5.11: Facial Nerve Grafting- Indications and Techniques
- Page ID
- 53282
OPEN ACCESS ATLAS OF OTOLARYNGOLOGY, HEAD & NECK OPERATIVE SURGERY
FACIAL NERVE GRAFTING: INDICATIONS AND TECHNIQUES
Vincent Darrouzet & Erwan de Mones del Pujol
The facial nerve is at risk in trauma of the temporal bone and face. It can also be harmed in surgery for tumors of the parotid, temporal bone or cerebellopontine angle (CPA). Often it is only a transient injury, and purely inflammatory in nature. But sometimes, despite the progress of otoneurosurgery and technical refinements limiting the risk of severe damage to a healthy nerve, and especially because some lesions interrupt or invade the nerve, the surgeon may have to manage a loss of actual nerve tissue. In either situation, nerve graft is the best solution, provided that it is technically possible and that it is carried out according to the very precise rules that we describe in this chapter. It should be noted that the technique used depends on the site and the nature of the trauma.
1. Influence of the site of the lesion
The site of the lesion is a key element when electing a strategy when a nerve graft is required. The approach also differs depending on whether the lesion involves 1 or more of the 3 distinct regions of the nerve’s course
- CPA and internal auditory meatus (IAM)
- Temporal bone
- Parotid
Making this distinction is important because in the CPA and IAM, the nerve does not have an epineurium covering. It is therefore technically impossible to perform needle sutures without transfixing the nerve fibers and causing axonal loss and fibrosis that may impede nerve regrowth. Sutures are also very difficult to place at this level, because of the narrowness and depth of access, the pulsatility of the area and the immersion in cerebrospinal fluid (CSF). In the temporal bone and parotid, conditions are more favorable because the nerve is more superficial and accessible. It is also protected by an increasingly strong and defined epineurium that serves as an excellent anchor for sutures.
The mobility of the nerve in the anatomical regions in which the graft must be performed must also be taken into account. The nerve in the parotid region is subject to movements of the mandible ; hence the nerve should be sutured when grafting in this region. On the other hand, in the temporal bone, a technique of fibrin-bonding can be used because the nerve repair will remain stable due to lack of movement.
2. Expected results
A well performed nerve transplant should provide a satisfactory House Brackmann Grade 3 facial mobility at one year postoperatively, regardless of the region repaired, even in the CPA where the surgery is the most challenging1. There is one caveat to this good result: recovery of frontalis muscle mobility is always disappointing, not that reinnervation does not take place, but because of the antagonistic competition between recovery of the frontalis muscle and the rhizorius. which is always at the expense of the first. This is referred to as the paradox of Stennert2, and mirrors our own experience.
To achieve good results it is necessary to operate as early as possible after the nerve injury, and if possible, immediately. After an interval of 1-2 years the results are much more disappointing. If one is compelled by circumstances to intervene beyond this time, it is probably preferable to apply other surgical techniques such as free muscle transfers or pedicles (See Chapter: Surgical reanimation techniques for facial palsy/paralysis).
Patients must be warned that functional recovery can take 4-10 months, depending on the location of the injury; proximal lesions take longer to recover. After this time further recovery may take 1-2 years. Surgery should be followed by rehabilitation, excluding any electrical stimulation.
In some situations it is possible to reroute the petrous and intraparotid facial nerve segments to gain additional length and achieve a direct, end-to-end, tension-free repair, thus avoiding the need for an interposition graft. However, such a diversion may be as time-consuming as harvesting and placing a graft, and mobilization of the nerve from the Fallopian canal can cause nerve ischemia and hence disappointing results. In practice, an end-to-end repair does not result in a better functional result than a well-done nerve graft3.
3. Five prerequisites for grafting
Nerve grafting is only feasible and effective under certain conditions:
- There should be no tension along the suture line. Failure to adhere to this principle leads to poor outcomes.
- The proximal end of the nerve must be perfectly healthy. To be sure, it is advisable to transect the nerve in a clearly healthy area, even if this leads to a longer graft. This situation generally does not apply in tumor resections in the CPA, because of difficulties related to compression and edema of the nerve. The nerve endings must be histologically free of tumor invasion on frozen section.
- A traumatic neuroma must be resected widely. The proximal end of the nerve must be transected in a healthy area.
- The graft must be placed in a vascularized area. This is a principle that is often neglected, and explains many poor outcomes. In the temporal bone in particular, the graft needs to be placed in contact with the bone that provides vascular support. It must not be surrounded by fat as it will become necrotic or move as the fat resorbs.
- It should not be delayed too long; this will be referred to again later.
4. Indications for grafting
4-1 Temporal bone trauma (See Chapter: Temporal bone trauma)
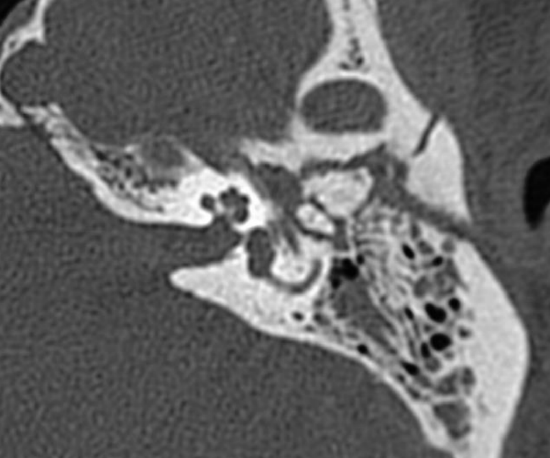
Figure 1: CT scan of left temporal bone with extralabyrinthine comminuted fracture. The fracture line points to the geniculate ganglion
Temporal bone fractures are less frequent today. They are mostly longitudinal extralabyrinthine fractures (Figure 1).
The facial nerve is generally injured at the geniculate ganglion, and is severed by a bony spur. Grafts are difficult to place in a trauma situation due to contusion of the nerve endings, associated extradural hematoma and the delicateness of the labyrinthine portion of the nerve.
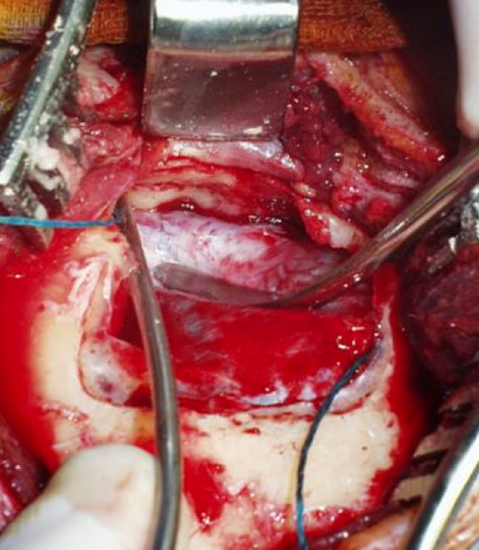
Figure 2: Right middle fossa craniotomy
A craniotomy is required for access (Figure 2). It allows good exposure of a sufficient length of the labyrinthine segment of the nerve without injuring the labyrinth, while ensuring access to transect the nerve in a healthy area to achieve a good result. However, it requires specialized otoneurosurgical training and expertise.
The situation is very different and simpler when the fracture is translabyrinthine, as the nerve is generally interrupted in the tympanic segment. A transmastoid, translabyrinthine approach is then simpler to use, and access is less restricted which makes it much easier to control the nerve endings.
4-2 Iatrogenic lesions in the temporal bone or parotid
The most common situations are the following:
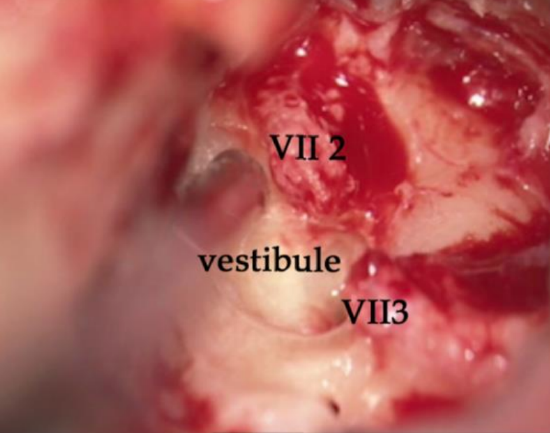
Figure 3: Iatrogenic lesion of the right facial nerve by a cutting burr associated with an accidental opening of the vestibule. Note the loss of nerve substance in the area over the vestibular cavity
- In the temporal bone, the nerve is usually damaged at the 2nd genu4. Injury with a cutting burr creates a substantial loss of nerve substance that can only be treated with a graft (Figure 3). In cases of thermal injury, which always has a poor prognosis, where the nerve has been transected is more important.
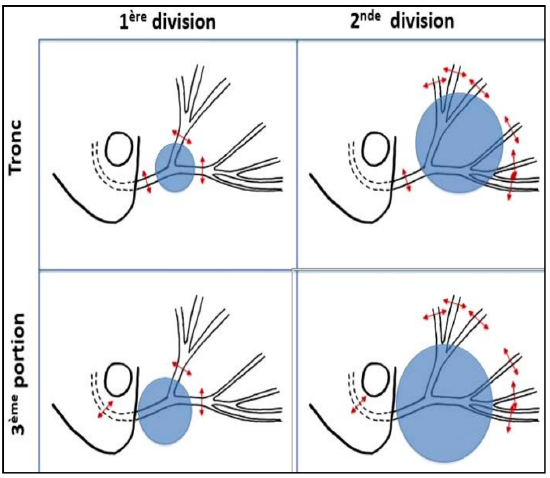
Figure 4: Different lesional locations along the intraparotid facial nerve.
- In the parotid, anything is possible. Injuries range from a localized lesion of the facial nerve trunk to loss of nerve affecting the first division of the nerve or beyond this to involve distal subdivisions of the nerve. When the lesion is near the stylomastoid foramen, it becomes necessary expose the nerve in the mastoid to permit proximal anastomosis of the graft (Figure 4).
4-3 Cancer of temporal bone and parotid
Nerve reconstruction should not be ignored when treatment is with curative intent and the excision follows oncological principles. The nerve should be resected to ensure tumor free nerve endings. This applies especially to adenoid cystic carcinoma as perineural invasion can even extend intracanially. In such cases it is advisable to be extremely cautious to secure clear margins.
4-4 Benign tumors or intermediate malignancies of the temporal bone, intrinsically or extrinsically affecting the facial nerve
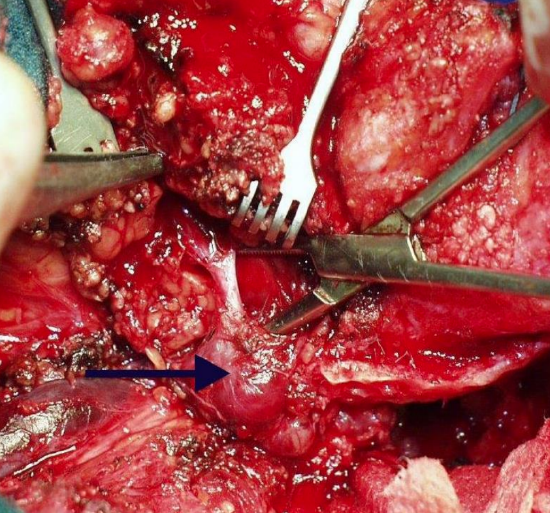
Figure 5: Neuroma of left facial nerve at stylomastoid foramen
This includes congenital cholesteatoma, jugulotympanic paraganglioma, adenocarcinomas, endolymphatic sac tumors, facial nerve neuromas and hemangiomas (Figure 5). Here again, it should be ensured that the nerve ends are free of tumor.
4-5 CPA tumors (schwannomas of vestibular and facial nerves, meningiomas, epidermoid cysts)
The most complex situation is that of neuromas arising from the facial nerve itself, as the discovery of the origin of the tumor may only be made intraoperatively, especially as the facial nerve is very rarely functionally impaired preoperatively. It is sometimes not possible to continue the surgery without interrupting the continuity of a functional nerve. Nerve monitoring helps to be sure. But often the nerve is already no longer continuous, and the decision to graft should be made during the procedure after a very precise assessment of the extent of the tumor along the nerve. Very often the nerve invasion extends within the temporal bone. In such cases the graft must be interposed between the proximal end of the nerve in the CPA and the healthy part of the intrapetrous nerve. Translabyrinthine and retrolabyrinthine approaches are the only ones that permit nerve grafting. The retrosigmoid approach makes it difficult to manage these situations.
We recommend planning for this possibility in any CPA tumor surgery by incorporating the upper cervical region in the surgical field to make the greater auricular nerve accessible as a graft. When the proximal end of the facial nerve in the CPA is not visible or is nonviable, the best alternative, even if it does not offer the same results, is to perform a hypoglossal-facial end-to-end or end-to-side anastomosis, the latter having the advantage of avoiding weakness of the tongue5. This intervention can take place at the same time or is most often delayed by 2-3 months depending on the general condition of the patient. (See Chapter: Surgical reanimation techniques for facial palsy/ paralysis).
5. Graft material
5-1 Greater auricular nerve
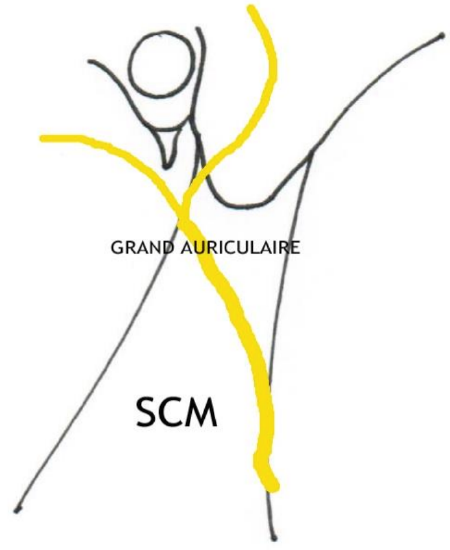
Figure 6: Diagram of the left great auricular nerve as it crosses over the sternocleidomastoid. An incision 1 to 2 cm below the mastoid tip will identify it before it divides
This is the most commonly used graft. It is conveniently closely situated to otological, otoneurosurgical and cervical approaches. The nerve is located 1 cm below the mastoid tip on the surface of the sternocleidomastoid muscle via a 4 cm transverse incision (Figure 6). Add 1 cm to the required graft length to permit trimming of the ends of the nerve graft. The greater auricualr nerve has the disadvantage of not providing a long length of graft because it divides quickly (<10 cm). Similarly, it is not advisable to use it in ipsilateral parotid or temporal bone cancers when the nerve is likely to be affected by the disease.
5-2 Sural nerve
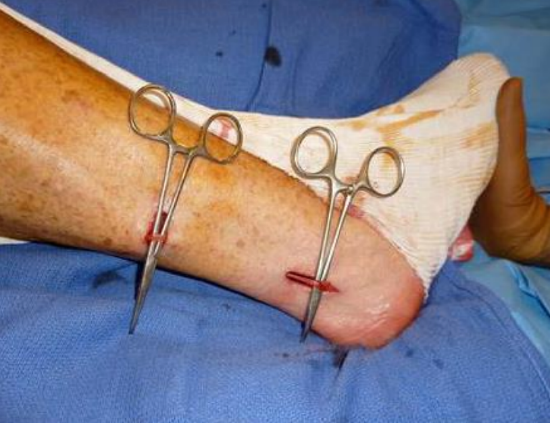
Figure 7: Finding and isolating the right sural nerve by a retromalleolar incision and a horizontal step incision in the leg
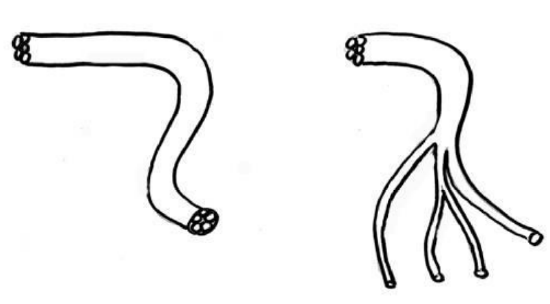
Figure 8: The fascicles of the sural nerve allows nerve subdivision particularly suited to parotid surgery
It is much less accessible and locating it is more difficult. It requires a 2nd operating field and its removal must be anticipated. It has two important advantages: it is a very fasciculated nerve that is easy to subdivide and a long length can be harvested as it divides late. These important benefits make it the preferred choice when it comes to placing split grafts in the parotid or temporal bone-to-parotid. It is located 2 cm behind the lateral malleolus, almost subcutaneously (Figure 7 and 8). It is possible to harvest it via horizontal incisions along the leg. Postoperatively the patient may have pain in the foot and leg causing transient functional impairment. It may be necessary to prescribe anticoagulants to avoid secondary venous thrombosis. Sensory deficits caused by harvesting the sural nerve are limited to the lateral edge of the foot and are not very troublesome.
6. Grafting technique
6-1 In the CPA
In the CPA, the authors favor a fibrin-bonding technique with an "aponeurotic sleeve". This replaces suturing, and provides much better results.
- Harvest a 4 cm temporalis fascia graft to wrap the "sutures"

Figure 9: Temporalis fascia spread and divided for fibrin-bonding
- Carefully spread the fascia on a metal plate and divide it into two pieces of equal size (Figure 9)
- Identify the proximal end of the facial nerve
- Carefully clean and cut the nerve. If necessary, bevel it to increase the anastomotic surface
- Ensure that the nerve is perfectly healthy, undamaged and tumor free
- Do the same for the nerve graft, and bevel it at both ends
- The nerve graft must be long enough to be positioned without tension and to make good contact with the petrous bone which must provide vascularization
- Before grafting, ensure perfect hemostasis in the CPA
- One of the two fragments of fascia, which serves as the bed of the graft, is then spread across the nerve defect
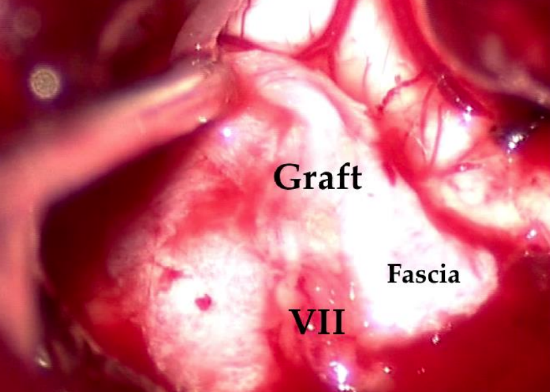
Figure 10: Perfect apposition of the proximal tip of the facial nerve and graft on a bed of fascia positioned across the defect
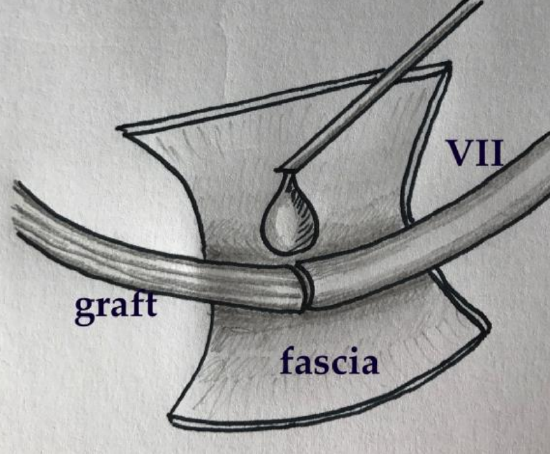
Figure 11: Diagram illustrating the fibrin-bonding and wrap technique used in CPA
- With the greatest precision possible and using fine forceps, place the ends of the facial nerve and graft in contact with one another (Figure 10).
- Use 2 drops of biological glue to secure the assembly (Figure 11)
- Fold the fascial graft over the nerve junction to wrap and protect it
- Glue is again used to secure the repair in this pulsatile environment
- A Surgicel® sheet, also glued to the area of repair, may be used as an additional layer
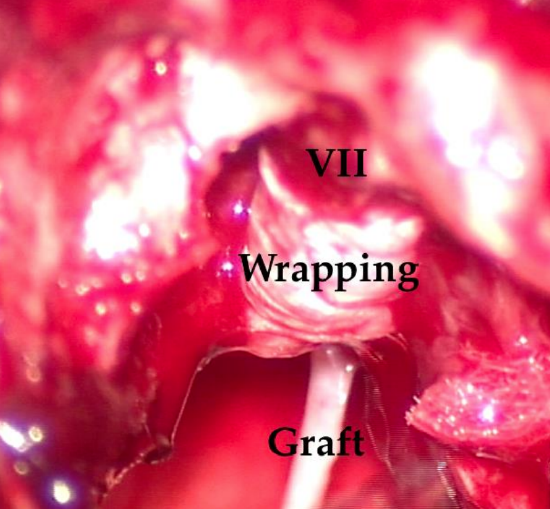
Figure 12: Fibro-bonding of the distal end of the graft and the distal end of the facial nerve to the medial bony wall of the internal auditory meatus (left translabyrinthine route)
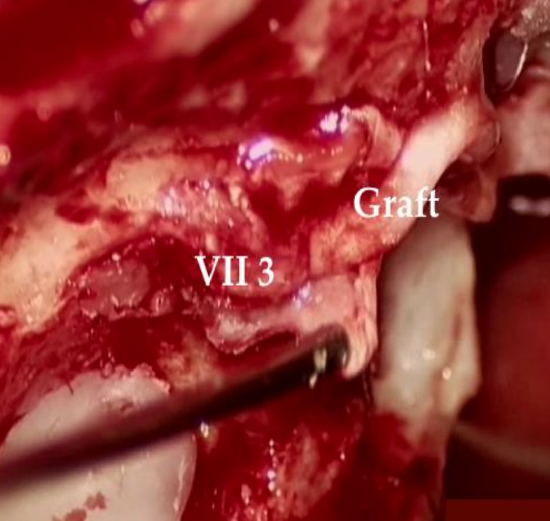
Figure 13: Distal fibrin-bonding in a case of facial nerve neuroma invading the geniculate ganglion and the initial portion of the tympanic portion of the facial nerve. The graft is positioned here in contact with the mastoid portion of the facial nerve (left translabyrinthine pathway)
- The distal anastomosis is done using the same steps, and wrapped with the second fragment of fascia (Figure 12).
- If the tumor extends in the nerve along the Fallopian canal, the graft is joined to the distal branch in the temporal bone, and is laid in a bony canal created to provide good stability and vascular support (Figure 13). This technique is quick and much simpler to implement in this "hostile" environment (narrow approach, pulsation, LCS outcome) than using conventional sutures
6-2 In the mastoid
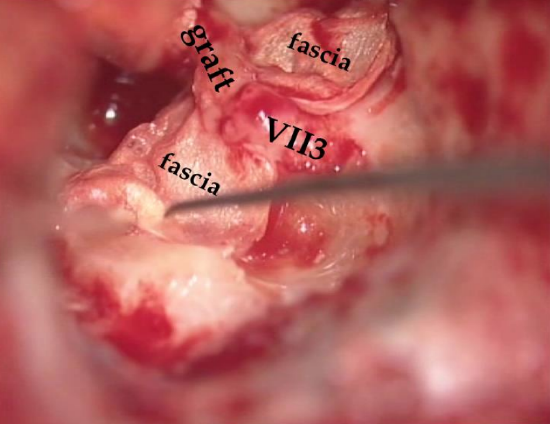
Figure 14: Iatrogenic traumatic lesion of the right facial nerve. Temporalis fascia wrap and distal fibrin-bonding of the graft in contact with the mastoid portion of the facial nerve
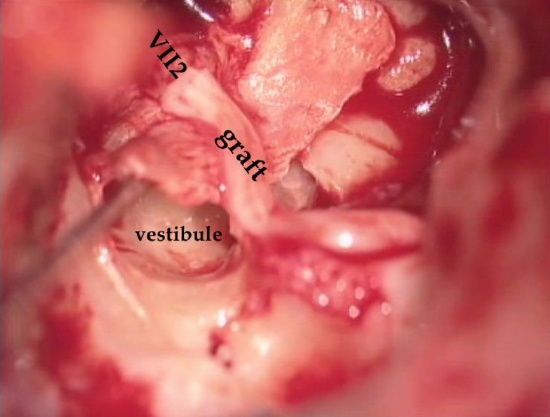
Figure 15: Iatrogenic traumatic lesion of the right facial nerve. Temporalis fascia wrap and proximal fibrin-bonding of the graft in contact with the initial tympanic portion of the facial nerve. The transplant is in the bridge over the vestibule
Here the graft is naturally stabilized in the Fallopian canal. It is also necessary to create bone contact for good vascularization of the graft (Figure 14 and 15). Unless the proximal branch is intradural in the IAM, the sleeve does not need to be circumferential and a simple fascial covering on the nerve repair will be sufficient. Here again there is no need for a nerve suture. Fibrin-bonding is sufficient.
Difficulties encountered are twofold:
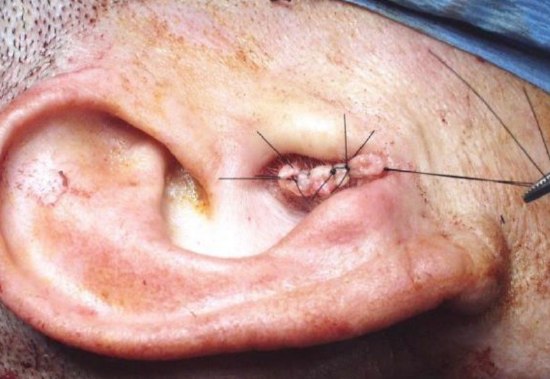
Figure 16: Closing the skin of the external auditory meatus
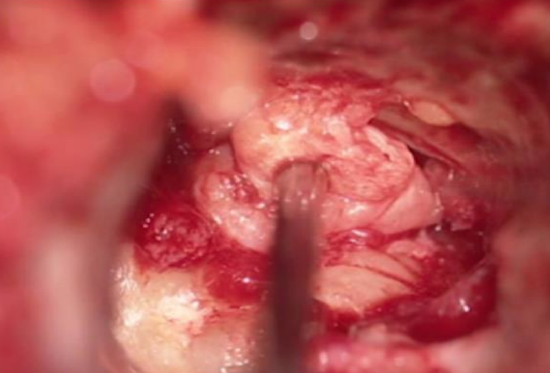
Figure 17: Iatrogenic traumatic lesion of the right facial nerve. The Eustachian tube is closed by successive layers of bone wax and fascia
6-2.1 The proximal fragment of the nerve is difficult to access without affecting the anatomy of the middle ear. In cases of iatrogenic trauma at the genu of the nerve, access to the tympanic portion or to the geniculate ganglion may require sacrificing all or part of the ossicular chain or external auditory meatus bone This may require isolating the middle ear after closure of the external cutaneous auditory meatus (Figure 16) and obliteration of the Eustachian tube by fascia (Figure 17). The decision may also depend on associated lesions (ossicular and labyrinthine).
6-2.2 The viable proximal fragment is deeply located in the labyrinthine portion of the VIIn or even in the IAC, whereas labyrinthine function is preserved and a translabyrinthine approach is therefore not possible. If the temporal bone is highly pneumatized, the labyrinthine portion of the nerve is partially accessible. On the other hand, if the narrowness of the temporal bone does not permit access or if the graft must be done in the IAM, it becomes necessary to include a middle fossa approach to allow both to connect the graft in the IAM or to the labyrinthine portion and a transmastoid pathway to connect to the mastoid portion.
6-3 In the parotid
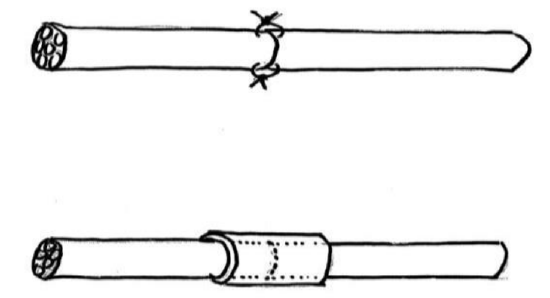
Figure 18: Diagram illustrating the microsuture-wrap technique adapted to parotid surgery
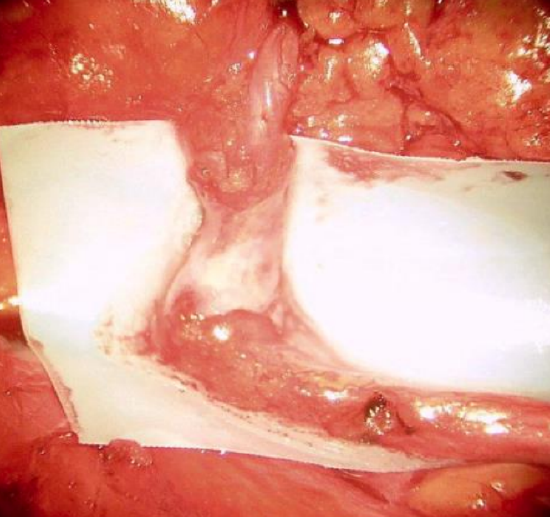
Figure 19: Operative photo of micro-suture-wrap in parotid surgery
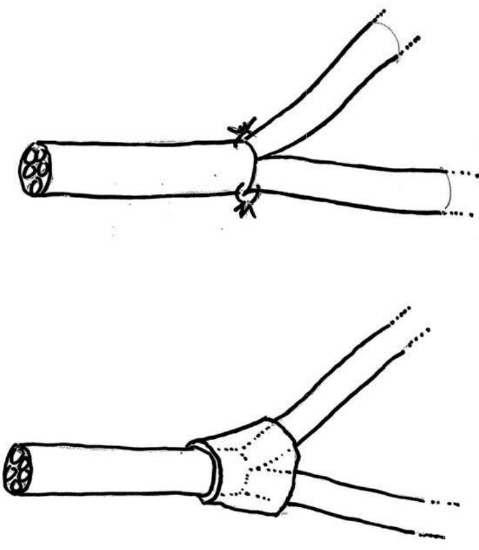
Figure 20: Diagram illustrating technique of microsuture-wrap at nerve division adapted to parotid surgery
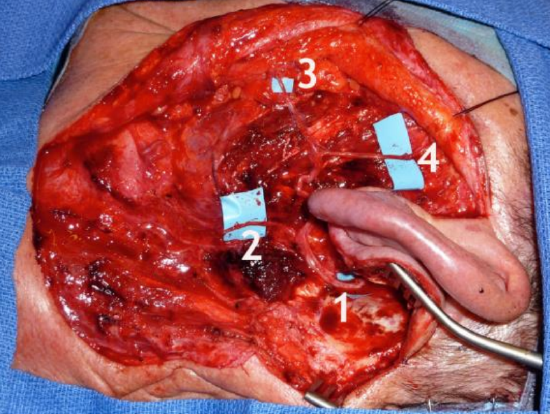
Figure 21: After resection of left parotid carcinoma, a split sural nerve graft is interposed between the mastoid facial nerve (1) and distal branches (2, 3, 4)
Nerve grafts are frequently used with malignancy and sacrifice of the facial nerve, provided the prognosis is favorable and the resection is considered curative. Radiotherapy does not preclude a good functional result, provided that the graft is placed on a well-vascularized bed and that the assessment of nerve invasion by tumor is correct. Particular attention should be paid with adenoid cystic carcinoma, as tumor can ascend along the facial nerve into the CPA. It is recommended that the nerve is sutured at one or two points with 9 or 10/0 sutures. A wrap and extra glue are useful (Figures 18 to 20). If it is necessary to address tumor invasion beyond the stylomastoid foramen and to expose the mastoid portion of the facial nerve, grafting at this level can be done without sutures. When the intraparotid facial nerve is completely sacrificed due to nerve infiltration (preoperative facial palsy), a sural nerve graft is split to provide a multiple branches to the frontal, orbicularis oculi, orbicularis oris, and marginal mandibular branches (Figure 21).
Stab or firearm injuries are sometimes encountered. In cases of stab wounds, a simple approximation-suture is sometimes possible. The prognosis is good. With firearm injuries the local mechanical and thermal trauma always requires trimming and a significant nerve graft in accordance with the protocol previously described. The prognosis is more uncertain, because infection may occur in a potentially polluted setting.
References
- Ramos DS, Bonnard D, Franco-Vidal V, Liguoro D, Darrouzet V. Stitchless fibrin glue-aided facial nerve grafting after cerebellopontine angle schwannoma removal : technique and results in 15 cases. Otol Neurotol. 2015 Mar;36(3): 498-502
- Stennert E. Why does the frontalis muscle "never come back"? Functional organization of the mimic musculature. Eur Arch Otorhinolaryngol. 1994 Dec: S91-5
- Charachon R, Tixier C, Lavieille JP, Reyt E. End-to-end anastomosis versus nerve graft in intratemporal and intracranial lesions of the facial nerve. Eur Arch Otorhinolaryngol. 1994 Dec: S281-3
- Linder T, Mulazimoglu S, El Hadi T, Darrouzet V, Ayache D, Somers T, Schmerber S, Vincent C, Mondain M, Lescanne E, Bonnard D. Iatrogenic facial nerve injuries during chronic otitis media surgery: a multicentre retrospective study. Clin Otolaryngol. 2017 Jun;42(3):521-527
- Franco-Vidal V, Blanchet H, Liguoro D, Darrouzet V. L’anastomose hypoglossofaciale latéro-terminale. Résultats à long terme et indications. A propos de 15 cas sur 10 ans. Rev Laryngol Otol Rhinol (Bord). 2006; 127(1-2):97-102
Authors
Prof Vincent Darrouzet
Service d’ORL
Fédération de Chirurgie de la Base du Crâne
CHU Bordeaux, Université de Bordeaux
vincent.darrouzet@chu-bordeaux.fr
Dr Erwan de MONES del PUJOL
Service d’ORL
Fédération de Chirurgie de la Base du Crâne
CHU Bordeaux, Université de Bordeaux
erwan.de-mones-delpujol@chu.bordeaux.fr
Editor
Johan Fagan MBChB, FCS(ORL), MMed
Professor and Chair
Division of Otolaryngology
University of Cape Town
Cape Town, South Africa
johannes.fagan@uct.ac.za


