1.17: Laryngectomy - Vertical Partial
- Page ID
- 17628
OPEN ACCESS ATLAS OF OTOLARYNGOLOGY, HEAD & NECK OPERATIVE SURGERY
VERTICAL PARTIAL LARYNGECTOMY
Jonas Johnson
Management of small tumors involving the true vocal folds can be contentious. Tumor control is achieved in over 95% of patients presenting with T1 glottic cancer employing external beam radiation therapy, vertical hemilaryngectomy, or transoral endoscopic resection. Given adequate resources, the weight of evidence favors endoscopic transoral resection as the most cost-effective approach with very high rates of tumor control.
Patient Selection
Young patients presenting with involvement of the anterior commissure or partial impairment of motion (T2) and those who have failed attempts at curative irradiation therapy for small glottic lesions may be suitable candidates for vertical partial laryngectomy.
When the tumor requires removal of the anterior commissure, the proper term for this procedure is frontolateral partial laryngectomy. Frontolateral vertical hemilaryngectomy can be accomplished to include one arytenoid and, when necessary, both true vocal folds. Under these circumstances, some surgeons may choose supracricoid partial laryngectomy with cricohyoidoepiglottopexy to manage extensive cancer of the anterior commissure involving both vocal cords and the paraglottic space.
Vertical partial laryngectomy is not suitable for tumors involving the supraglottic larynx and the paraglottic space. Similarly, it is not appropriate for patients with greater than 10 mm of subglottic extension. Involvement of both arytenoids is also a strict contraindication.
Patients presenting with multifocal lesions, diffuse dysplasia and carcinoma in situ, involvement of the arytenoids and the posterior commissure, and those in whom poor health compromises suitability for surgery may be better served with external beam irradiation.
Preoperative Planning
When considering surgical intervention for cancer involving the true vocal cords, precise staging and patient selection are crucial for good outcomes. High resolution fine-cut CT compliments microlaryngoscopy and examination under anesthesia.
In the setting of failed prior irradiation therapy, the surgeon must be aware of the potential for multifocal persistent cancer. In general, the entire field of the original tumor should therefore be considered for removal.
Careful preoperative tumor mapping employing flexible fiberoptic endoscopy and fine cut CT allows the surgeon to estimate the extent of resection and the reconstructive needs of each individual patient. For instance, patients requiring frontolateral hemilaryngectomy in which both arytenoids are intact and 80% of one vocal cord can be maintained will require no added tissue for reconstruction. Conversely, if an arytenoid is resected or if more than 20% of the contralateral vocal fold must be removed, some reconstruction must be undertaken to afford good swallowing without aspiration and to allow adequate diameter of the lumen for respiration.
Operative steps
The procedure is undertaken under general anesthesia. Perioperative prophylactic antibiotics are administered. Tracheotomy is accomplished through a separate incision. A second horizontal incision is made at approximately the level of the thyroid notch (Figure 1).
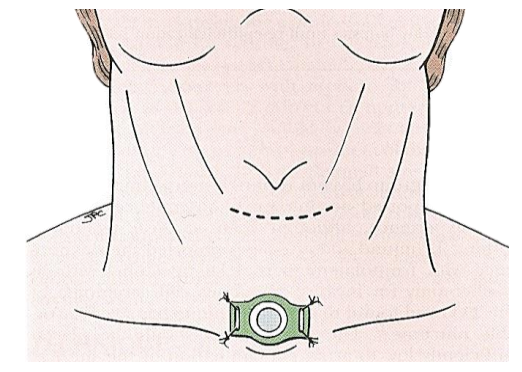
Figure 1: Incision for vertical partial laryngectomy. Care should be taken to ensure separation of the tracheostomy incision from that required for the partial laryngectomy
Skin flaps are elevated superiorly to the hyoid and inferiorly to the cricoid cartilage. The strap muscles are then elevated, exposing the thyroid lamina.
The external perichrondrium of the thyroid cartilage is incised at the midline and a posteriorly-based perichrondrial flap is elevated bilaterally (Figures 2 & 3). The extent of the elevation should reflect the intended thyroid cartilage resection.
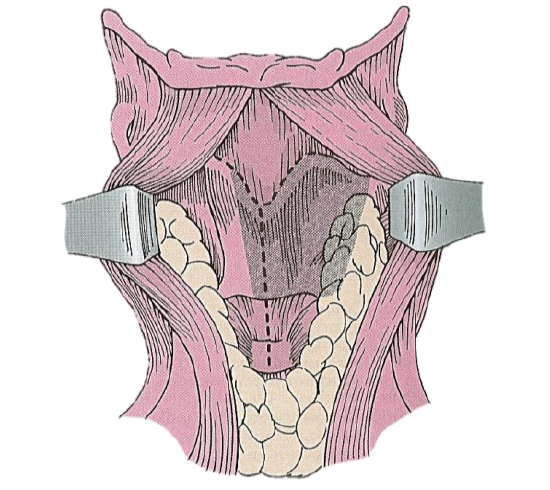
Figure 2: Incision of the perichondrium to expose the thyroid ala
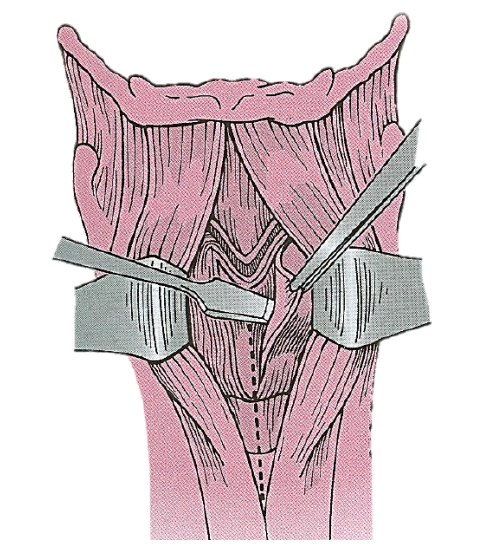
Figure 3: Elevation of the perichondrium with the overlying strap muscles
Vertical cuts are then made through the thyroid cartilage, maintaining as much of the posterior lamina of the cartilage as basic oncologic principles will allow (Figure 4).
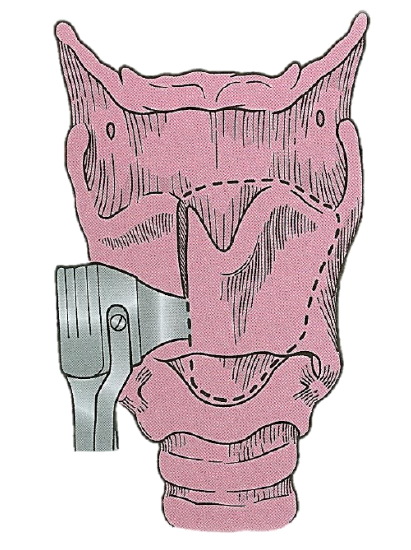
Figure 4: The vertical cartilage cuts are designed to correspond with soft tissue resection margins within the larynx
It is essential that the cartilage cuts be made perpendicular to the plane of the cartilage and that soft tissue cuts be made with a knife (not with a saw) (Figure 5).
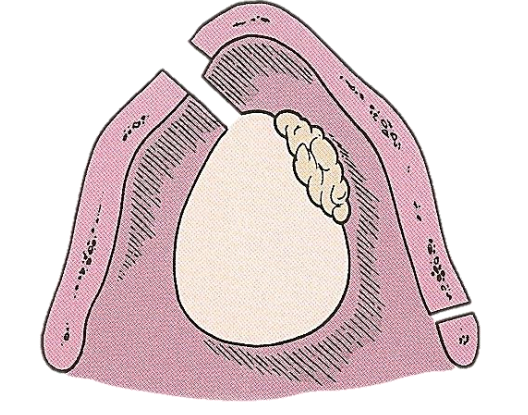
Figure 5: The cartilage cuts must be perpendicular to the cartilage to allow for corresponding soft tissue cuts in the larynx
A wide cricothyrotomy allows access to the subglottis such that the surgeon working with a headlight and a knife can visualize the undersurface of the vocal cords and to place the initial cut to allow a 2 mm margin from the tumor without unnecessarily removing uninvolved vocal fold. The thyrotomy is then opened widely such that the cut across the posterior aspect of the involved vocal fold can be accomplished with a 2 mm margin and the specimen removed. New margins from both sides of the vocal cord should be submitted for frozen section assessment. A nasogastric tube is introduced and fixed to the nasal septum with a suture.
When two-thirds or more of one vocal fold has been preserved, no extra soft tissue reconstruction is required. The free end of the residual vocal fold must be fixed anteriorly to the residual thyroid cartilage with a vicryl suture (Figure 6).
Reconstruction is accomplished by closing the external perichondrium in the midline (Figure 7). The strap muscles are reapproximated in the midline as the second layer of closure.
A Penrose drain is inserted. Suction drains should be avoided because they will tend to pull secretions through the wound. The skin is reapproximated in layers and a cuffed tracheotomy tube is introduced.
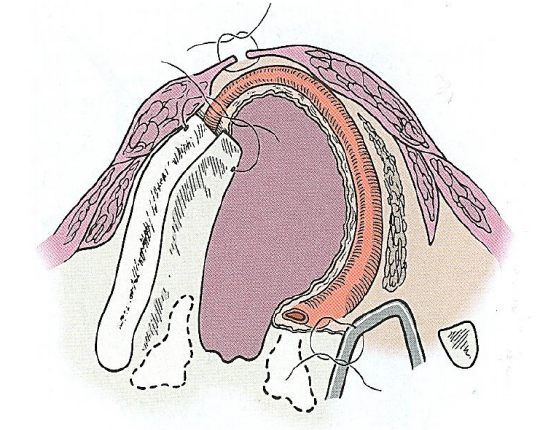
Figure 6: Reattachment of the vocal cord remnant to the thyroid ala
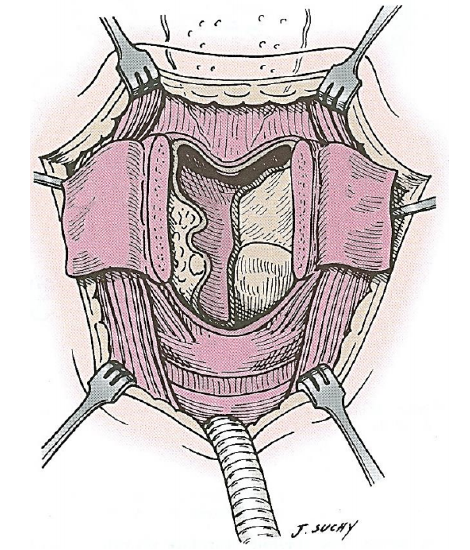
Figure 7: The remaining larynx after the specimen has been removed. Closure is initiated by suturing the external perichondrium in the midline
Reconstruction Considerations
When more than 80% of the contralateral vocal fold cannot be maintained, some form of tissue reconstruction is required to reconstitute the lumen to maintain an adequate airway. I prefer employing an epiglottopexy. The petiole of the epiglottis is grasped with an Allis forceps, and the hyoepiglottic ligament freed such that the epiglottis can be mobilized and pulled inferiorly and fixed to the superior surface of the cricoid cartilage (Figures 8 & 9).
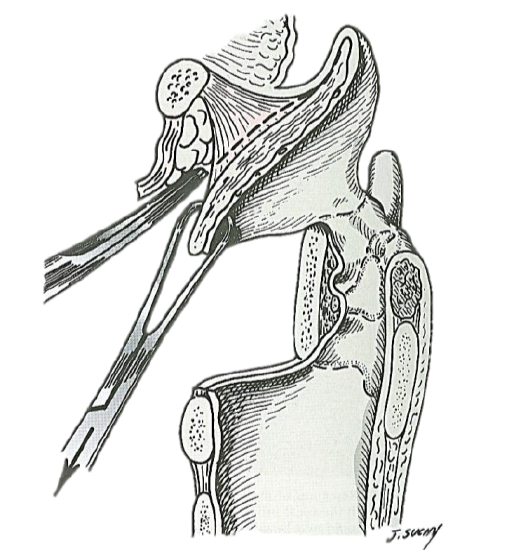
Figure 8: Epiglottopexy: The lingual surface of the epiglottic cartilage is undermined in the subperichondrial plane to divide the hyoepiglottic ligament
When tumor considerations require removal of the body of the arytenoid, it, too, must be reconstructed. The critical aspect is to re-establish the height of the arytenoid to prevent secretions from flowing directly from the pharynx into the larynx. A multitude of techniques have been reported employing cartilage, muscle or just fat. The tissue needs to be fixed to the top of the cricoid and then covered by advancing mucosa from the medial wall of the pyriform sinus over the reconstructive materials. Patients requiring arytenoid reconstruction invariably have prolonged recovery and temporary dysphagia.
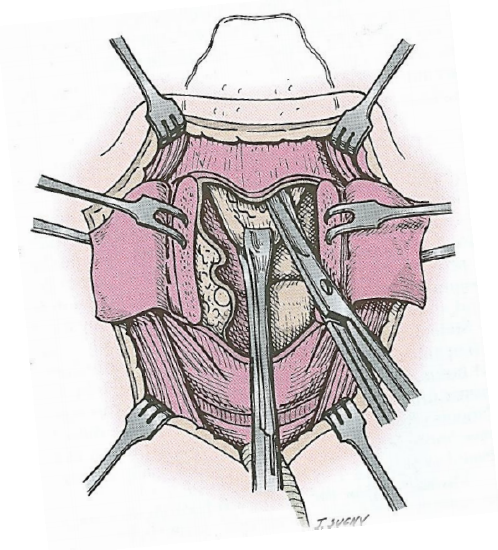
Figure 9: Epiglottopexy: The epiglottis is pulled inferiorly
Postoperative Care
At completion of the procedure, the patient should have a cuffed tracheotomy tube in place with the balloon inflated. Antibiotic administration is terminated on the first postoperative morning. The patient’s nutritional needs are supported by feeding through a nasogastric tube.
The dressing supporting the Penrose drain should be changed for good hygiene. The drain can be removed when it is no longer producing mucoid secretions. Ordinarily that occurs on the third or the fourth postoperative day.
Routine tracheotomy care is provided. The tracheotomy cuff should be maintained inflated for 4-5 days to divert coughed secretions through the tracheotomy tube rather than into the newly reconstructed wound.
On the fifth postoperative day, the tracheotomy cuff can be deflated. This allows the treatment team to determine if the patient can protect his own airway without aspiration. Patients who have previously been treated with radiation therapy may have a delayed decannulation process. If the patient tolerates deflating the cuff on the tracheotomy tube, the treatment team can change the tracheotomy tube to a smaller, uncuffed tube. The adequacy of the reconstructed airway can be tested by plugging the tracheotomy tube. Patients who tolerate this overnight are candidates for decannulation. Soft foods are reintroduced, and the patient can be discharged.
Salvage following radiation: Special considerations and problems
The most common call for frontolateral partial hemilaryngectomy in the era of transoral endoscopic technology is to treat patients with persistent cancer after having had radiation therapy. Such patients have increased edema, slower healing, and a high incidence of chondritis. The drain frequently needs to be maintained beyond the first week. Antibiotic administration should be instituted for cellulitis and erythema. Return to oral eating is often delayed and the nasogastric tube may need to be maintained for 2-3 weeks. Under these circumstances, most patients can be discharged to home with home nursing and care of the tracheotomy and nasogastric tube until the wound has stabilized, edema has resolved, at which time the treatment team can proceed with the decannulation and oral feeding process.
Therapeutic Expectations
Partial vertical hemilaryngectomy is highly effective (>90%) in properly selected patients. Surgical removal of previously irradiated tumors always has the potential that multifocal tumor is overlooked and if not completely removed, will recur.
All patients having open partial laryngectomy have dysphonia postoperatively. The overwhelming majority of patients do however have a serviceable voice.
Longstanding dysphagia and aspiration are unusual if both arytenoids are preserved or, when one arytenoid has been removed, it has been appropriately reconstructed.
Failure to properly reconstruct the glottis after resection of most of both vocal folds will result in laryngeal stenosis. Prevention is the key to relief of this problem. The treatment team must recognize the need for extra soft tissue and incorporate the reconstruction at the time of resection. Reconstructing the stenotic larynx is beyond the scope of this chapter.
Useful reference
https://developingworldheadandneckca...ottic-cancers/
Author
Jonas T. Johnson, M.D.
Eugene N. Myers
Professor & Chairman
Department of Otolaryngology
Eye and Ear Institute
200 Lothrop Street Suite
500, Pittsburgh, PA 15213, USA
johnsonjt@upmc.edu
Editor
Johan Fagan MBChB, FCS (ORL), MMed
Professor and Chairman
Division of Otolaryngology
University of Cape Town
Cape Town, South Africa
johannes.fagan@uct.ac.za

The Open Access Atlas of Otolaryngology, Head & Neck Operative Surgery by Johan Fagan (Editor) johannes.fagan@uct.ac.za is licensed under a Creative Commons Attribution - Non-Commercial 3.0 Unported License



