10.4: Riboflavin
- Page ID
- 40983
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\dsum}{\displaystyle\sum\limits} \)
\( \newcommand{\dint}{\displaystyle\int\limits} \)
\( \newcommand{\dlim}{\displaystyle\lim\limits} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\(\newcommand{\longvect}{\overrightarrow}\)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)A student once asked this question:
"I started taking the Mega Man Sport Multi-vitamin from GNC and about an hour or two after consumption, with a meal, my pee is bright, practically neon yellow. What does that mean?"
Since this question is leading off the riboflavin section, you have probably surmised that riboflavin is somehow involved. Indeed, flavin means yellow in Latin, and riboflavin is bright yellow as shown in Figure \(\PageIndex{1}\).

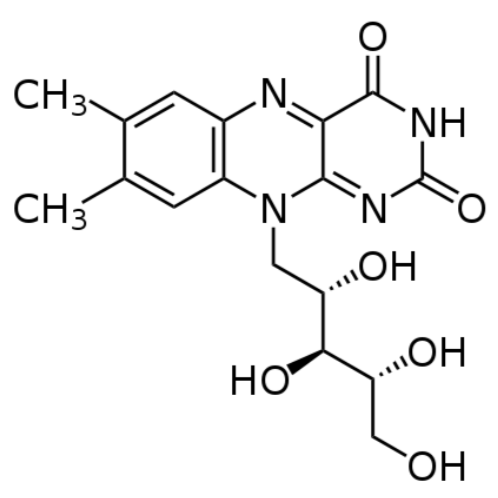
Riboflavin is a water-soluble B vitamin, so the student was excreting large amounts of riboflavin in his urine, leading it to become "bright, practically neon yellow." The structure of riboflavin is shown in Figure \(\PageIndex{2}\).
Riboflavin is important for the production of two cofactors: flavin adenine dinucleotide (\(\ce{FAD}\)) & flavin mononucleotide (\(\ce{FMN}\)).
\(\ce{FAD}\) has been introduced before, but structurally you can see where riboflavin is within the compound below.
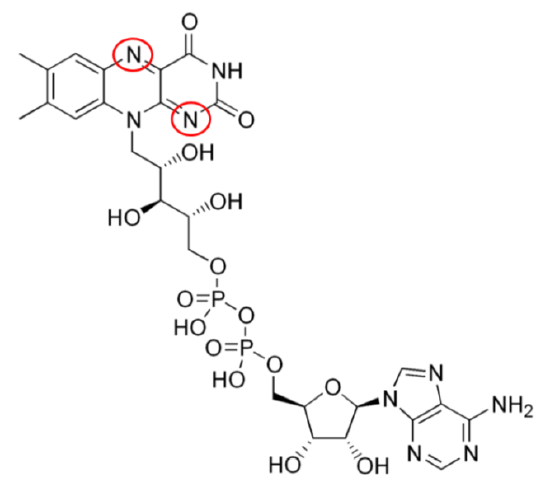
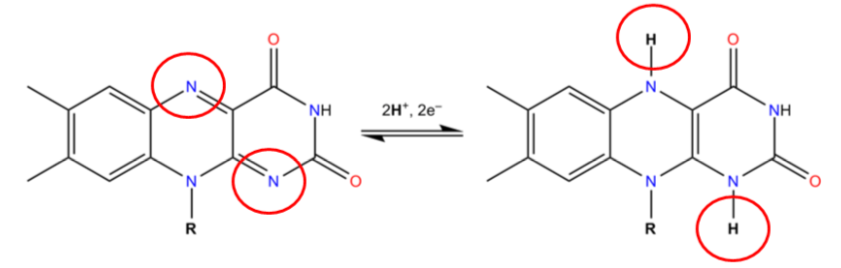
The 2 circled nitrogens are the sites that accept hydrogen to become \(\ce{FADH2}\) as illustrated below.
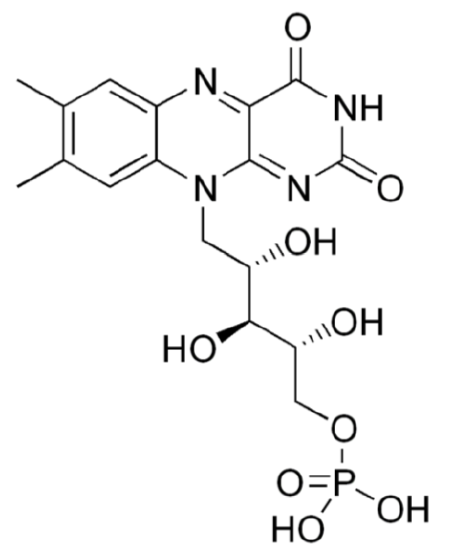
The structure of \(\ce{FMN}\) as shown below, is similar to \(\ce{FAD}\), except that it only contains one phosphate group (versus 2) and doesn't have the ring structures off the phosphate groups that are found in \(\ce{FAD}\).
Riboflavin is photosensitive, meaning that it can be destroyed by light. This was a problem in the old days when the milkman delivered milk in clear glass bottles. These have now been replaced by cartons or opaque plastic containers to help protect the riboflavin content of the milk.

Riboflavin in foods is free, protein-bound, or in \(\ce{FAD}\) or \(\ce{FMN}\). Only free riboflavin is taken up so it must be cleaved, or converted before absorption6. Riboflavin is highly absorbed through an unresolved process, though it is believed that a carrier is involved7. As you would guess from the description above, riboflavin is primarily excreted in the urine.
Query \(\PageIndex{1}\)
Riboflavin Functions
Riboflavin is required for the production of \(\ce{FAD}\) and \(\ce{FMN}\). Below are some of the functions of \(\ce{FAD}\) and \(\ce{FMN}\)6:
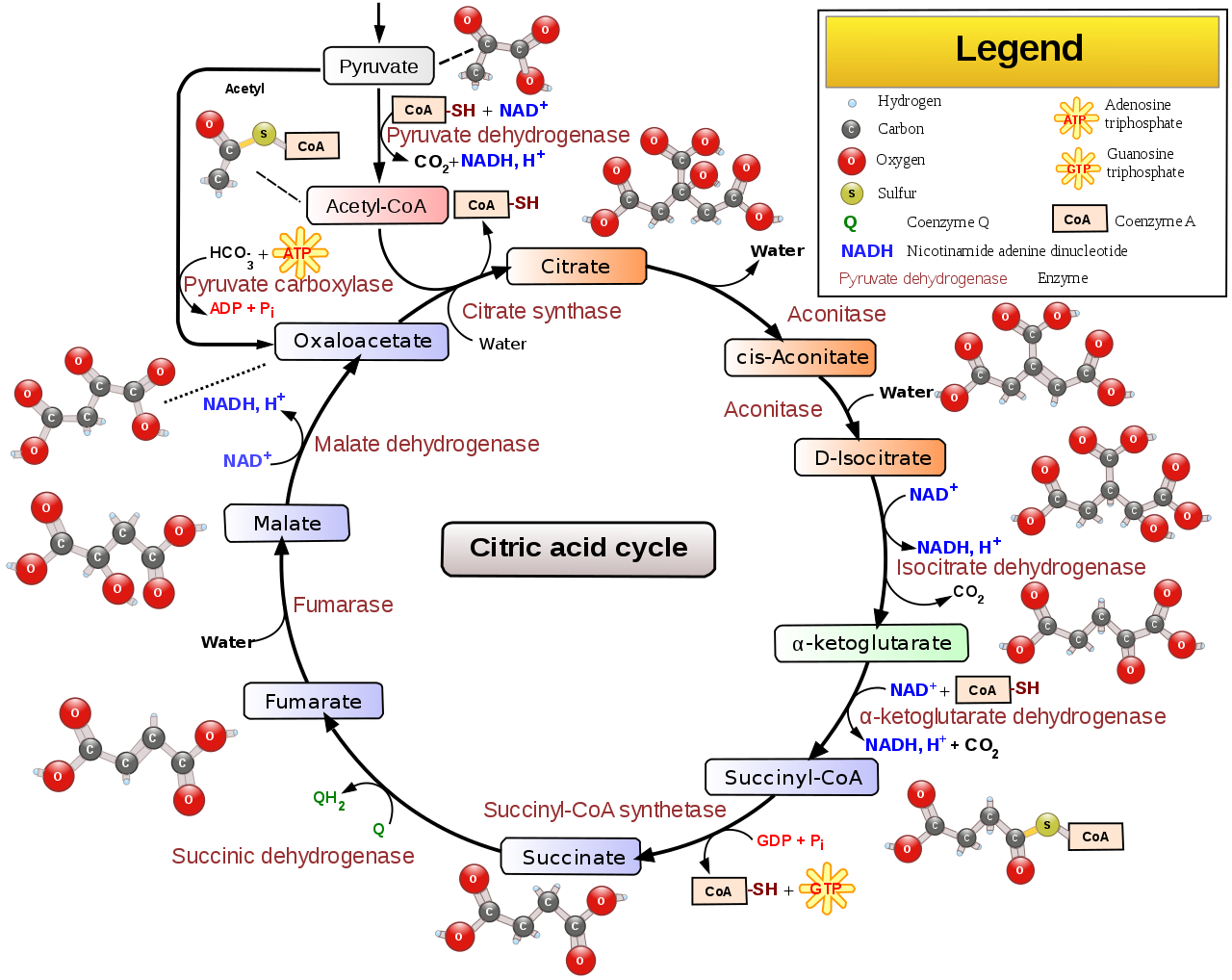
Citric Acid Cycle
\(\ce{FAD}\) is reduced to \(\ce{FADH2}\) in the citric acid cycle when succinate is converted to fumarate by succinic dehydrogenase as circled below.
Electron Transport Chain
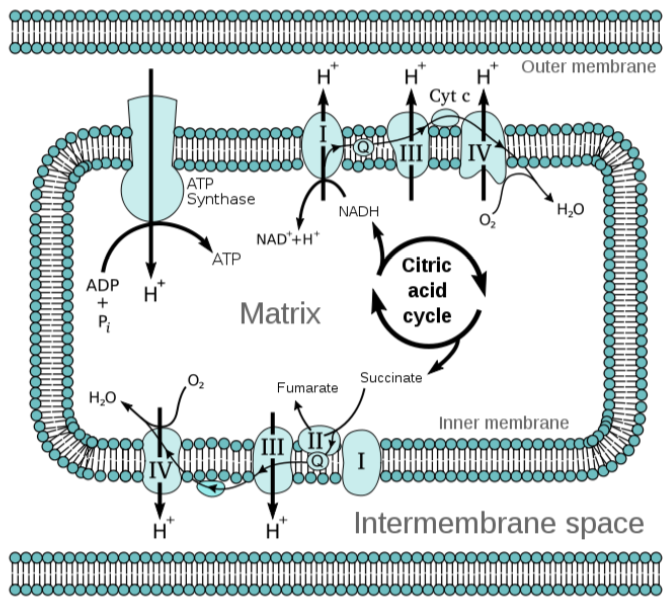
Under aerobic conditions, the electron transport chain is where the \(\ce{FADH2}\) is used to produce ATP. Complex I of the electron transport chain includes an \(\ce{FMN}\) molecule. The electron transport chain is shown below.
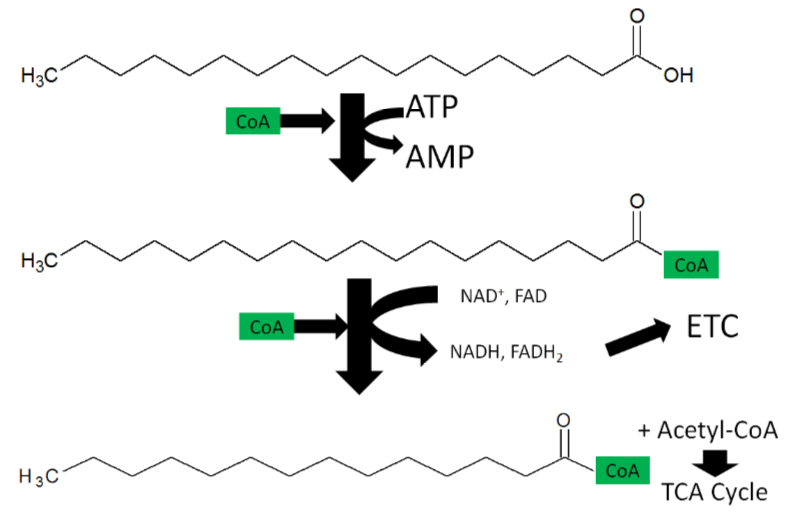
Fatty Acid oxidation
During fatty acid oxidation \(\ce{FAD}\) is converted to \(\ce{FADH2}\) as shown below.
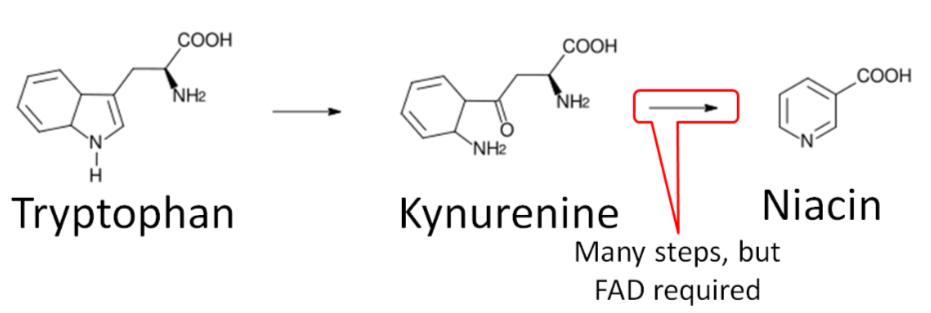
Niacin synthesis
As you will hear more about in the niacin section, niacin can be synthesized from tryptophan as shown below. An intermediate in this synthesis is kynurenine, and one of the multiple steps between kynurenine to niacin requires \(\ce{FAD}\).
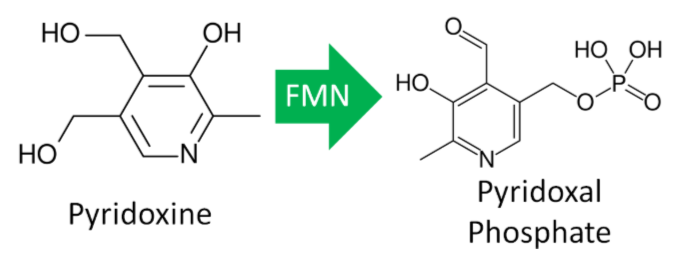
Vitamin \(B_6\) Activation
The enzyme that creates the active form of vitamin \(B_6\) (pyridoxal phosphate) requires \(\ce{FMN}\).
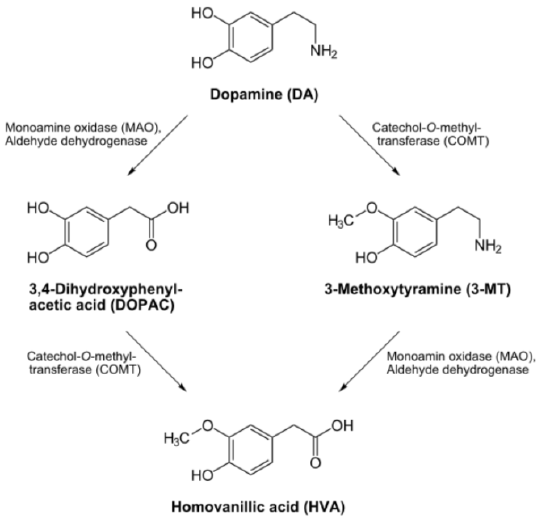
Neurotransmitter Catabolism
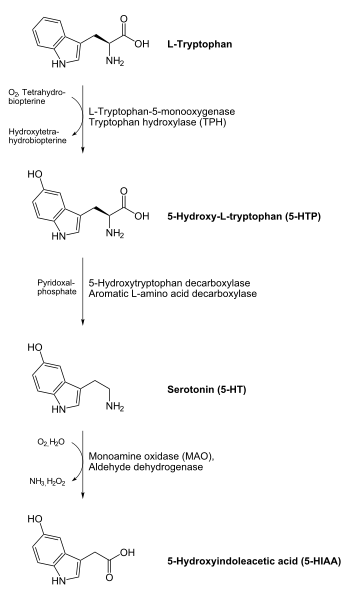
The enzyme monoamine oxidase (\(\ce{MAO}\)) requires \(\ce{FAD}\). This enzyme shown below is important in the catabolism of neurotransmitters such as dopamine and serotonin.
Antioxidant Enzymes
The antioxidant enzymes glutathione reductase and thioredoxin reductase both require \(\ce{FAD}\) as a cofactor. Thioredoxin reductase is a selenoenzyme. The function of glutathione reductase is shown in the following link. Glutathione reductase can reduce glutathione that can then be used by the selenoenzyme glutathione peroxidase to convert hydrogen peroxide to water.
In addition to the functions listed above, \(\ce{FAD}\) is also used in folate activation, choline catabolism, and purine metabolism6.
Query \(\PageIndex{2}\)
Riboflavin Deficiency & Toxicity
Ariboflavinosis, riboflavin deficiency, is a rare condition that often occurs with other nutrient deficiencies. The symptoms of this condition are shown in the figure below.
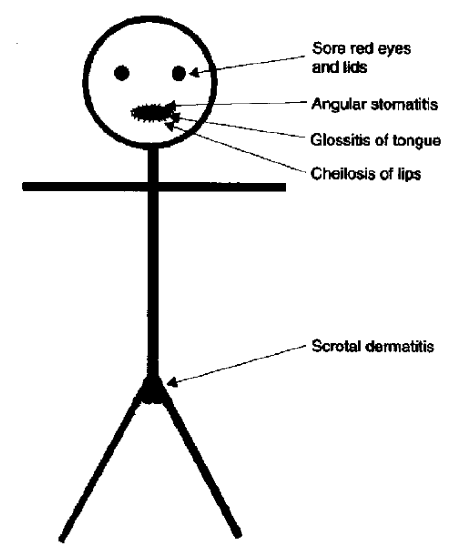
The most notable symptoms include angular stomatitis (aka angular cheilitis, cheilosis), which is a lesion or cracking that forms at the corners of the mouth as shown below.
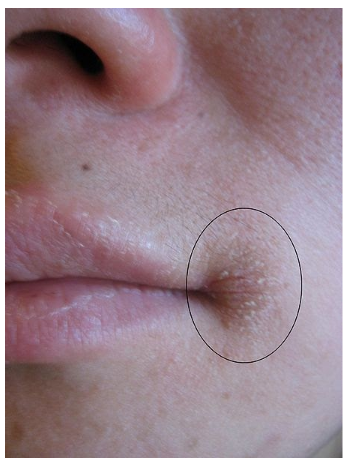
Glossitis is the inflammation of the tongue, which can be accompanied by redness or inflammation of the oral cavity. Dermatitis (skin inflammation) is also frequently a symptom6,17. Rare reports of riboflavin toxicity has resulted in optic problems18.
Query \(\PageIndex{3}\)
References
- http://www.chm.bris.ac.uk/motm/vitam...n-solution.jpg
- en.Wikipedia.org/wiki/File:Riboflavin.svg
- https://courses.lumenlearning.com/suny-nutrition/chapter/6-11-cofactors/
- https://courses.lumenlearning.com/suny-nutrition/chapter/6-11-cofactors/
- https://en.Wikipedia.org/wiki/Flavin_mononucleotide#/media/File:Flavin_mononucleotide.png
- Gropper SS, Smith JL, Groff JL. (2008) Advanced nutrition and human metabolism. Belmont, CA: Wadsworth Publishing.
- Said H, Mohammed Z. (2006) Intestinal absorption of water-soluble vitamins: An update. Curr Opin Gastroenterol 22(2): 140-146.
- https://en.Wikipedia.org/wiki/Citric_acid_cycle#/media/File:Citric_acid_cycle_with_aconitate_2.svg
- https://en.Wikipedia.org/wiki/Electron_transport_chain#/media/File:Mitochondrial_electron_transport_chain%E2%80%94Etc4.svg
- https://courses.lumenlearning.com/suny-nutrition/chapter/6-32-fatty-acid-oxidation-beta-oxidation/
- https://courses.lumenlearning.com/suny-nutrition/chapter/10-41-riboflavin-functions/
- https://courses.lumenlearning.com/suny-nutrition/chapter/10-7-vitamin-b6/
- https://courses.lumenlearning.com/suny-nutrition/chapter/10-41-riboflavin-functions/
- https://courses.lumenlearning.com/suny-nutrition/chapter/10-41-riboflavin-functions/
- https://courses.lumenlearning.com/suny-nutrition/chapter/10-42-riboflavin-deficiency-toxicity/
- https://www.medicalnewstoday.com/articles/320053#what-is-angular-cheilitis
- Byrd-Bredbenner C, Moe G, Beshgetoor D, Berning J. (2009) Wardlaw's perspectives in nutrition. New York, NY: McGraw-Hill.
- Pinto JT, Zempleni J. (2016) Riboflavin. Advances in Nutrition 7(5): 973–975.


