2.2: Chemical Bonds
- Page ID
- 11102
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)Learning ObjectivES
- Define chemical bonding, molecules, salts, and compounds; and list 3 types of chemical bonds important for the study of human physiology
- Define ionic bonds and describe how they form, and define salts
- Define covalent bonds and describe how they form, and differentiate between the two types of covalent bond
Atoms and ions can combine by chemical bonds. A chemical bond is an interaction between atoms or ions that stabilizes their outer shells. The interaction happens among the valence electrons (the ones in the outermost orbital shell). As a product of this interaction, participating atoms complete eight electrons in their outermost shells, form a chemical bond, and become stable.
Chemical bonding is responsible for the formation of molecules and salts. Molecules are substances composed of two or more atoms held together by a chemical bond. For example, in a molecule of carbon dioxide (CO2) the atom of carbon and the two atoms of oxygen are held together by chemical bonds. Salts are substances composed of ions held together by a chemical bond. For example, in a crystal of NaCl, table salt, Na+ and Cl- are held together by a chemical bond.
Salts and molecules made up of two or more atoms of different elements are called compounds. For example CO2, H2O, and NaCl are compounds, whereas O2 or H2 are not.
There are three types of chemical bonds important for the study of the human physiology. These are ionic bonds, covalent bonds, and hydrogen bonds.
Ionic bonds occur between ions with opposite charges (between anions and cations); covalent bonds occur between atoms of the same molecule; and hydrogen bonds occur between atoms in different molecules (one of them being hydrogen), or different parts of the same molecule.
Concepts, terms, and facts check
Study Questions Write your answer in a sentence form (do not answer using loose words)
1. What is a chemical bond?
2. What subatomic particles participate in a chemical bond?
3. What is a molecule?
4. What is a salt?
5. What is a compound?
6. What is an ionic bond?
7. What is a covalent bond?
8. What is a hydrogen bond?
Ionic bonds are formed by the electrical attraction between ions of opposite charges. The attraction between a cation and an anion forms an ionic bond.
Ions form when atoms take on or give up electrons following the octet rule.
Salts are a class of compounds formed by ionic bonds between ions. For example NaCl, table salt, forms when Na+ forms an ionic bond with Cl-. See the structure of NaCl below in figure \(\PageIndex{1}\). When a salt crystal is placed in water –as you can observe by placing table salt crystals in a glass of water-, it dissociates (separates) into its forming ions, or electrolytes, in a way that we will explore in an upcoming learning objective.
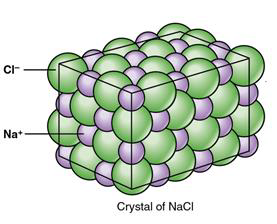
Figure \(\PageIndex{1}\) The structure of a NaCl crystal is given by the interaction of its forming ions, Na+ and Cl-, which are linked by ionic bonds
Concepts, Terms, and Facts Check
Study Questions Write your answer in a sentence form (do not answer using loose words)
1. What is an ionic bond?
2. What is a salt?
Instead of taking on or giving up electrons completely as happens in ionic bonding, atoms with four or more valence electrons may share pairs of electrons so both atoms’ outer shells complete eight electrons (or two for the case of hydrogen). Each pair of shared electrons moves in an orbital cloud around the nuclei of the two atoms. This sharing of electrons between atoms is called a covalent bond (co- = "together, mutually, in common", valent = relative to the valence electron).
Figure \(\PageIndex{2}\) below shows in (a) that a molecule of hydrogen has two atoms of the element hydrogen. This can be represented by either a molecular formula, H2, which shows how many atoms of each different type of element form a molecule or a structural formula, H-H, which shows the single covalent bond involved as one line, representing the pair of shared electrons. The oxygen molecule is formed by two atoms of the element oxygen as shown in (b). The molecular formula of the gas oxygen is O2, while the structural formula of this molecule is O=O, where the double line shows a double covalent bond indicating two pairs of shared electrons. A molecule of carbon dioxide is shown in (c) and has a molecular formula of CO2, showing that it is made of one atom of the element carbon and two atoms of the element oxygen. The structural formula of carbon dioxide in O=C=O, shows that the molecule has two double covalent bonds.
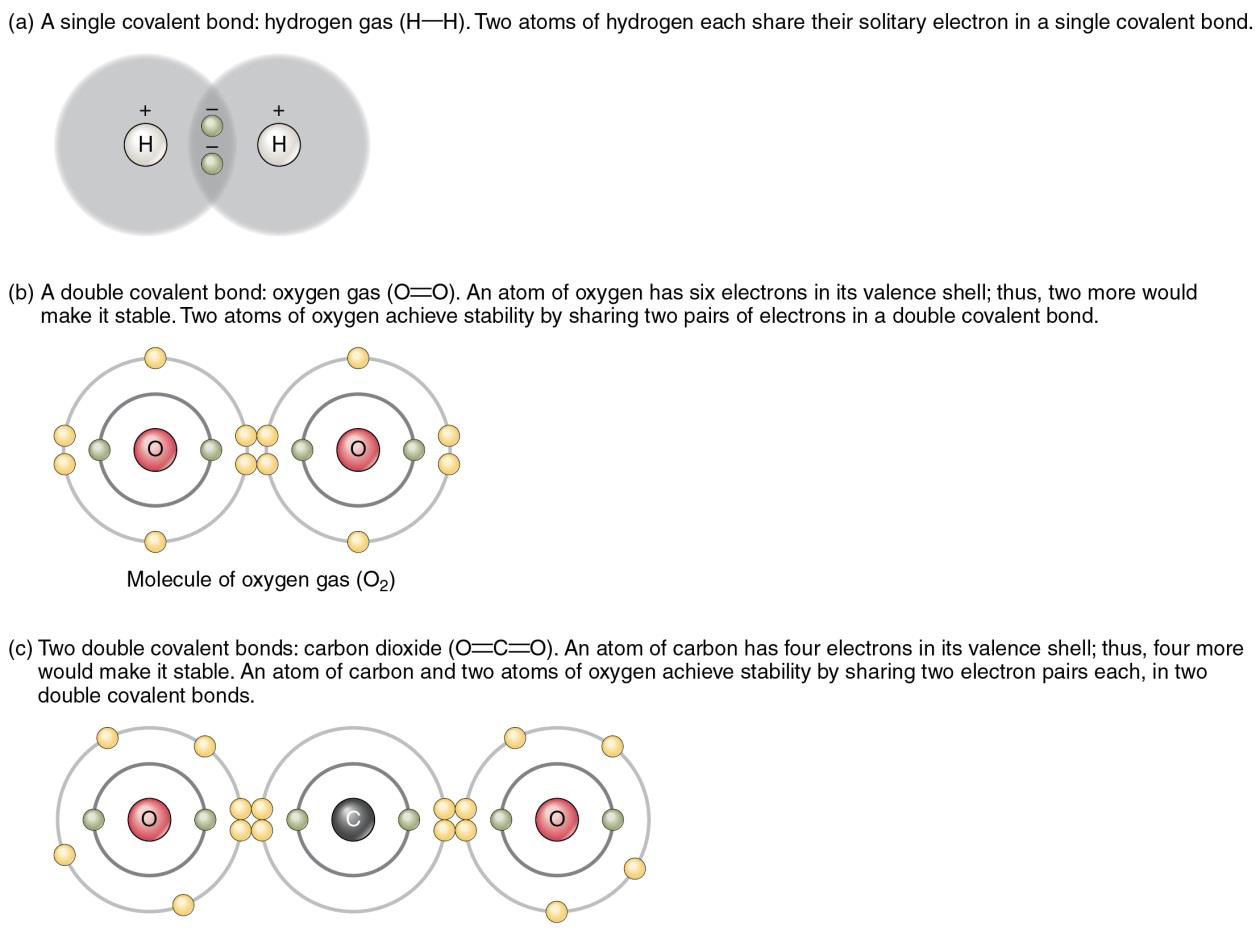
Figure \(\PageIndex{2}\) The examples shown above show sharing of electrons between atoms with similar a number of electrons (carbon and oxygen have six and eight, respectively). In these cases, the shared electrons spend about the same time moving around each one of the nuclei of both atoms. This type of covalent bond where electrons are equally shared is called a non-polar covalent bond. Nonpolar covalent bonds do not have a charge and are electrically neutral.
A second type of covalent bond occurs when electrons between atoms are shared in an unequal manner and is called a polar covalent bond. A polar covalent bond is common when either oxygen or nitrogen is sharing electrons with hydrogen. As shown in Figure \(\PageIndex{3}\) below, water (H2O) is a polar covalent molecule, in which one oxygen atom is covalently bound to two hydrogen atoms. The oxygen atom has 8 protons (and 8 electrons) and the hydrogen atom has just one proton (and one electron). The eight protons in the oxygen atom draw the pair of shared electrons toward oxygen and away from hydrogen. This is because the eight protons in the nucleus of oxygen attract the shared electrons with a stronger force than the single proton in the nucleus of hydrogen. This unequal share of electrons results in the atom that attracts the electrons more, in this example oxygen, having a slightly negative charge density (noted as δ-) and the other atom, in this example hydrogen, having a slightly positive charge density (noted as δ+). Note that we refer to charge density, as opposed to net charge, because neither oxygen nor hydrogen gains or loses electrons.
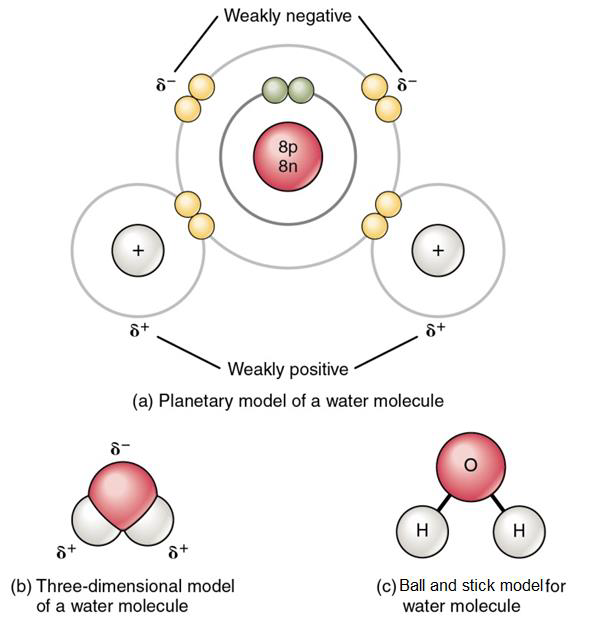
Figure \(\PageIndex{3}\) Water: a polar covalent molecule (a) shows two polar covalent bonds in the water molecule. See the explanation in the paragraph above; (b) and (c) show two other ways to represent a water molecule, other than the molecular formula, H2O, and the structural formula, H-O-H.
Summarizing
Covalent bonds are formed when atoms in a molecule share electrons. There are two
types of covalent bonds. Molecules with non-polar covalent bonds equally share
electrons between atoms and have an even arrangement of charges. Molecules with
polar covalent bonds share electrons unequally between atoms and have an
uneven arrangement of changes.
Concepts, terms, and facts check
Study Questions Write your answer in a sentence form (do not answer using loose words)
1. What is a covalent bond?
2. What is the difference between a single covalent bind and a double covalent bond?
3. What is the difference between a molecular formula and a structural formula?
4. What is a non-polar covalent bond?
What is a polar covalent bond?

