2.16: Surgery for Temporal Bone Trauma
- Page ID
- 17718
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)OPEN ACCESS ATLAS OF OTOLARYNGOLOGY, HEAD & NECK OPERATIVE SURGERY
SURGERY FOR TEMPORAL BONE TRAUMA
Vincent Cousins
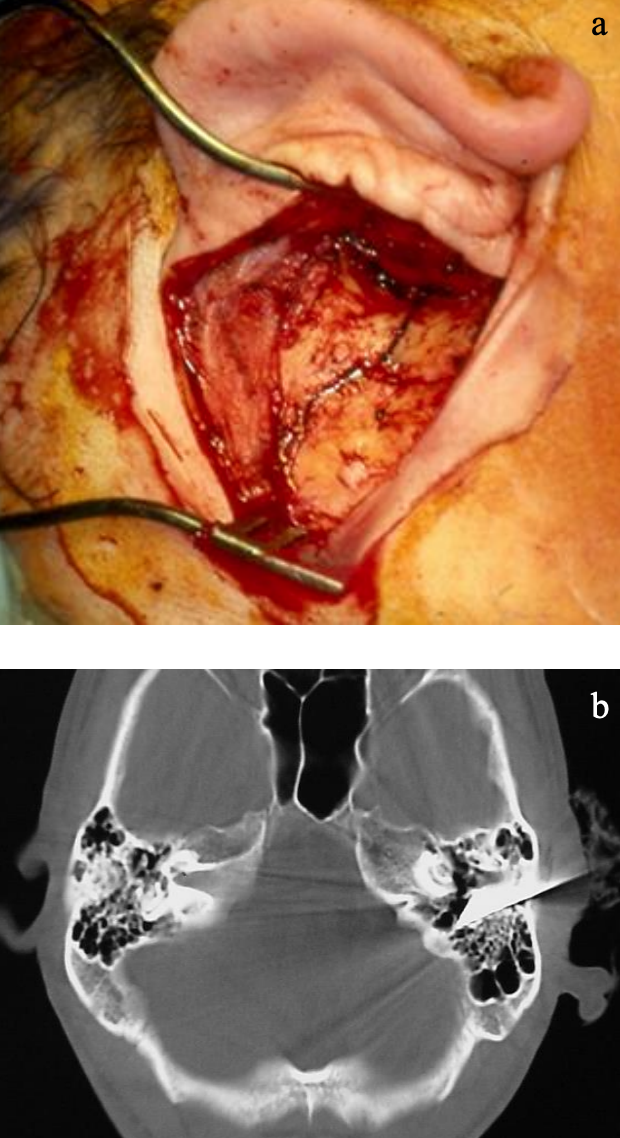
Figures 1a, b: Fracture of mastoid bone entering the ear canal; Knife that transected the facial nerve
Trauma to the temporal bone is commonly seen. It occurs most frequently as part of blunt head trauma but may also be due to penetrating injuries including gunshots and stabbings (Figures 1a, b).
Classifications
Fractures may be unilateral or bilateral. Classically, fractures of the temporal bone are classified into longitudinal or transverse according to its long axis which passes from its base posterolaterally to the petrous apex anteromedially. The direction of a single trauma force may cause a longitudinal fracture on one side and a transverse on the other. Any combination of all fracture types is possible depending on the nature and severity of the trauma involved.
Longitudinal fractures generally pass through the external auditory canal, the middle ear and Eustachian tube. They commonly involve the bony external canal, lacerate the tympanic membrane and may disrupt the ossicular chain. The inner ear is generally preserved and facial nerve lesions are mostly compressive from hematoma or displaced bone fragments.
Transverse fractures cross the long axis of the temporal bone and more commonly involve the inner ear. Facial nerve injuries are more likely to cause partial or complete disruption of the facial nerve.
Isolated fracturing of the anterior wall of the external canal may also occur from posterior displacement of the mandibular condyle seen with front-on facial injuries.
Fractures may be more complex and not readily fit the above classification. This led Kelly & Tami to propose a different classification based on involvement of the inner ear1.
Clinical Presentation
A fracture of the temporal bone should be suspected in any case of significant head trauma. This may be part of a high impact injury as seen in motor vehicle collisions, or low speed injuries such as falls onto a hard surface. Penetrating trauma involving the temporal bone or the external ear canal alone may also cause the same pattern of injuries.
The classical sign of a fracture of the temporal bone is bleeding from the ear canal. A conscious patient may complain of hearing loss, vertigo and facial weakness.
Physical Examination
Assessment and treatment of associated head, spinal and other life-threatening injuries must take precedence over those of the temporal bone trauma.
The ear should be examined and the source of bleeding identified. Skin lacerations of the external ear, scalp or neck should be identified. The ear canal should be examined and blood gently cleared from its outer end.

Figure 2: Battle’s sign that appeared four days after the temporal bone fracture
“Battle’s sign” refers to bruising that may appear over the mastoid process four or more days after a fracture of the temporal bone has occurred (Figure 2).

Figure 3: Halo sign of CSF mixed with blood
Cerebrospinal fluid (CSF) may be identified issuing from the ear canal. If mixed with blood it may not be easily detected. Dry stains on the bedding may show a “Halo sign” where the blood forms the inner darker stain and the CSF seeps more peripherally producing a paler rim or halo (Figure 3).
The canal should not initially be cleared aggressively as clotting seals the canal helping to prevent contamination of the canal which may be transmitted to the subarachnoid space. The patency of the canal should however be assessed as it may be crushed requiring early packing to prevent stenosis. The condition of the tympanic membrane should also be assessed as perforations accompanying temporal bone fractures are commonly seen in the posterosuperior quadrant.
If the patient is conscious, hearing should be assessed. Speech and whisper tests can be performed on each side. The Weber and Rinne tuning fork tests are also very pertinent to distinguish between conductive and sensorineural hearing loss.

Figures 4: Longitudinal fracture of temporal bone (yellow). Note separation of incus and malleus (red)
A conductive hearing loss may be caused by blood in the middle ear (hemotympanum), tympanic membrane trauma or disruption of the ossicular chain (Figure 4).
Sensorineural loss will be present if the inner ear is involved; this may be more minor in a concussive type of injury, or complete if the fracture passes through the cochlea or vestibular apparatus (Figure 6). Inner ear trauma also causes vestibular loss and may cause nystagmus. Full clinical vestibular assessment should not be performed in the acute phase, particularly until the cervical spine has been cleared of significant injury.
Facial movement must be assessed as early as possible. Facial grimacing on the side of the fracture in a semiconscious patient is an important sign to elicit. A conscious patient should be asked to perform the full range of facial movement looking for presence and strength of eyebrow elevation, eye closure, cheek wrinkling, smiling or dental exposure and lip pursing. The presence of definite movement indicates that the nerve is anatomically intact. Delayed facial palsy may occur after hours or days and early knowledge of movement has significant management and prognostic implications. Facial palsy that is present immediately after temporal bone trauma or where the time of onset is not known and where no facial movement has been observed raises the possibility of disruption of the nerve and possible need for surgical intervention.
Investigations
Imaging
Although plain x-rays of the temporal bone will show most larger or displaced fractures, CT scanning is the investigation of choice. Fracture lines and displaced fragments will be visible. The mastoid air cell system will be opacified by blood early on and mucosal edema and fluid later on. The ossicular chain should be reviewed. Displacement or dislocation of one or more of the ossicles may be seen e.g. the gap between the incus and malleus may be widened in a subluxation injury (Figure 4).
The facial nerve canal should be examined along its course from the internal auditory canal to the stylomastoid foramen for any direct involvement by a fracture.
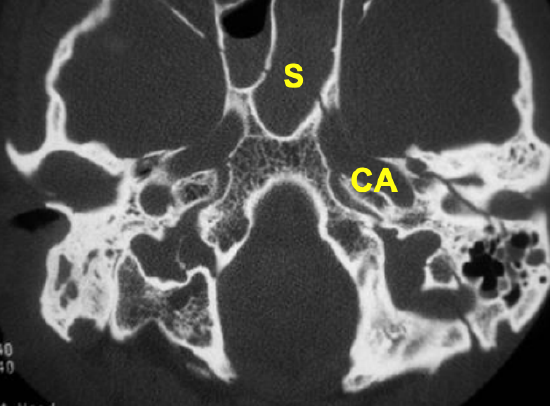
Figure 5: Note proximity of longitudinal fracture line to the internal carotid artery (CA) and traversing the opacified sphenoid sinus (S)
The medial extent of the fracture should be assessed. It may involve the carotid artery as well as the sphenoid sinus (Figure 5). Suspected carotid artery involvement may be further assessed by CT angiography.

Figure 6a: Transverse fracture of the temporal bone. Fracture seen passing into the vestibule which also contains air

Figure 6b: Transverse fracture of the temporal bone. Fracture seen passing into the vestibule
A fracture involving any compartment of the inner ear (cochlea, vestibule or semicircular canals) is associated with complete ipsilateral hearing loss (Figures 6a,b). Air is sometimes seen in the inner ear in such injuries (Figure 6a).

Figure 7: Anterior wall fracture with posterior displacement of bone fragments (arrow). Note blood and air in temporomandibular joint *
Crushing injuries of the external auditory canal with posterior displacement of bone fragments leading to narrowing and potential stenosis is best seen on axial CT scans (Figure 7).
Hearing and vestibular assessment
Once a patient is stable and able to cooperate, pure tone and speech audiometry should be performed. Impedance testing should be avoided until the ear canal and tympanic membrane have fully healed.
Formal vestibular function tests may be warranted if the patient has ongoing vertigo or disequilibrium after three months or more.
Management
External Auditory Canal
If severe enough and left untreated, a disrupted ear canal with displacement of its bony walls leads to permanent narrowing or stenosis. Early packing of a collapsed canal helps prevent permanent stenosis. This should be done with appropriate anesthesia and in an aseptic manner. Care needs to be taken to prevent additional trauma, especially to the facial nerve if there is a mobile posterior wall segment.
Meatoplasty for scarring of the outer canal and canalplasty for permanent bony stenosis can be performed as delayed procedures. Small defects of the posterior or other walls due to bone loss can be repaired with cartilage grafting. In cases where there is extensive loss of the posterior wall, conversion to a canal wall down cavity is occasionally warranted.
Hearing Loss
Conductive hearing loss can be treated expectantly in the first instance. Time should be given for middle ear blood and fluid effusions to resolve. Myringoplasty may be indicated if a perforation does not heal spontaneously after three months.
Ossicular chain reconstruction can be performed as a delayed procedure if the conductive hearing loss is significant enough to warrant surgical intervention. A displaced incus can be retrieved, reshaped and interposed. More extensive ossicular disruption may be better treated with alloplastic partial or total ossicular prostheses.
Hearing aids may be appropriate in some cases in line with usual tympanoplasty indications and contraindications and if there is a sensorineural component to the hearing loss.
Facial Paralysis
Facial paralysis can be a significant permanent sequela of a fracture of the temporal bone.
When definite ipsilateral facial movement has been documented one generally adopts an expectant approach as good recovery mostly occurs. The severity of the weakness, the duration of time before movement begins to return and a patient’s age are all relevant prognosticators for return of function. Complete paralysis, no sign of recovery for more than four weeks and advanced age are likely to be associated with poorer outcomes. When no movement returns, consideration needs to be given to exploring the relevant segments of the nerve and to repair or graft it.
Where there is immediate paralysis or the time of onset has not been documented, and there is involvement of the bony facial nerve canal by the fracture on CT scan and no response to electrical stimulation, the author favors surgical exploration with the intention of proceeding to nerve repair if necessary. This is better done sooner rather than later, although the patient’s general health and other injuries will often necessitate delay.

Figure 8: Greater auricular nerve
The nerve is explored and the involved segment isolated. Granulation tissue, bone fragments and scar are removed. The continuity of the nerve is assessed. If intact, the author generally opens the nerve sheath. If partially severed but >50% of the nerve’s cross-section is intact the author leaves the nerve in its position and repairs it as is possible. The author may even place a small inlay nerve graft into the missing segment. The ipsilateral greater auricular nerve usually provides enough graft material (Figure 8).
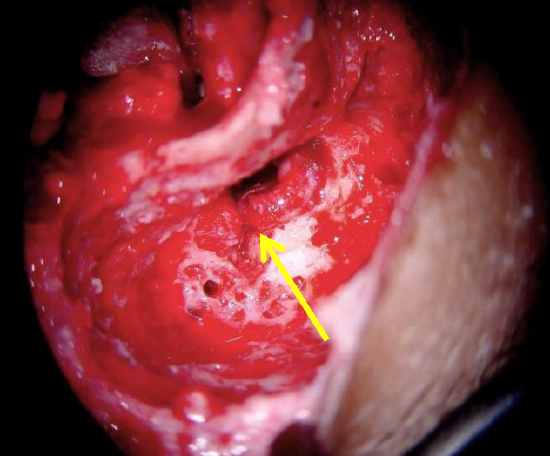
Figure 9: Transected nerve just distal to 2nd genu as viewed via cortical mastoidectomy (Same case as in Figure 1)
Severe crushing or severance of >50% of the nerve’s cross-section is an indication for resection of the involved segment and cable grafting of the nerve (Figure 9).
In the setting of total hearing loss, the author may drill out the labyrinth to mobilize the nerve to allow for a single anastomosis.
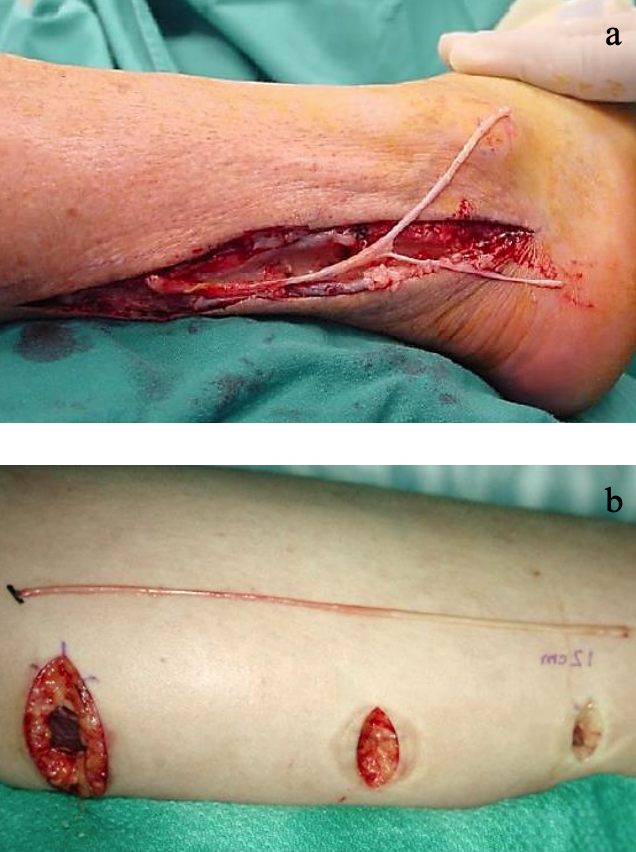
Figures 10a,b: Sural nerve harvesting techniques
When hearing has been preserved, a middle cranial fossa approach may be required to access the internal auditory meatus and labyrinthine segments of the nerve. Although the greater auricular may be used in some cases, the sural nerve is generally used for cable grafting in such cases due to its size and length (Figure 10).
The best nerve repair or cable graft results are achieved when done early after nerve injury but acceptable results can be achieved after 3 months and even up to 12 months, but no better than equivalent to a House-Brackman level Grade 3 to 4.
Also see chapters:
Dysequilibrium & Vertigo
Vertigo occurs commonly after temporal bone trauma. Benign positional vertigo (BPV) is often seen and generally responds to particle repositioning maneuvers or specific exercises. Permanent loss of vestibular function from a fracture involving the inner ear may cause significant vertigo. Some patients compensate easily. Others may require vestibular rehabilitation physical therapy; a very small number may require formal vestibular ablation, either chemical or surgical.
Reconstructive procedures
A variety of reconstructive procedures may be necessary depending on the specific injuries of the external ear, middle ear and mastoid.
Surgical management of traumatic CSF leak
The author has been required to surgically repair persistent CSF leaks only on rare occasions as CSF leaks with fractures of the temporal bone almost always resolve spontaneously.

Figure 11: Fracture of tegmen mastoideum

Figure 12: Fracture involving posterior cranial fossa plate with intracranial air
The likely site(s) of persistent CSF leaks are generally apparent on high resolution CT scans, and can originate from both middle and posterior fossa dural defects (Figures 11, 12).
When hearing is still serviceable, an attempt should be made to preserve the anatomy of the external auditory canal and mesotympanum. With a localized defect of the posterior fossa dural plate, a cortical mastoidectomy with local repair of the dural defect using temporalis fascia and possibly also a cartilage plate harvested from the tragus or concha, and placed between the bone and the dura, may suffice.
This can be covered with a fat graft taken from the abdominal wall with the mastoid being obliterated as far as the aditus. A small piece of fat can be placed in the aditus and bone wax placed over this to seal the communication with the mastoid. A small amount of fat extending into the attic is likely to atrophy with time and have minimal permanent effect on ossicular mobility. Additional reinforcement of these repairs can be applied when available. Soft tissue dural grafts can be reinforced with tissue glues. Bony defect repairs can be augment-ted with cements, e.g. hydroxylapatite.
Localized defects in the tegmen mastoideum (that are posterior to the aditus) can be managed in the same way. Defects in the tegmen tympani, however, will mostly require removal of the incus and malleus head for access from below. Ossicular chain reconstruction can be performed once the repair is secure.
Larger defects of the tegmen may be associated with a secondary encephalocoele, and are best handled by combined mastoid and middle cranial fossa approaches which permit placement of larger extradural grafts of fascia, bone and pedicled muscle.
Complex defects of the tegmen and/or posterior fossa plate are best treated with subtotal petrosectomy, fascial reinforcement of traumatized dura, Eustachian tube obliteration, and blind sac closure of the external auditory canal. Fat obliteration of the middle ear cleft ensures closure of a leak via the final common pathway, the Eustachian tube. This technique is particularly applicable if there is no useable hearing in the ear.
Blind sac closure of the external auditory canal technique
Figure 13: Blind sac closure of external auditory canal
The external auditory canal is permanently occluded
- Transect the bony canal skin circumferentially with a Plester knife
- Reflect the skin of the lateral canal off the underlying bone from the incision line
- Remove the tympanic membrane, medial canal skin, malleus, and incus after formal separation of the incudostapedial joint. The stapes superstructure can also be removed with crurotomy scissors
- Gently elevate the skin of the remaining lateral part of the canal off the cartilage, working from the medial aspect of the pinna
- Take care to keep the skin intact, creating a tube of approximately 1 cm in length
- Place 2 sutures in the subcutaneous tissue at the medial end of the skin tube, one superiorly and the other inferiorly; they are initially left long and held in an artery forceps
- Pass a small artery forceps through the external meatus and clip it onto one suture’s end which is then gently pulled through the tube from medial to lateral
- Use a 2nd forceps to deliver the second suture in the same manner
- Gently pull both suture ends laterally to completely evert the tube of skin of the lateral canal. This ensures that the epidermal surface of the tube is completely exteriorized
- Oversew the end of the tube with interrupted absorbable sutures
- Create a 2nd layer of closure medial to the oversewn skin with an anteriorly based musculoperiosteal flap raised from surface of the mastoid bone; this is best created at the time of initial exposure of the mastoid cortex
- Make superior, posterior and inferior incisions in this layer to create a rectangular flap that is based on the soft tissues posterior to the external meatus
- Elevate the flap from the cortex and reflected it forward with the pinna
- Once eversion of the canal skin has been completed, rotate the rectangular flap over its medial end and secure it in place with absorbable sutures
Fat graft (Figure 14)
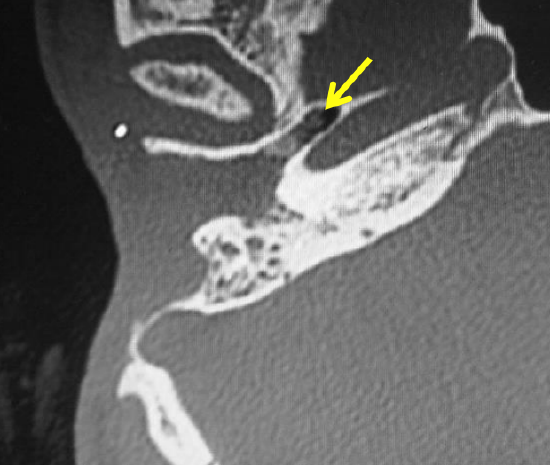
Figure 14: Mastoid cavity obliterated with fat. Note bone pate in Eustachian Tube orifice (arrow)
- This is generally taken from the left lower abdominal quadrant (A right-sided scar may be mistaken for a prior appendicectomy)
- Take care not buttonhole or excessively undermine the skin
- Dissection must remain superficial to the aponeurosis of the external oblique muscle
- The volume of fat taken must be slightly greater than the volume of the bony defect of the mastoidectomy in order to achieve some gentle compression on the dural repair and to allow for subsequent atrophy of the fat
- Close the abdominal wall wound in multiple layers after meticulous hemostasis has been achieved
- A suction drain is left in situ for 48 hours to minimize troublesome hematoma, seroma or abscess
Mastoid, middle ear and Eustachian tube
- Clear the mastoid of all accessible air cells and mucosa
- Denude the mesotympanum of as much mucosa as possible
- Drill out the protympanum (area around the Eustachian tube) to clear surrounding air cells
- Reflect the mucosa of the Eustachian tube into its lumen taking great care not to injure the internal carotid artery which is an immediate medial relation and sometimes without a bony covering in this area (Figure 14)
- Do a multiple layered seal using a small piece of fascia placed over the mucosa followed by a small ball of bone wax gently advanced to seal the raw bone of the Eustachian canal. This is covered with an additional small piece of fascia
- Cover the protympanum with bone pate and overly this with a sheet of fascia
- Fill the cavity of the mesotympanum with a small piece of fat
- Place one or more large pieces of fat to fill the mastoid
- Close the wound in multiple layers
- Suction tube drainage is not used, especially in cases of CSF leak
- Apply a well-padded mastoid dressing for 24 hours
Concluding remarks
- Temporal bone fractures are relatively common
- They may involve the outer, middle and/or inner ears
- Early, detailed assessment of the effects of the trauma need to be made
- In most cases, an expectant approach can be adopted and appropriate reconstruction undertaken in a delayed manner
- Facial palsy needs to be fully assessed as early as possible as early surgical intervention may be required to achieve the best outcome
Reference
- Kelly, KE and Tami, TA. Temporal Bone and Skull Base Trauma in Neurotology Eds Jackler, RK & Brackman, DE (1994) Mosby-Year Book, Inc, Missouri, USA
Author
Vincent C Cousins BMedSci (Hons), MBBS(Hons), FRACS
Head of ENT-Otoneurology Unit
The Alfred Hospital
Melbourne, Australia
Clinical Associate Professor
Monash University
Melbourne, Australia
vccousins@iinet.net.au
Editor
Johan Fagan MBChB, FCS(ORL), MMed
Professor and Chairman
Division of Otolaryngology
University of Cape Town
Cape Town, South Africa
johannes.fagan@uct.ac.za


