2.3: Water
- Page ID
- 11103
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Learning oBjectiveS
- Define hydrogen bonds and describe how they form
- Describe the behavior of ionic compounds when placed in water
- Differentiate between hydrophobic and hydrophilic substances
- Define the terms solution, solvent, and solute
Atoms in the water molecule are held together by polar covalent bonds, as we saw in the previous Learning Objective. The unequal share of electrons in the O-H polar covalent bonds results in the oxygen atom having a slightly negative charge density (δ-) and hydrogen atom having a slightly positive charge (δ+). The slightly negative Oδ- in one water molecule is attracted to the slightly positive Hδ+ of a neighboring water molecule as shown in figure /(PageIndex{1}/) below.
The electrical attraction between Hδ+ in one molecule and Oδ- in another molecule is called a hydrogen bond. (Hydrogen bond can also be formed between Hδ+ in one molecule and N in another molecule). A hydrogen bond is a weaker type of attraction, but many hydrogen bonds add up. Hydrogen bonds explain many of the properties of water.
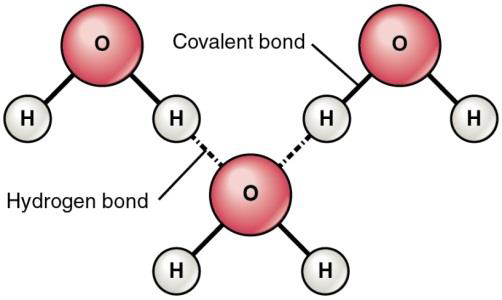
Figure \(\PageIndex{1}\) Hydrogen bonds are relatively weak. They occur between atoms of different molecules and are shown as a dotted lines rather than a solid line. Covalent bonds are stronger, occur between atoms of the same molecule and are shown as solid lines
Concepts, Terms, and Facts Check
Study Question Write your answer in a sentence form (do not answer using loose words)
1. What is a hydrogen bond?
When an ionic compound, or salt, (e.g. NaCl) is placed in water, its ionic components (e.g. Na+ and Cl-) dissociate (separate) and water molecules surround each ion. This is shown in figure \(\PageIndex{2}\) below. The small positive charges on hydrogen atoms in water molecules attract the anions (Cl-), and the small negative charges on the oxygen atoms attract the cations (Na+). Thus, because water is a polar covalent liquid, many salts and some polar covalent molecules (e.g. glucose) tend to ‘dissolve’ in water, while non-polar covalent molecules (e.g. fat) do not.
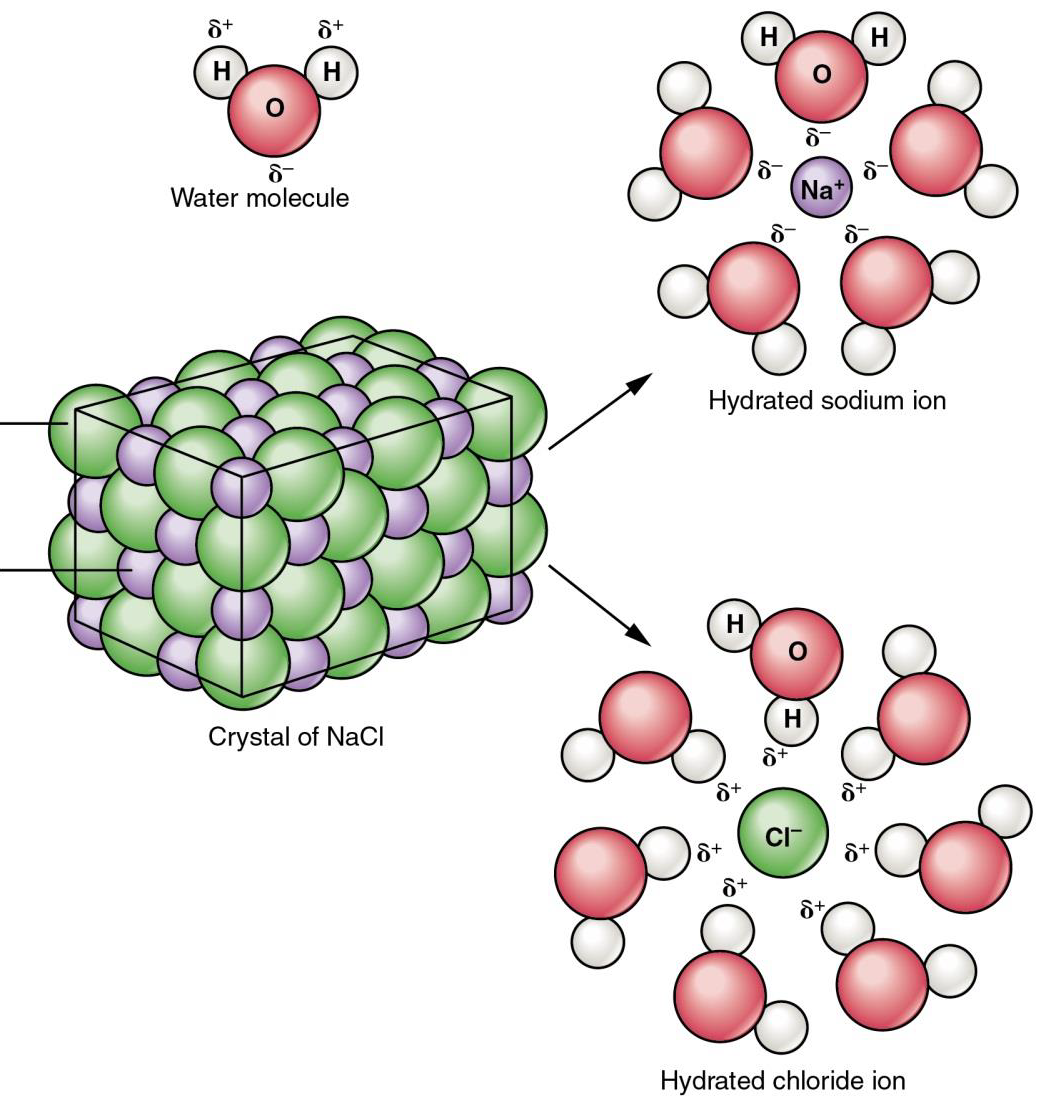
Figure \(\PageIndex{2}\) A sodium chloride crystal dissociates not into molecules of NaCl, but into Na+ cations and Cl– anions, each completely surrounded by water molecules.
Concepts, terms, and facts check
Study Question Write your answer in a sentence form (do not answer using loose words)
1. What happens to an ionic compound (salt) when placed in water?
Polar molecules are generally water soluble (they mix with water well), so they are referred to as hydrophilic, or “water-loving”. (hydro = water; philia = loving, tendency toward). Alcohols, salts and some sugars are examples of hydrophilic substances. Nonpolar molecules do not interact with water, and they are not water soluble (they do not mix with water), so they are referred to as being hydrophobic, or “water-fearing” (phobos = fear). Oils, fats and waxes are examples of hydrophobic substances. See examples of hydrophobic and hydrophilic substances in figure \(\PageIndex{3}\) below.


Figure \(\PageIndex{3}\) Sucrose (sugar table) molecules are hydrophilic and mix well with water, whereas unsaturated fats (mineral oils) are hydrophobic and do not mix well with water
Concepts, terms, and facts check
Study Questions Write your answer in a sentence form (do not answer using loose words)
1. What does hydrophilic mean?
2. What does hydrophobic mean?
A mixture is a combination of two or more substances, where each substance maintains its own properties. When we mix sucrose (table sugar) with water, we have a liquid mixture. Water molecules have not changed, and sucrose molecules have not changed either; they are mixed together. Each one maintains its own chemical properties.
A solution is a type of liquid mixture that consists of a solvent that dissolves a substance called a solute. A “sugary water” solution consists of water (solvent) that dissolves sucrose, or table sugar (solute). A drop of ink is a solution of water (solvent) and tiny colorful particles (solute). See another example in figure \(\PageIndex{4}\) below.
Most chemical reactions that happen inside and outside our body occur among compounds dissolved in water. For this is reason water is considered the “universal solvent”.

Figure \(\PageIndex{4}\) A saline water solution can be prepared by dissolving table salt (NaCl) in water. Salt is the solute and water the solvent.
Concepts, terms, and facts check
Study Questions Write your answer in a sentence form (do not answer using loose words)
1. What is a mixture?
2. What is a solution?
3. What is a solvent?
4. What is a solute?

