4.15: Endoscopic Dacryocystorhinostomy (DCR) Surgical Technique
- Page ID
- 51033
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)OPEN ACCESS ATLAS OF OTOLARYNGOLOGY, HEAD & NECK OPERATIVE SURGERY
ENDOSCOPIC DACRYOCYSTORHINOSTOMY (DCR) SURGICAL TECHNIQUE
Hisham Wasl, Darlene Lubbe
Endoscopic dacryocystorhinostomy (DCR) is a surgical intervention for epiphora caused by obstruction of the nasolacrimal duct. Understanding endonasal anatomy, wide endoscopic marsupialization of the lacrimal sac, and meticulous care of the nasal mucosa are important to achieve good results.
Patients develop symptoms of tearing when there is an imbalance between tear production and drainage function of lacrimal system. Tearing can therefore be caused by
- Hypersecretion
- Epiphora
- Combinations of the above
Hypersecretion (lacrimation) is excessive tearing caused by reflex hypersecretion due to irritation of the cornea or conjunctiva e.g. trigeminal nerve stimulation in corneal disease.
Epiphora occurs with poor lacrimal drainage due to
- Mechanical obstruction of the lacrimal drainage system related to trauma, dacryocystolithiasis, sinusitis, and congenital nasolacrimal duct obstruction in children
- Lacrimal pump failure (functional epiphora) may be caused by eyelid laxity (as in facial nerve palsy), eyelid malposition, and punctum eversion
Relevant anatomy
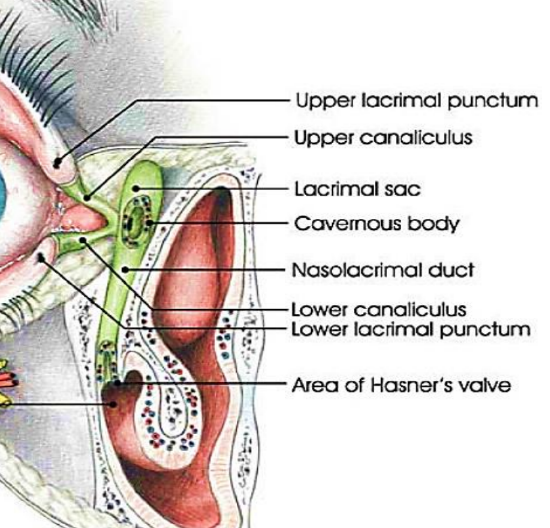
Figure 1: Ocular and nasolacrimal duct anatomy
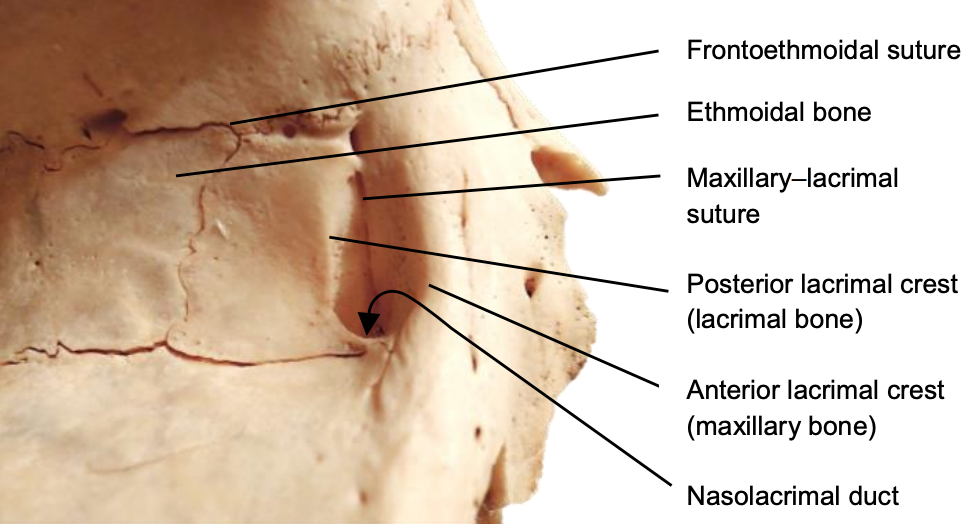
Figure 2a: Anterior and posterior lacrimal crests are formed by the frontal process of maxillary bone and lacrimal bones

Figure 2b: Area of bone to be removed
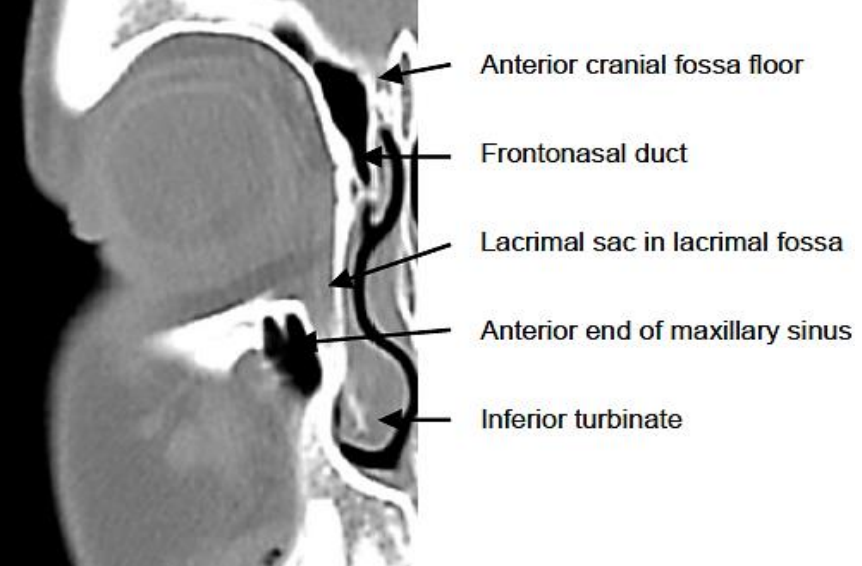
Figure 3a: Coronal CT scan through the lacrimal sac
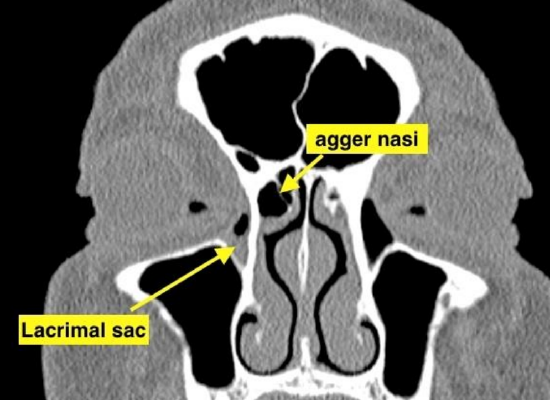
Figure 3b: Coronal CT scan through the lacrimal sac and agger nasi cell
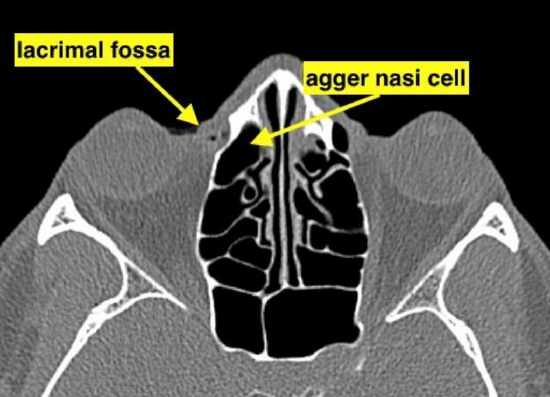
Figure 3c: Axial CT scan demonstrates anatomical relations between lacrimal fossa and agger nasi cell
The lacrimal puncta open at the medial ends of the upper and lower eyelids and drain into the lacrimal sac via the upper and lower canaliculi (Figure 1). The lacrimal sac is located in the lacrimal fossa and drains into the nasolacrimal duct (Figures 2, 3). The nasolacrimal duct runs within a bony canal created by the maxillary and lacrimal bones and opens into the inferior meatus of the nose (Figures 1-3).
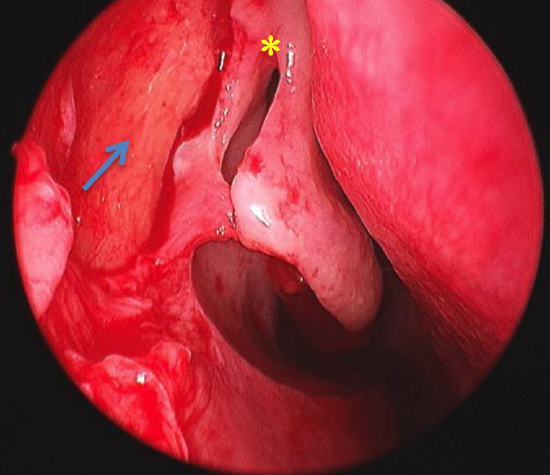
Figure 4: Endoscopic view of the axilla of the right middle turbinate (*) with mucosal flap raised to expose bone covering the lacrimal sac (arrow)
The lacrimal bone extends between the frontal process of the maxilla anteriorly (Figure 2), to the attachment of the uncinate posteriorly. It is important to note that the lacrimal bone and sac are located just anterior to the orbit. The retrolacrimal region of the lamina papyracea is thin and careless surgery to the uncinate at this point may lead to penetration of the orbit. The lacrimal sac extends approximately 9 mm above the axilla of the middle turbinate (Figure 4). The common canaliculus opens high on the lateral wall of the sac; this area should be exposed, and all bone removed during endoscopic DCR for a better result.
Evaluation of patients with epiphora
Clinical History and Examination
A detailed history and clinical examination help to
- Differentiate between hypersecretion, lacrimation and epiphora
- Define the pathological process
- Distinguish whether tearing is due to a functional or anatomical disorder
- Identify the site of the blockage
- Define a surgical approach (if required)
Examination includes
- Eyelids: Lower lid laxity, ectropion, entropion, punctum eversion, trichiasis, blepharitis
- Medial canthus: Lacrimal sac enlargement below the medial canthal tendon
- Palpation of lacrimal sac: Reflux of mucopurulent material from the punctum; pressure over sac in acute dacryocystitis causes pain
Special Investigations
Diagnostic tests are used to identify the cause of an obstruction and to choose appropriate treatment1. Diagnostic tests can be classified as follows:
- Anatomical tests to locate the site of obstruction
- Diagnostic probing
- Syringing (irrigation)
- Dacryocystography
- Nasal examination
- CT, MRI
- Physiological/Functional tests
- Fluorescein dye disappearance
- Scintigraphy
- Saccharine test
- Secretion tests
- Schirmer’s test
- Bengal rose test
- Tear-film breakup
- Tear lysozyme
Diagnostic probing & lacrimal syringing (irrigation)
Diagnostic probing and irrigation of the lacrimal system are important anatomical tests. They provide information about the site of obstruction but are unable to give information about functional insufficiency. Teaming up with ophthalmologists help ENT surgeons to obtain the required skills of probing and syringing the lacrimal system. The required instrumentation is shown in Figure 5.
Syringing
This is generally done by ophthalmologists in an outpatient clinic under local anesthesia
- Apply topical anesthesia by applying 1-2 drops of Oxybuprocaine or Benoxinate HCl 0.4% (Novesin Wander ® by Novartis) onto the puncta
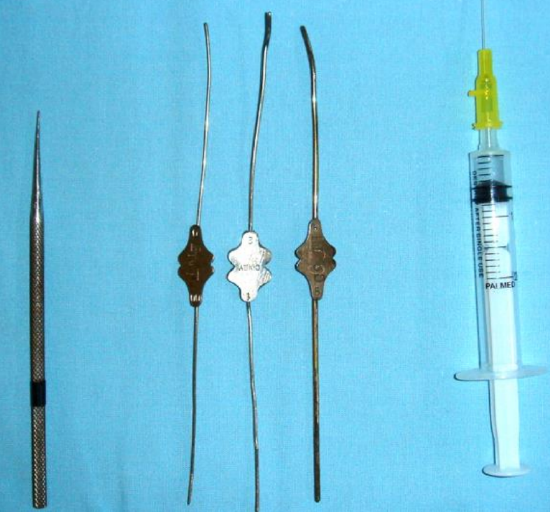
Figure 5: Dilator, probing and syringing instrumentation
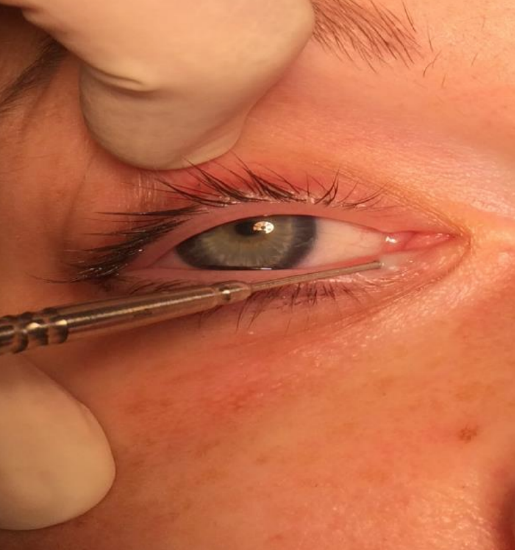
Figure 6: Punctum dilated with a punctum dilator
- Dilate the puncta with a punctum dilator if the puncta are small (Figure 5,6)
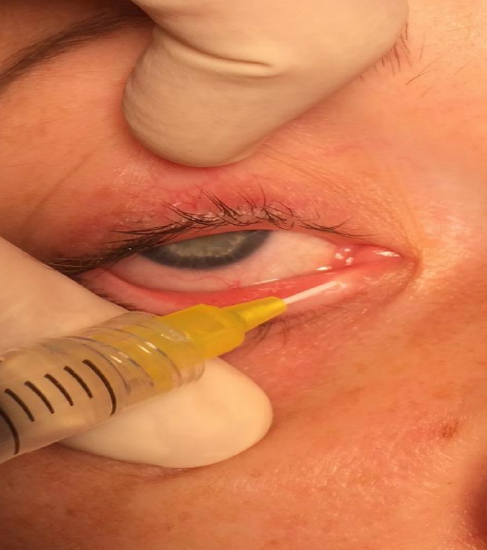
Figure 7: Syringing via lower canaliculus
- Insert a 24G (yellow) intravenous cannula into the inferior canaliculus, first aiming vertically and then horizontally (Figures 5, 7)
- Straighten the lower canaliculus by pulling the lower eyelid downwards and laterally (Figure 7)
- Advance the tip of the cannula 3-6 mm into the canaliculus
- Irrigate the lacrimal drainage system with sterile water
How to interpret syringing tests
- Reflux through the opposite punctum suggests obstruction in the common canaliculus or distal to it
- Fluid coming directly back through the same punctum indicates a canalicular obstruction; repeat syringing through the other canaliculus
- Distention of the lacrimal sac indicates obstruction of the nasolacrimal duct
- Irrigation into the nose indicates an anatomically patent system, but not necessarily a functional system
Diagnostic Probing Probing is indicated only if syringing demonstrates obstruction and the site and extent of the obstruction need to be determined. If fluid refluxes through the opposite punctum, it suggests obstruction of the common canaliculus or more distally; this distinction should be made with diagnostic probing
- Apply topical anesthesia by using 1-2 drops of Oxybuprocaine or Benoxinate HCl 0.4% Novesin Wander ® by Novartis) onto the puncta and wait 20 seconds
- Dilate the puncta with a punctum dilator
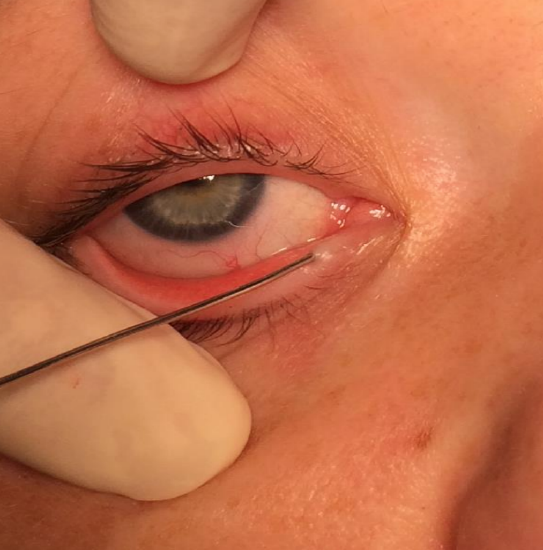
Figure 8: Diagnostic probing
- Advance an appropriately sized lacrimal probe into the canaliculus (Figures 5, 8)
- First pass the probe vertically through the punctum, and then horizontally until it encounters the lacrimal bone or meets the canalicular obstruction
How to interpret a diagnostic probing test
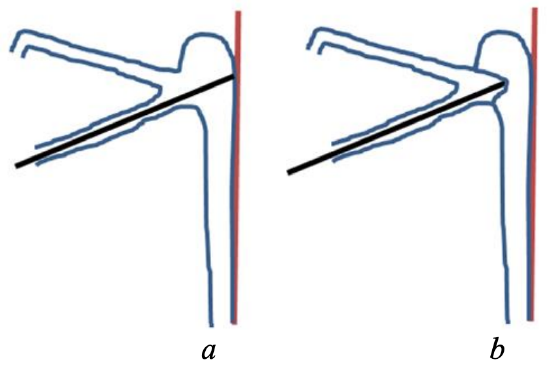
Figure 9a: “Hard stop” when probe is passed into lacrimal sac and hits medial bony wall; b: “Soft stop” when probe displaces common canaliculus towards medial wall of sac…is associated visible medial shift of inner canthus
- A hard stop is when the probe is advanced along the canaliculus and encounters the lacrimal bone. This means that the probe has passed into the sac and touched its bony medial wall, and that the common canaliculus is patent (Figure 9a)
- A soft stop is a spongy feeling encountered when there is an obstruction proximal to the lacrimal sac and the probe cannot be passed into the sac. In such cases the obstruction is at the common canaliculus and the lacrimal probe presses the common canaliculus and the lateral wall against the medial wall of the sac (Figure 9b). There will also be a medial shift of the inner canthus when advancing the probe toward the lacrimal bone due to the probe displacing the common canaliculus medially
- Reflux through the opposite punctum when syringing a hard stop suggests obstruction of the sac or the nasolacrimal duct
It is important to note the presence of a hard stop or a soft stop, because the treatment of an obstruction at the lacrimal sac or duct vs. at the common canaliculus requires different surgical interventions.
Radiological investigations
Radiological investigations are done if doubt exists about the surgery that is required2, 3. Both dacryocystography and scintigraphy provide some idea of the level of obstruction and whether a tight common canaliculus is contributing to the epiphora.
Dacryocystography is indicated when there is obstruction in the lacrimal system with syringing. It can assist with understanding the internal anatomy of the lacrimal system. Indications for dacryocystography include
- Complete obstruction: size of sac, determining exact site of obstruction (common canaliculus or sac)
- Incomplete obstruction and intermittent tearing: site of stenosis, diverticula, stones, and absence of anatomical pathology (functional disorders)
- Failed lacrimal surgery: size of the sac
- Suspicion of sac tumors
Nuclear lacrimal scintigraphy is a functional test, and is useful to assess the site of delayed tear transit. It is especially helpful in difficult cases with an incompletely obstructed system e.g. questionable eyelid laxity, and questionable epiphora.
Computed tomography (CT) is used with tumors, rhinosinusitis, facial trauma, and following facial surgery. With concomitant sinus disease, CT assists a surgeon to address the sinuses at the same time as the DCR.
Magnetic resonance imaging (MRI) is rarely used to investigate tearing patients.
Nasal endoscopic examination
Endoscopic examination of the nasal cavity is obligatory. It provides very important information about e.g. nasal polyps, nasal masses, rhinosinusitis, tumors, anatomical variations, and nasal septal deviation.
Endoscopic DCR surgery technique
Anesthesia and preparation of surgical field
- Surgery is performed under general anesthesia with the endotracheal tube out of the way of the endoscope and instruments
- The patient is placed supine, either flat or slightly flexed to 15 degrees, and slightly rotated towards the surgeon
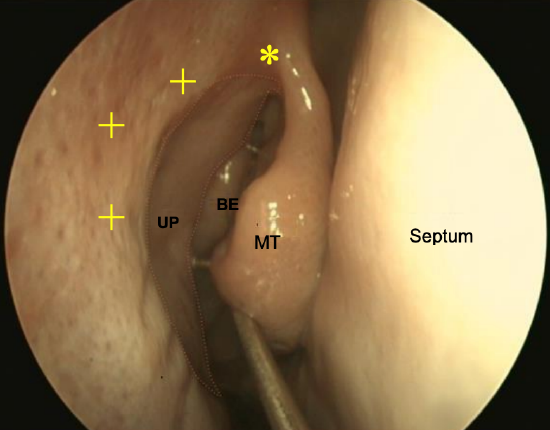
Figure 10: Injection points of lidocaine with 1:100,000 adrenaline into axilla of middle turbinate (*) and frontal process of maxilla (+)
- Using a dental syringe, inject 2 mL of 1% lidocaine with 1:100,000 adrenaline into the axilla of the middle turbinate and the frontal process of maxilla (Figure 10)
- Insert ribbon gauze or neurosurgical patties soaked in 2 mL of 1:1000 adrenaline between the inferior turbinate and the nasal septum and in the middle meatus to achieve topical decongestion
- A dose of co-amoxiclav or cefazolin is given at induction of anesthesia
Surgical instruments

Figure 11a: Kerrison punch
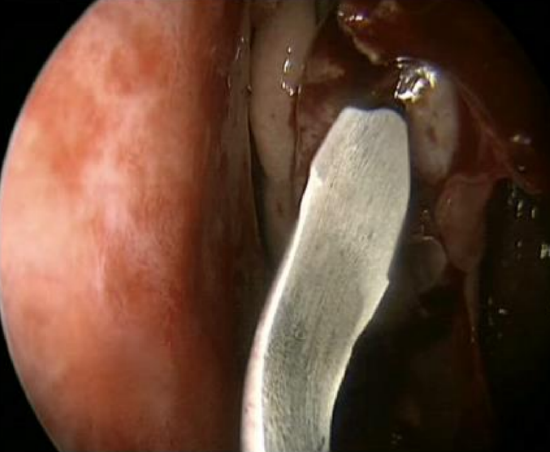
Figure 11b: 2.75 mm Cataract knife
- Surgical instruments required:
- Dental syringe
- No 15-scalpel blade
- Suction Freer elevator
- Kerrison punch (Figure 11a)
- 2.75 mm Cataract knife (Figure 11b)
- Small Blakesley forceps
- Punctum dilator and probes
- DCR intubation set
It is quicker to remove bone with a Kerrison’s punch than with a DCR burr. The DCR bur is only used when the punch is unable to engage the bone adequately.
Surgical steps
1. Septoplasty
- Have a low threshold for performing a septoplasty if a septal deviation hampers access to the middle meatus and lateral nasal wall
- Ideally place the septal incision on the side contralateral to the DCR
2. Create a posteriorly based mucosal flap to expose the lacrimal bone
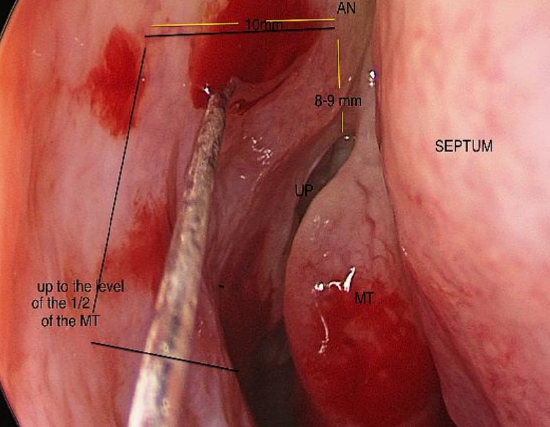
Figure 12: Superior and inferior incisions and the area where the local anesthetic is injected
- Use a no 15 scalpel blade to make a superior incision that runs horizontally 8-9 mm above the axilla of the middle turbinate (Figure 12)
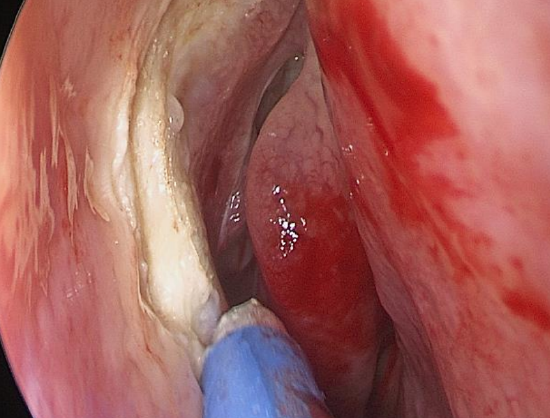
Figure 13: Suction diathermy can be to cauterize the incision sites for hemostasis
- One can use suction diathermy to cauterize the incision line before making the cut (Figure 13)
- Extend the incision anteriorly for approximately 10 mm onto the frontal process of the maxilla
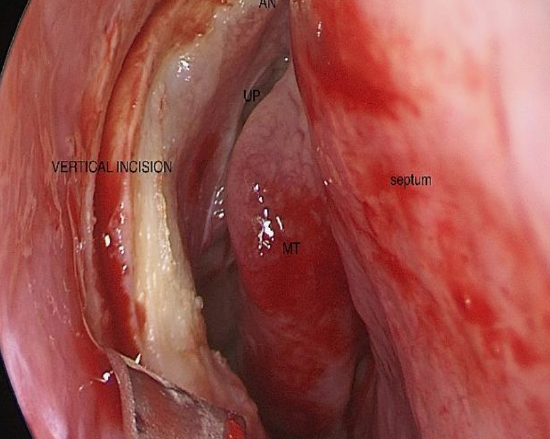
Figure 14: Vertical incision
- Turn the blade vertically and make a cut onto the frontal process of the maxilla from the superior incision to just above the insertion of the inferior turbinate (Figure 14)
- Turn the blade horizontally and make the inferior incision from the insertion of the uncinate to join the vertical incision (Figures 12, 14)
3. Raise a mucosal flap
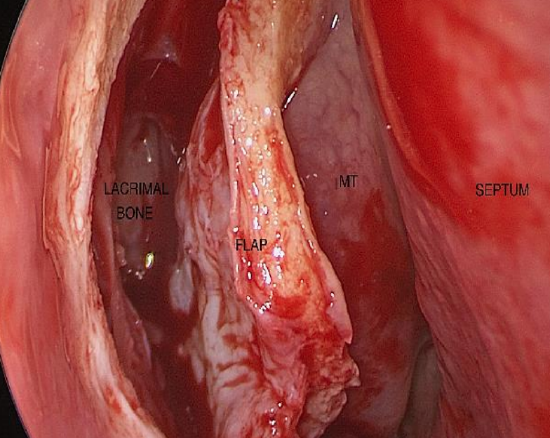
Figure 15: Raising the mucosal flap
- Use a suction Freer dissector to raise a mucosal flap and to expose the underlying bone (Figure 15)
- It is important to stay on bone to avoid losing the surgical plane
4. Remove bone to expose lacrimal sac
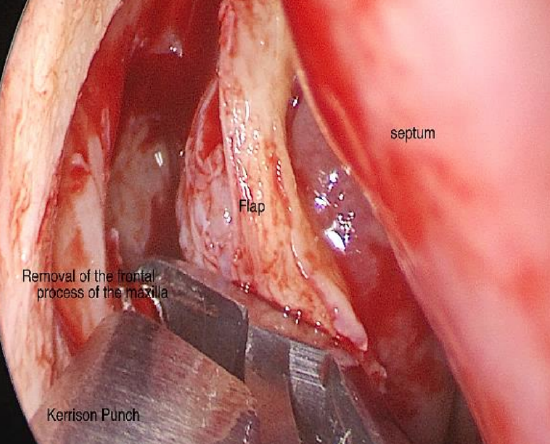
Figure 16: Removal of bone of the frontal process of the maxilla exposes the lacrimal sac
- Use a Kerrison’s punch to remove the bone of the frontal process of the maxilla overlying the lacrimal sac (Figure 16)
- It is quicker to remove bone with a Kerrison’s punch than with a DCR bur
- Only use a DCR bur when the punch is unable to adequately engage the bone
- Expose the sac by removing bone up to the mucosal incisions superiorly, inferiorly, and anteriorly, so the sac forms a prominent bulge into the nasal cavity (Figure 16)
- Remove all the lacrimal bone up to the insertion of the uncinate, but do not disturb the uncinate itself
- The lacrimal probe should be passed from the puncta through to the nasal cavity without feeling any bone obstructing the pathway of the probe. If this is not so, then more bone should be removed superiorly
- This retrolacrimal region where the uncinate inserts into the lamina papyracea is extremely thin, so take care not to cause an accidental orbital injury
- The common canaliculus opens high on the lateral wall of the sac; this area should be exposed during endoscopic DCR for a better result
5. Expose agger nasi cell
- The agger nasi cell is situated medially, superiorly and more posteriorly to the lacrimal fossa (Figure 3b)
- Open the agger nasi cell using a Kerrison’s punch
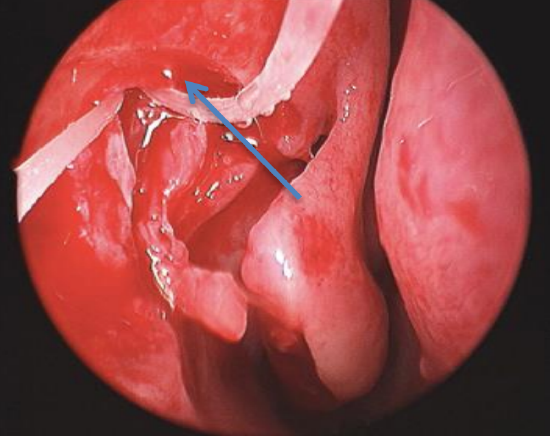
Figure 17: Exposure of agger nasi (arrow); 2 silastic tubes visible
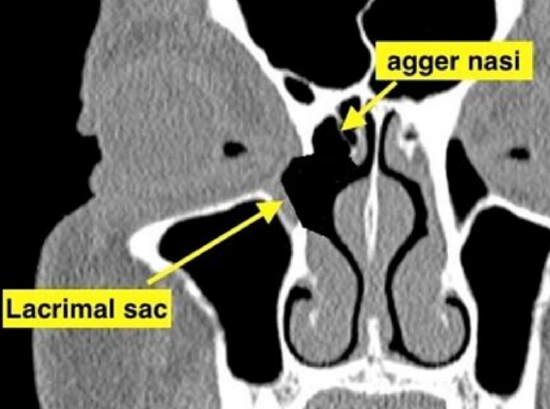
Figure 18: Agger nasi (arrow) has been opened (compare to Figure 3b)
- This allows better exposure of the lacrimal sac and allows the mucosa of the sac to lie against the agger nasi mucosa (Figures 17, 18)
6. Marsupialize the lacrimal sac
- Cannulate the superior or inferior canaliculus with a probe, taking great care not to make a false passage in the delicate lacrimal system
- Both the upper and lower canaliculi have an angulated course that needs to be carefully followed to avoid creating such a false passage
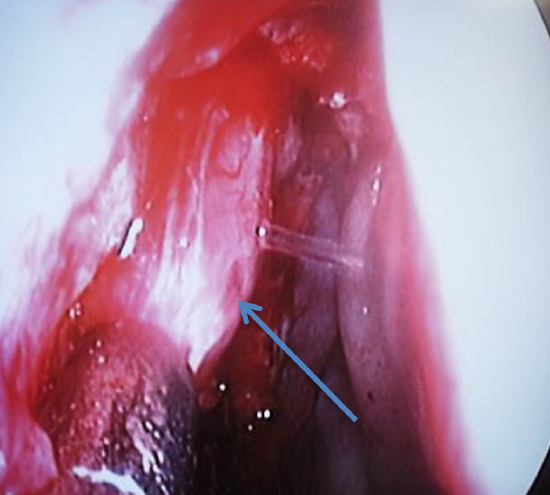
Figure 19: Tenting the wall of the sac with a probe before incising the lacrimal sac
- Tent the lacrimal sac with the lacrimal probe under endoscopic vision (Figure 19)
- Incise the medial wall of the sac only when the lacrimal probe can be clearly seen tenting the sac wall
- Use a DCR knife or cataract knife to vertically incise the sac
- Cut releasing incisions in the posterior and the anterior flaps of the sac
- Either Bellucci micro ear scissors or a sickle knife can be used for the release incisions
- The sac must be widely marsupialized, and be widely open and lying flat on the lateral nasal wall (Figure 17)
7. Trimming the nasal mucosal flap
- Preserve the mucosal flap until the end of the procedure to protect the nasal septum during the surgery
- Trim the mucosal flap so that only small superior and inferior rims remain, and the lacrimal sac remains wide open
- Return the remainder of the flap onto the lateral wall and ensure that the exposed bone is covered
8. Inserting silastic stents
- The authors prefer to stent the DCR neo-ostium (not all surgeons do so)
- Two silastic lacrimal tubes are used to stent the lacrimal system (Figures 17, 19)
- Dilate the puncta with a punctum dilator if the puncta are small (Figure 6)
Figure 20: Pass the lacrimal tubes through the inferior and superior puncta into the nose
- Pass the silastic lacrimal tubes through the superior and inferior puncta into the nasal cavity (Figure 20)
- Secure the tubes by tying the ends together, using 4-6 knots, on the nasal side
- It is important not to tie the tubes too tightly as this can cause adhesions between the puncta at the medial canthus; this is avoided by passing the tip of an artery clip through the loop at the medial canthus while knotting the tubes
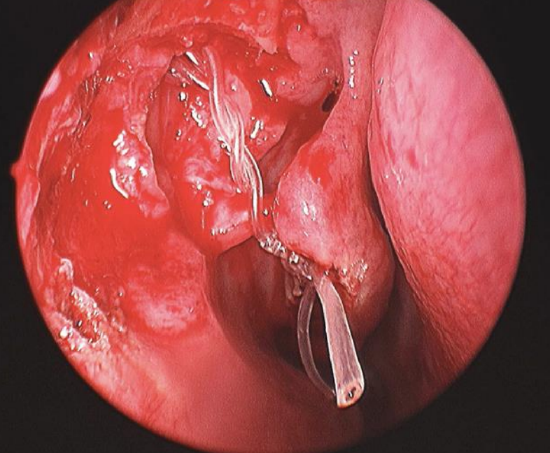
Figure 21: Endoscopic view of tied ends of silastic tubes passing through the common canaliculus into the nasal cavity
- Use a Tilley’s nasal forceps to advance the knot, which is tied outside the nasal cavity, down the nasal cavity (Figure 21)
- The first knot should be about 5 mm below the opened sac to avoid the knots getting stuck within the sac itself
- Check that there is no tension on the tubes at the medial canthus or in the nose
Postoperative Care
- Patients are generally discharged within a few hours
- Prescribe a 5-day course of decongestant nasal drops
- Apply antibiotic eye drops for 2 weeks
- Irrigate the nose with saline
- Patients are again seen at 2 weeks to clean the nose, remove crusts and to remove early adhesions
- Silastic tubes are removed in the office after 4-6 weeks
Complications
Early
- Adhesions can form if care is not taken to preserve the mucosa of the nasal septum and middle turbinate
- If the tubes are tied too tightly, an adhesion can form at the medial canthus between the upper and lower puncta, and may require difficult oculoplastic corrective surgery
- If the tubes are tied too tightly in the nasal cavity the knots can cause granulation tissue at the neo-ostium
- Epistaxis is unusual unless there has been significant trauma to the nasal mucosa or turbinates
Late
- Restenosis
- Failed surgery
Revision surgery
Revision surgery is occasionally indicated. It is imperative that the reason for failure is ascertained, which may include:
- Not enough bone removed superiorly
- Not enough bone removed inferiorly causing a sump effect with tears collecting in the sac
- Sac not marsupialized widely enough
- Missed foreign body or stones within the sac
- Stenosis of the superior or inferior canaliculi
- Stenting that caused excessive granulations
References
- Hurwitz JJ. The Lacrimal System. Lippincott-Raven Publishers, Philadelphia (1996).
- Hurwitz JJ, Welham RAN. Radiography in functional lacrimal testing. Br J Ophthalmol 1975; 59:323–31
- Hurwitz JJ, Molgat Y. Radiological test of lacrimal drainage. Diagnostic value versus cost-effectiveness. Lacrimal system. Symposium on the Lacrimal System, Brussels, 23–24 May 1992, pp 15– 26
Authors
Hisham Wasl MD, FCORL
University of Cape Town
Karl Storz Rhinology Fellow
Division of Otolaryngology
University of Cape Town
Cape Town, South Africa
hisham707@yahoo.com
Darlene Lubbe MBChB, FCORL
Associate Professor
Division of Otolaryngology
University of Cape Town
Cape Town, South Africa
doclubbe@gmail.com
Editor
Johan Fagan MBChB, FCS (ORL), MMed
Professor and Chairman
Division of Otolaryngology
University of Cape Town
Cape Town, South Africa
johannes.fagan@uct.ac.za


