4.2: The Basic Equation of Photosynthesis
- Page ID
- 56181
Joseph Priestley was a somewhat radical English minister and politician, who was eventually forced to emigrate to the United States for political and religious sanctuary. But he’s best remembered for his secondary interest, chemistry, which led to the untangling of some of the first mysteries of photosynthesis.
One day in 1780, Priestley lit a candle that stood in a shallow dish of water. He inverted a jar over the candle, the water sealing the jar from the outside air. He was limiting the ability of the candle to keep burning, since a candle burns by oxidation. The carbon in the wax combines with oxygen from the air and the reaction produces heat and light.
The inverted jar was sealed, so the candle burned only as long as oxygen remained in the jar. As the oxygen was exhausted, the candle flickered and went out. Air, of course, is a mixture of several gases, mainly nitrogen. But of these gases, only the oxygen could support the flame.
In the burning, the oxygen from the air combined with the carbon of the candle wax to yield carbon dioxide. What Priestley now had was a dead candle in an atmosphere of carbon dioxide and some other gases.
He then did one more thing. He placed a sprig of mint in the oxygen-free jar, with its stem in water. The mint could receive light through the glass, and water through its stem. A few days later, Priestley tested the gas from the inverted jar and found “…that the air would neither extinguish a candle, nor was it at all inconvenient to a mouse which I put into it.”
In other words, somehow the living sprig of mint had used light and water and turned the carbon dioxide back into oxygen again.
Within three years, French scientist Antoine Lavoisier had grasped the basics of the other side of the equation. Lavoisier found that animal life used up oxygen (just as Priestley’s candle did) and gave off carbon dioxide (in which Priestley’s mint had grown). In the process, heat was given off, as it was by the candle.
By 1800 it was becoming clear—with parts of the puzzle coming from scientists in many countries—that there was a balance between plants and animals, and that that balance involved the use of energy.
Dutch scientist Jan Ingenhousz had followed Priestley’s lead and reported, “I observed that plants not only have the faculty to correct bad air…by growing in it…but that they perform this important office in a complete manner in a few hours; that this wonderful operation is by no means owing to vegetation of the plant, but to the influence of light of the sun upon the plant.”
Closing the ring, Swiss scientists showed that both carbon dioxide and water were used up by plants during photosynthesis. And finally, in 1842, German surgeon Julius Mayer drew the correct fundamental conclusion about how it all worked. It was simple. He explained it in one sentence: “The plants take in one form of power, light; and produce another power, chemical difference.”
The Chemical Birth of Food
It can be said that all our food begins with plants. For example, when we drink milk or eat beef, we get the energy that the cow stored by eating grass and other vegetable matter. And that vegetable matter got this energy from the sun— by a process called photosynthesis, which means “putting together with light.”
The light in this case is, of course, the light of the sun (“solar energy”). What this light links together are two chemicals which the earth has in abundance—carbon dioxide and water. If our body could put these two chemicals together itself, we could create our own energy sources.
But it can’t. We could sit all day in the sun, taking in both water and carbon dioxide by sipping carbonated beverages, and we’d end up with only a sunburn.
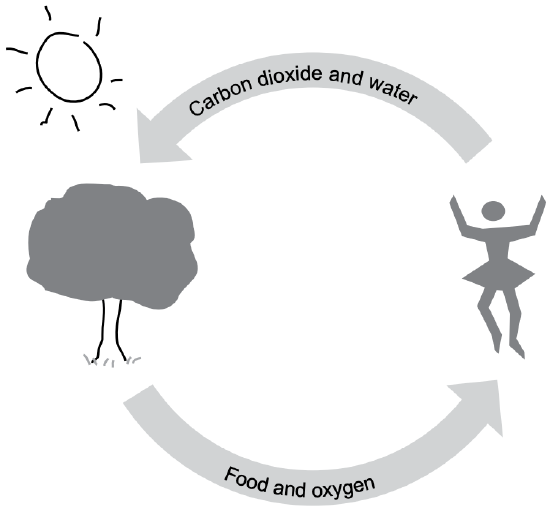
But plants have something we don’t have—the phenomenal green chemical, chlorophyll. In the presence of chlorophyll, plants can use the sun’s energy to combine carbon dioxide and water.
So plants combine the three atoms found in carbon dioxide (CO2) and the three found in water (H2O), and use solar energy to reassemble them. They end up with two products: two of the oxygens join together to become oxygen gas. What’s left is a combination of one carbon, two hydrogens, and a single oxygen. In effect, the carbon has been combined with water (C-H2O). The carbon has been hydrated—thus carbohydrate.
Photosynthesis’s juggling act is:
H2O + CO2 + Energy = C-H2O + O2
This hydrated carbon (C-H2O) is combined with other hydrated carbons into the many forms of a plant’s carbohydrate family. The carbohydrate form that’s a major source of energy for us is starch—a carbohydrate that has thousands of these hydrated carbon atoms. We, in fact, get energy from starch by “undoing” starch back into carbon dioxide and water (see Fig. 4-1).
Hydrate: To combine with water
It could be said that we are solar-powered, since sunlight is the original source of the energy in the food we eat.
In nature, CO2 in the atmosphere is kept in balance by animal life making CO2, and plant life using CO2. This balance can be upset by more CO2 produced (e.g., more people; burning of gasoline, wood, coal) and less CO2 removed (e.g., destroying forests). CO2 is a “greenhouse gas” in that it traps solar heat. An increasing amount of greenhouse gases causes global warming—the warming of the earth’s surface and lower atmosphere— that can be ecologically disastrous.
Most people “know” about photosynthesis. Yet, ask how a small acorn becomes a majestic oak tree. Where does that huge mass come from? Even college graduates often answer, “from the soil.” It’s quite incredible that the bulk of the tree comes from carbon dioxide in the air (with the help of water and sun, of course; see Fig. 4-1).


