Figure 29)
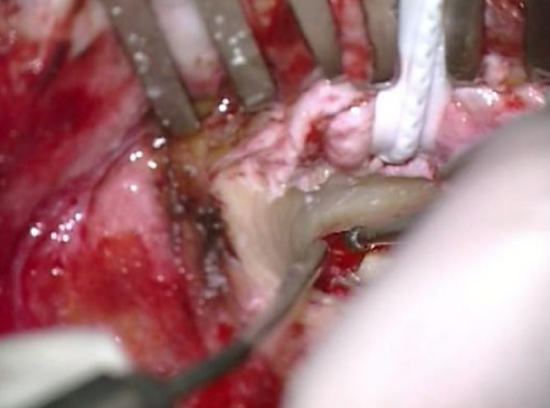
Figure 30: Bony overhang drilled away using a small diamond burr
- This bony overhang is then drilled away, using a small diamond burr (Figure 30)
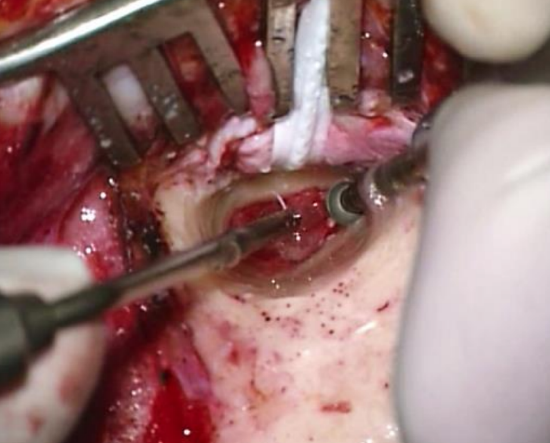
Figure 31: Inferior trough technique
- The inferior trough technique allows the surgeon to gauge the depth of the annulus: using a diamond burr, drill at 6 o’clock from medially to laterally to create a groove. Continue to drill until the white color of the annulus becomes visible at the level of the sulcus (Figure 31)
- Now continue to expand the drilling from the groove outwards and circumferentially
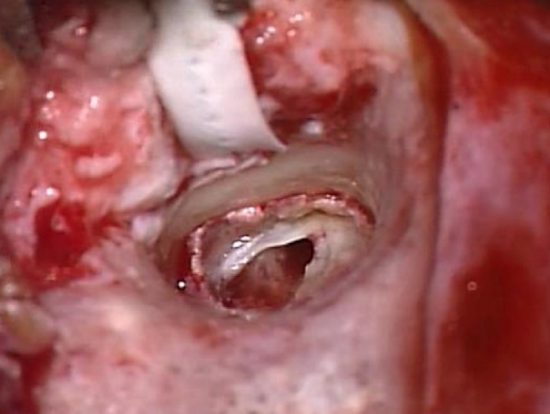
Figure 32: Completed canalplasty with entire annulus visible and no bony overhang
- Once the canalplasty has been completed the entire annulus should be visible with one view of the microscope and no bony overhang should remain (Figure 32)
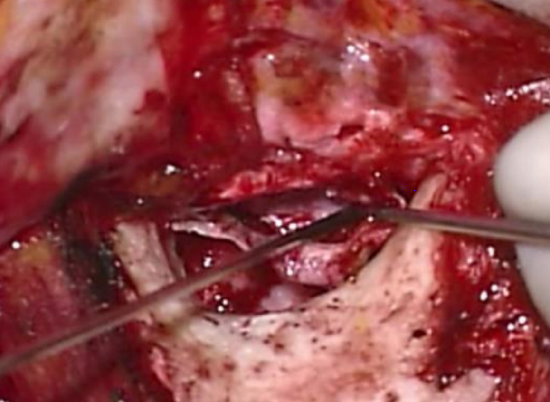
Figure 33: Meatal skin flap being replaced
- Following grafting of the tympanic membrane perforation (see later) the meatal skin flap is replaced (Figure 33)
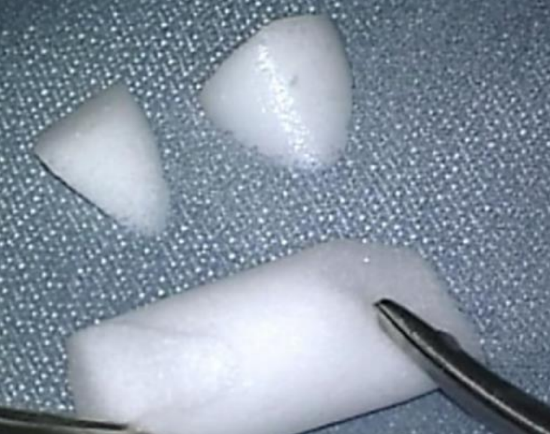
Figure 34: Ivalon®
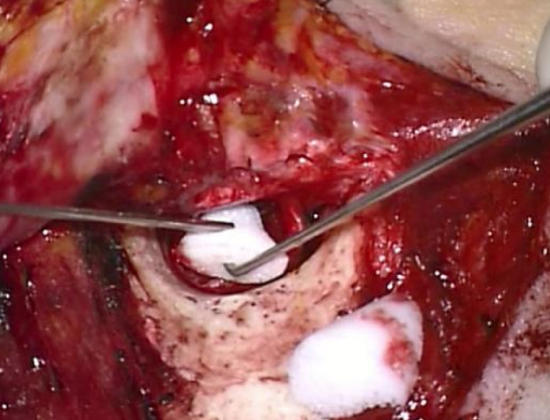
Figure 35: Ivalon® used to secure the meatal skin
- The meatal skin is secured with gelfoam pledgets placed medially over the graft. Two pieces are cut from an Ivalon® ear wick and inserted laterally into the ear canal over the meatal flap (Figures 34, 35). Ivalon® has a smooth surface which is placed on the outside (facing the meatal skin) to allow for atraumatic removal one week postoperatively
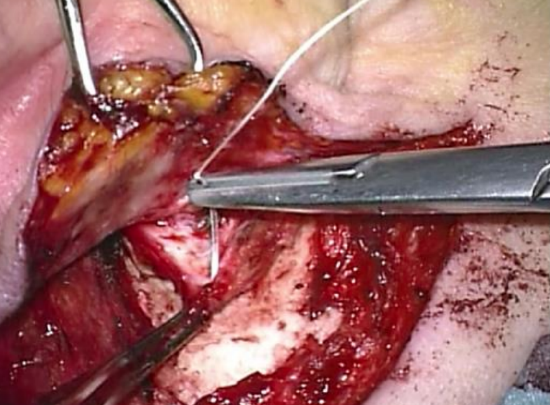
Figure 36: Securing the retroauricular periosteal flap
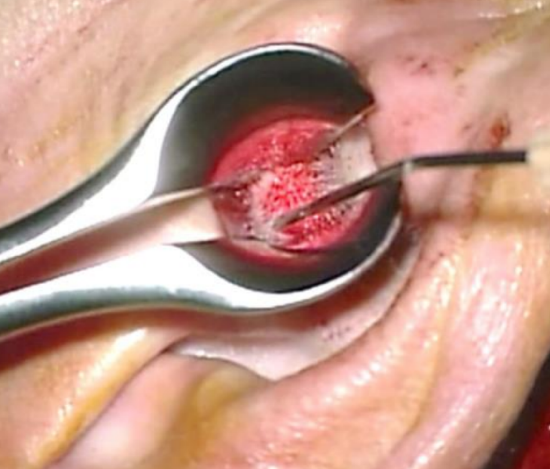
Figure 37: Checking correct placement of meatal skin
- The postauricular periosteal flap is replaced and secured with 3/0 Vicryl sutures (Figure 36)
- Ensure that the skin of the meatal flap is applied to the bony canal and extends lateral to the Ivalon® ear wick (Figure 37)
Antrotomy
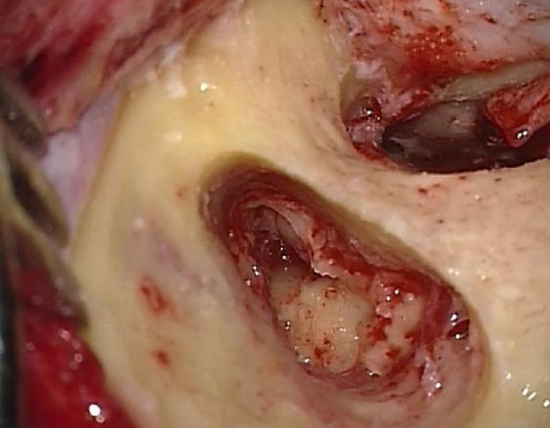
Figure 38: Antrotomy defect
An antrotomy is performed in conjunction with a myringo- or tympanoplasty when eustachian tube function is questionable or in the presence of polypoidal middle ear mucosa obstructing the epitympanum (Figure 38).
The detailed surgical steps for antrotomy are presented in the chapter on Mastoidectomy and epitympanectomy
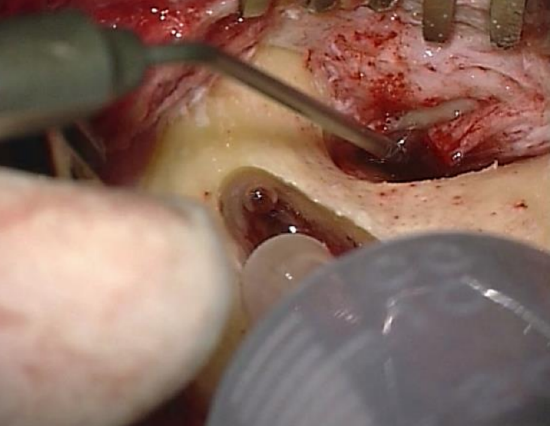
Figure 39: Antrum irrigated with Ringer’s solution
Epitympanic patency is determined by means of the water test: Ringer’s solution is irrigated into the antrum to test whether there is free communication between the antrum and the middle ear (Figure 39)
- If the test is positive, there is no need to further explore the epitympanum
- If the test is negative, then one needs to explore the epitympanum
- If abnormal mucosa is removed from the epitympanum but the ossicles are left intact, this is defined as an epitympanotomy.
- If malleus head and incus need to be removed to re-establish patency, this is referred to as an epitympanectomy
- Following the procedure, a groove is drilled posteriorly into the mastoid bone to accommodate a transmastoid drain
- A separate stab incision is made posterior to the retroauricular skin incision and the drain is fixed to the skin using a silk suture
- The patient may perform Valsalva maneuvers from the 2nd day following surgery
- The mastoid drain is usually removed after 2-4 days
Grafting tympanic membrane perforations
Choice of graft material
- Temporalis fascia is widely used to reconstruct the tympanic membrane. It is easily accessible and long-term results are comparable to that of cartilage tympanoplasty. An advantage of temporalis fascia is that recurrent or residual cholesteatoma can easily be identified behind the reconstructed tympanic membrane
- Tragal perichondrium
- Cartilage is preferred in certain instances due to its resilience: it may be used to reinforce an atrophic tympanic membrane; it is used in addition to fascia in a closed mastoidoepitympanectomy to reconstruct the posterior canal wall; and certain middle ear prostheses require reinforcement of the overlying tympanic membrane to prevent extrusion through tympanic membrane
Harvesting temporalis fascia
Temporalis fascia consists of a superficial and a deep layer. The deep layer is used for grafting because it is thicker and more resilient. The fascia is easily harvested from the retro-auricular region if a retroauricular approach has been used via the same incision. It is preferable to harvest the fascia at the end of the procedure once the surgeon knows how much fascia is required.
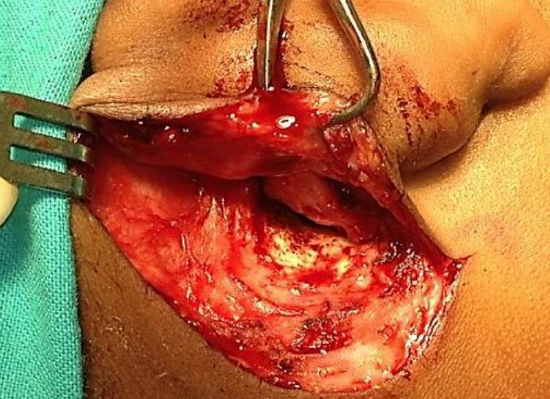
Figure 40: Exposing temporalis fascia
- An assistant exposes the fascia by superiorly retracting skin and soft tissue with a rake retractor (Figure 40)
- The superficial layer of fascia is divided and separated from the deep layer with tympanoplasty scissors
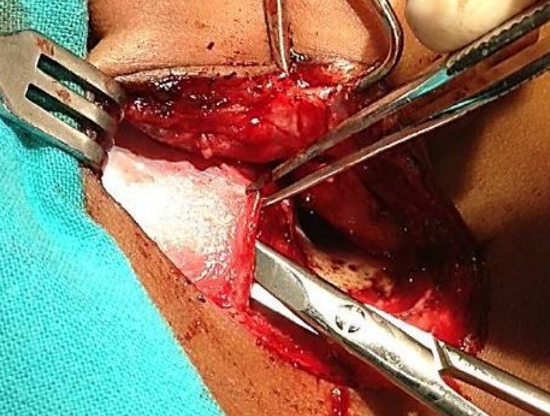
Figure 41: Undermining deeper layer of temporalis fascia
- A transverse incision is made in the deep layer of fascia parallel to linea temporalis with a #11 blade. The undersurface of the fascia is undermined through this incision and separated from the temporalis muscle with scissors (Figure 41)
- Harvest the appropriate amount of fascia with scissors taking only as much as is needed so that should the patient require revision surgery, additional fascia can be harvested
- The fascial graft is compressed between two gauze swabs, but not dried
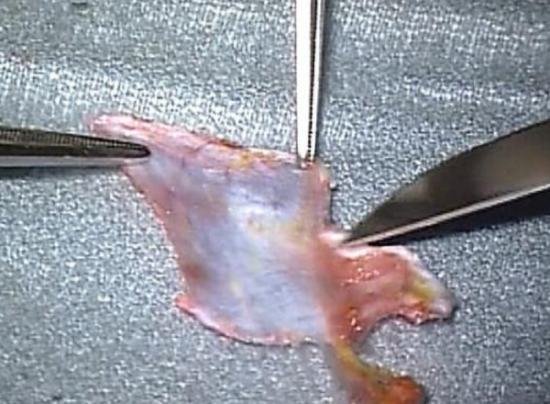
Figure 42: Scraping muscle off fascia
- The graft is then placed on a silicone block. The scrub nurse holds one corner of the graft, while the surgeon holds the other corner with anatomical forceps, and using a #10 blade, the graft is scraped clean of muscle fibers (Figure 42)
- Uneven edges are trimmed with a #10 blade
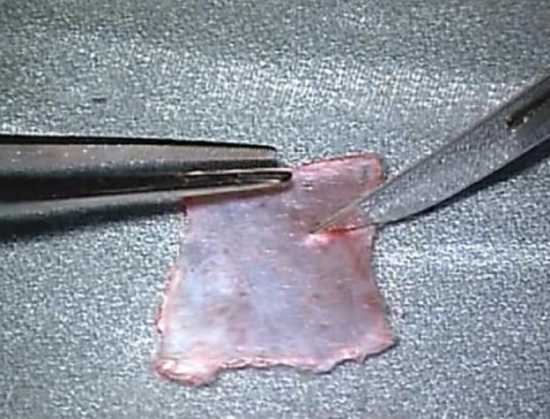
Figure 43: Incision to accommodate tensor tympani tendon
- An incision is made in the flap with a knife to accommodate the tensor tympani tendon at the anticipated location of the malleus handle (Figure 43)
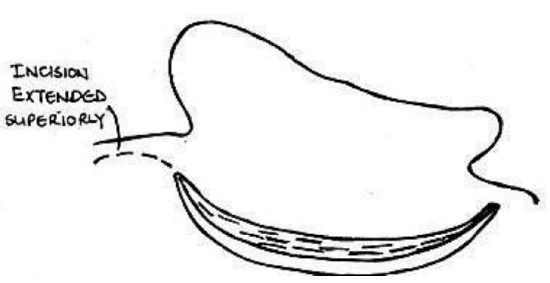
Figure 44: Extended postauricular incision for additional temporalis fascia
- When doing revision surgery, it is possible to extend the postauricular incision superiorly in order to find additional temporalis fascia (Figure 44)
Harvesting tragal cartilage and tragal perichondrium
Tragal cartilage is easily harvested by exposing the upper end of the tragal cartilage through an endaural approach.
- Hold the tragal cartilage with surgical forceps while dissecting the soft tissue off the cartilage with tympanoplasty scissors
- After exposing the amount of cartilage required for reconstruction, resect cartilage with overlying perichondrium with tympanoplasty scissors
- Using the operating microscope strip the tragal perichondrium from the cartilage with a microraspatory. The perichondrium may also be left attached to the tragal cartilage when reconstructing the posterosuperior canal wall
- To thin cartilage, hold it with Hudson Brown forceps and section the cartilage with a new #10 blade
Grafting Techniques
The authors use the terms “underlay” and “overlay” to refer to position of the graft relative to the bony (annular) sulcus. (In other texts it may refer to the position of the graft in relation to the tympanic membrane)
- With underlay technique the graft is placed medial to the remnant of the tympanic membrane and anterior tympanic sulcus
- With overlay technique the graft is placed lateral to the tympanic sulcus
Underlay technique is most often used for perforations involving the anterior quadrant. The graft is always placed medial to the malleus handle. Larger perforations often require a combination of anterior underlay and posterior/inferior overlay grafting technique.
Grafting limited perforations of the posterior quadrant (Figure 45)
Figure 45
These surgical steps are discussed in detail under endaural approach.
Grafting perforations that reach the anterior quadrant (Figure 46)
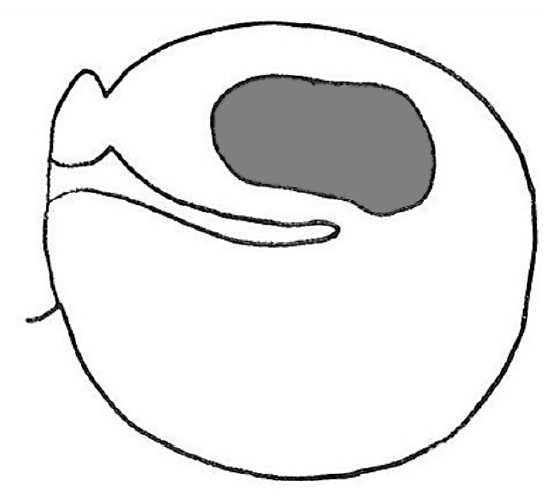
Figure 46
- Retroauricular approach is used
- Canalplasty is done
- The annulus is not elevated between 2 and 4 o’clock (right side) as this will cause blunting of the anterior tympanomeatal angle and lateralization of the tympanic membrane resulting in reduction of vibratory properties of the tympanic membrane
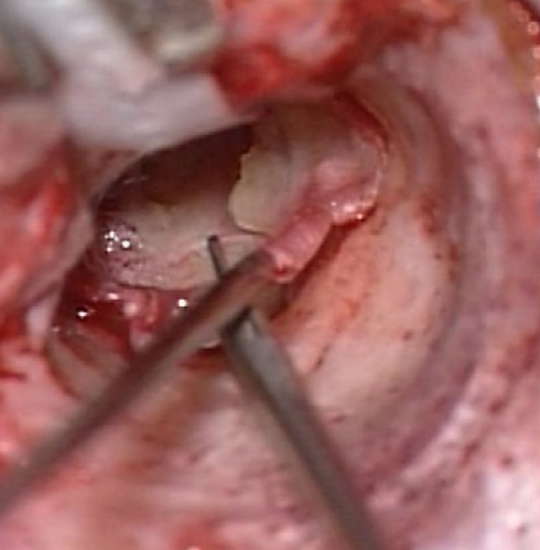
Figure 47: Dividing the tympanomeatal flap posteriorly
- If the perforation involves the anteroinferior quadrant then the tympanomeatal flap is divided posteriorly after it has been elevated (Figure 47)
- Scrape the undersurface of the tympanic membrane remnant with a 1.5 mm, 45° hook
- Anteriorly, the graft is underlaid under the edge of the tympanic remnant
- If the perforation extends inferiorly or posteriorly then the graft is underlaid anteriorly but overlaid posteriorly in the inferior or posterior quadrants
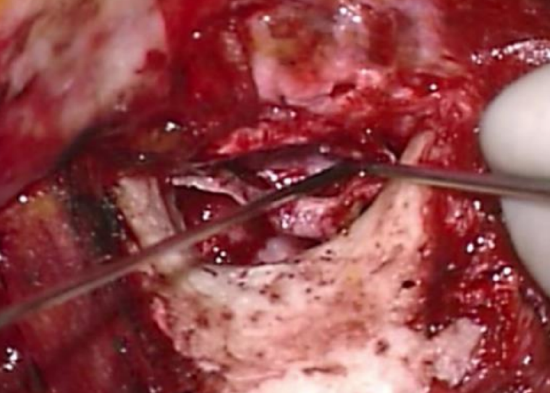
Figure 48: Meatal skin flap is being replaced
- The meatal skin flap is replaced (Figure 48)
- The fascia and meatal skin flap are secured with Gelfoam
Grafting perforations that reach the anterosuperior quadrant (Figure 49)
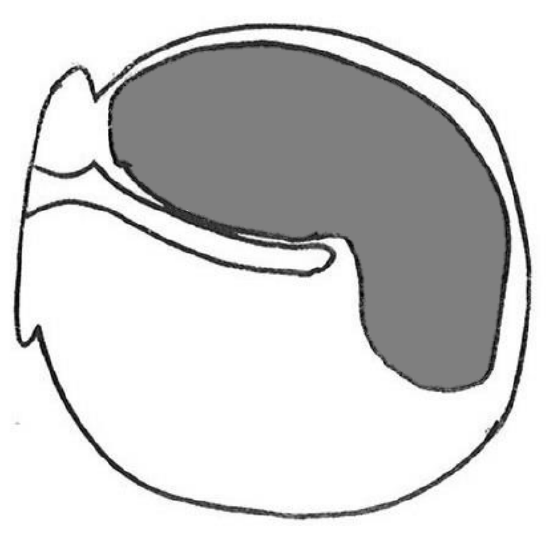
Figure 49
- Retroauricular approach is used
- Canalplasty is done
- Elevate and divide the tympanomeatal flap posteriorly (Figure 47)
- Scrape the undersurface of the tympanic membrane remnant with a 1.5 mm, 45° hook
- Fixing the graft anteriorly requires special anterior support
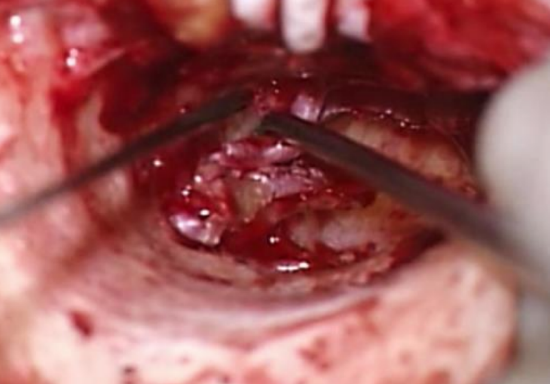
Figure 50: Detach tympanic annulus between 1 and 2 o’clock to create “buttonhole” anteriorly
- Detach the tympanic annulus between 1 and 2 o’clock (right ear) using a microraspatory to create a “buttonhole” anteriorly between annular ligament and bone (Figure 50)
- The facial graft is placed medial to the malleus
- A small “tongue” of the graft is maneuvered with a 1.5 mm 45° hook, to as close as possible to the “buttonhole”
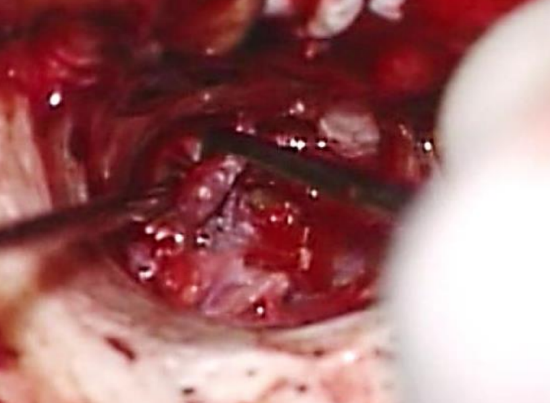
Figure 51: Use a microsuction tube to suck and pull “tongue” of temporalis fascia through “buttonhole”
- Use a microsuction tube to suck and pull the “tongue” of temporalis fascia through the “buttonhole” and then use a 1.5 mm hook to advance the graft into position (Figure 51)
Grafting subtotal perforations (Figure 52)
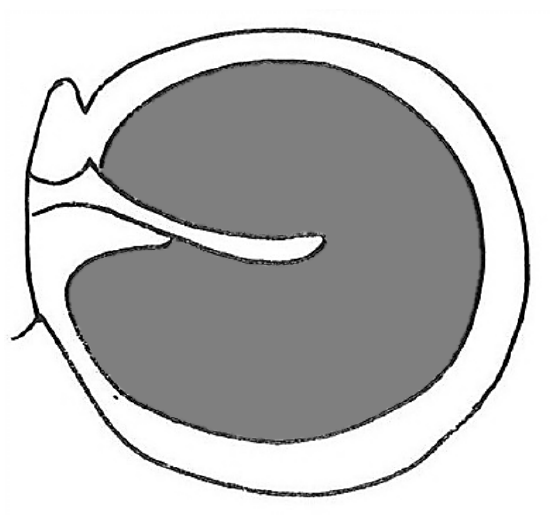
Figure 52
- Retroauricular approach is used
- Canalplasty is done
- Elevate and divide the tympanomeatal flap posteriorly (Figure 36)
- Subtotal perforations may only have a limited remnant of the tympanic membrane anteriorly
- Scrape the undersurface of the tympanic membrane remnant and adjacent bone with a 1.5mm, 45° hook
- The graft is underlaid both under the tympanic membrane and malleus handle
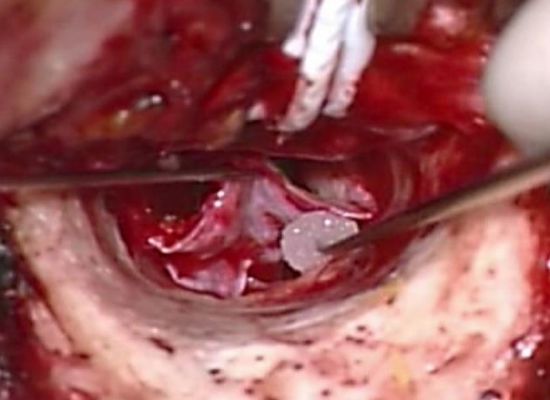
Figure 53: Graft supported anteriorly by Gelfoam soaked in Ringer’s lactate
- The graft is supported anteriorly by placing small pieces of Gelfoam soaked in Ringer’s lactate beneath the graft (Figure 53)
- Superiorly both limbs of the fascia overlap just above the neck of the malleus (Figure 43)
- Posteriorly and inferiorly, the graft is positioned as an overlay graft
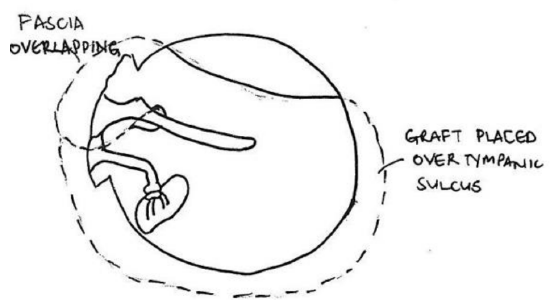
Figure 54: Fascia overlaps just above neck of malleus
- Gelfoam is then placed laterally over the graft to secure it in place over the tympanic sulcus (Figure 55)
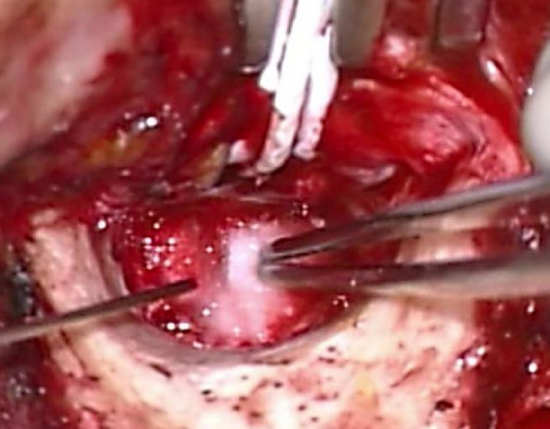
Figure 55: Gelfoam placed laterally to secure graft
Management of severely retracted malleus with chronic otitis media with
Occasionally patients with longstanding chronic otitis media do not develop erosion of the incus, but the malleus handle becomes medialized. In severe cases the umbo of the malleus touches or even attaches to the promontory. This presents a challenge insofar as that the tympanic membrane perforation needs to be reconstructed and at the same time the hearing needs to be improved. If fascia is placed as an underlay graft it will result in Grade 4 atelectasis and may subsequently reperforate. Lateralization of the malleus is easily achieved once the incus has been removed. The following is an effective technique to manage a severely retracted malleus:
- The malleus head and anterior mallear ligament are removed leaving the tensor tympani tendon intact
- An incus interposition is then done
- The perforation is grafted with fascia or cartilage by underlay technique
- Because the malleus has been lateralized it facilitates placement of the graft in the proper position
Reconstruction with closed mastoidoepitympanectomy
See the mastoidoepitympanectomy chapter for a description of reconstruction at the time of primary surgery
- The defect is often located superiorly in the epitympanum
- Cartilage is used to reconstruct the defect in the epitympanum
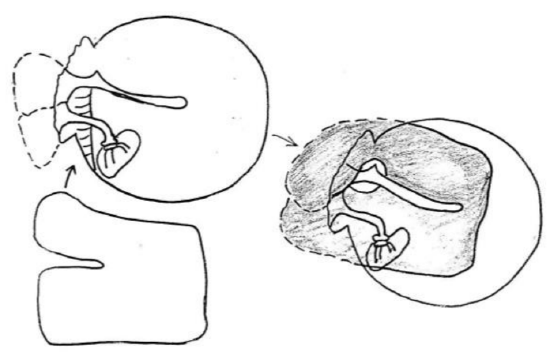
Figure 56: Temporalis fascia placed beneath handle of malleus so that two tongues of fascia overlap
- Temporalis fascia is placed beneath the handle of the malleus so that the two tongues of the fascia overlap each other in the epitympanum (Figure 56)
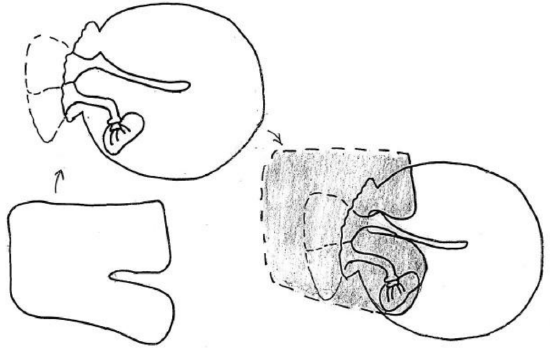
Figure 57: Two tongues overlap in middle ear beneath malleus handle
- An alternative technique is to position the graft such that the two tongues are positioned around the malleus and overlap each other in the middle ear beneath the malleus handle (Figure 57)
Reconstruction with open mastoidoepitympanectomy
See the Mastoidectomy and epitympanectomy chapter for a description of reconstruction at the time of primary surgery
- A circumferential tympanic groove is drilled with a small diamond burr to form a new tympanic sulcus
- The graft is placed as with repair of a subtotal perforation
- If a 2nd stage ossiculoplasty is planned, then it becomes necessary to place silastic sheeting in the middle ear and protympanum
Ossiculoplasty
Ossiculoplasty is defined as reconstruction of the hearing mechanism by establishing a connection between the tympanic membrane and the oval window. It is covered in detail in the chapter “Ossiculoplasty.”
References
- Fisch U, May J, Linder T. Tympanoplasty, Mastoidectomy, and Stapes Surgery. New York: Thieme; 2008
- Lerut B, Pfammatter A, Moons J, Linder T. Functional Correlations of Tympanic Membrane Perforation Size. Otol Neurotol. 2012; 33:379-86
- Hol KS, Nguyen DQ, Schlegel Wagner C, Pabst G, Linder TE. Tympanoplasty in chronic otitis media with an intact, but severely retracted malleus: A treatment challenge. Otol Neurotol. 2010; 31:1412-6
Acknowledgments
This surgical guide is based on the text by Professor Ugo Fisch (Tympanoplasty, Mastoidectomy, and Stapes Surgery) and the personal experience of Professor Linder, as well as course material for the temporal and advanced temporal bone courses conducted annually by Professors Fisch and Linder at the Department of Anatomy, University of Zurich, Switzerland.
Author
Tashneem Harris MBChB, FCORL, MMed (Otol), Fisch Instrument Microsurgical Fellow
ENT Specialist
Division of Otolaryngology
University of Cape Town
Cape Town, South Africa
harristasneem@yahoo.com
Senior Author
Thomas Linder, M.D.
Professor, Chairman and Head of Department of Otorhinolaryngology, Head, Neck and Facial Plastic Surgery
Lucerne Canton Hospital, Switzerland
thomas.linder@ksl.ch
Editor
Johan Fagan MBChB, FCS(ORL), MMed
Professor and Chairman
Division of Otolaryngology
University of Cape Town
Cape Town, South Africa
johannes.fagan@uct.ac.za


