12.1: Introduction to Antioxidants
- Page ID
- 36354
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\( \newcommand{\id}{\mathrm{id}}\) \( \newcommand{\Span}{\mathrm{span}}\)
( \newcommand{\kernel}{\mathrm{null}\,}\) \( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\) \( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\) \( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\id}{\mathrm{id}}\)
\( \newcommand{\Span}{\mathrm{span}}\)
\( \newcommand{\kernel}{\mathrm{null}\,}\)
\( \newcommand{\range}{\mathrm{range}\,}\)
\( \newcommand{\RealPart}{\mathrm{Re}}\)
\( \newcommand{\ImaginaryPart}{\mathrm{Im}}\)
\( \newcommand{\Argument}{\mathrm{Arg}}\)
\( \newcommand{\norm}[1]{\| #1 \|}\)
\( \newcommand{\inner}[2]{\langle #1, #2 \rangle}\)
\( \newcommand{\Span}{\mathrm{span}}\) \( \newcommand{\AA}{\unicode[.8,0]{x212B}}\)
\( \newcommand{\vectorA}[1]{\vec{#1}} % arrow\)
\( \newcommand{\vectorAt}[1]{\vec{\text{#1}}} % arrow\)
\( \newcommand{\vectorB}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vectorC}[1]{\textbf{#1}} \)
\( \newcommand{\vectorD}[1]{\overrightarrow{#1}} \)
\( \newcommand{\vectorDt}[1]{\overrightarrow{\text{#1}}} \)
\( \newcommand{\vectE}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash{\mathbf {#1}}}} \)
\( \newcommand{\vecs}[1]{\overset { \scriptstyle \rightharpoonup} {\mathbf{#1}} } \)
\( \newcommand{\vecd}[1]{\overset{-\!-\!\rightharpoonup}{\vphantom{a}\smash {#1}}} \)
\(\newcommand{\avec}{\mathbf a}\) \(\newcommand{\bvec}{\mathbf b}\) \(\newcommand{\cvec}{\mathbf c}\) \(\newcommand{\dvec}{\mathbf d}\) \(\newcommand{\dtil}{\widetilde{\mathbf d}}\) \(\newcommand{\evec}{\mathbf e}\) \(\newcommand{\fvec}{\mathbf f}\) \(\newcommand{\nvec}{\mathbf n}\) \(\newcommand{\pvec}{\mathbf p}\) \(\newcommand{\qvec}{\mathbf q}\) \(\newcommand{\svec}{\mathbf s}\) \(\newcommand{\tvec}{\mathbf t}\) \(\newcommand{\uvec}{\mathbf u}\) \(\newcommand{\vvec}{\mathbf v}\) \(\newcommand{\wvec}{\mathbf w}\) \(\newcommand{\xvec}{\mathbf x}\) \(\newcommand{\yvec}{\mathbf y}\) \(\newcommand{\zvec}{\mathbf z}\) \(\newcommand{\rvec}{\mathbf r}\) \(\newcommand{\mvec}{\mathbf m}\) \(\newcommand{\zerovec}{\mathbf 0}\) \(\newcommand{\onevec}{\mathbf 1}\) \(\newcommand{\real}{\mathbb R}\) \(\newcommand{\twovec}[2]{\left[\begin{array}{r}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\ctwovec}[2]{\left[\begin{array}{c}#1 \\ #2 \end{array}\right]}\) \(\newcommand{\threevec}[3]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\cthreevec}[3]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \end{array}\right]}\) \(\newcommand{\fourvec}[4]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\cfourvec}[4]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \end{array}\right]}\) \(\newcommand{\fivevec}[5]{\left[\begin{array}{r}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\cfivevec}[5]{\left[\begin{array}{c}#1 \\ #2 \\ #3 \\ #4 \\ #5 \\ \end{array}\right]}\) \(\newcommand{\mattwo}[4]{\left[\begin{array}{rr}#1 \amp #2 \\ #3 \amp #4 \\ \end{array}\right]}\) \(\newcommand{\laspan}[1]{\text{Span}\{#1\}}\) \(\newcommand{\bcal}{\cal B}\) \(\newcommand{\ccal}{\cal C}\) \(\newcommand{\scal}{\cal S}\) \(\newcommand{\wcal}{\cal W}\) \(\newcommand{\ecal}{\cal E}\) \(\newcommand{\coords}[2]{\left\{#1\right\}_{#2}}\) \(\newcommand{\gray}[1]{\color{gray}{#1}}\) \(\newcommand{\lgray}[1]{\color{lightgray}{#1}}\) \(\newcommand{\rank}{\operatorname{rank}}\) \(\newcommand{\row}{\text{Row}}\) \(\newcommand{\col}{\text{Col}}\) \(\renewcommand{\row}{\text{Row}}\) \(\newcommand{\nul}{\text{Nul}}\) \(\newcommand{\var}{\text{Var}}\) \(\newcommand{\corr}{\text{corr}}\) \(\newcommand{\len}[1]{\left|#1\right|}\) \(\newcommand{\bbar}{\overline{\bvec}}\) \(\newcommand{\bhat}{\widehat{\bvec}}\) \(\newcommand{\bperp}{\bvec^\perp}\) \(\newcommand{\xhat}{\widehat{\xvec}}\) \(\newcommand{\vhat}{\widehat{\vvec}}\) \(\newcommand{\uhat}{\widehat{\uvec}}\) \(\newcommand{\what}{\widehat{\wvec}}\) \(\newcommand{\Sighat}{\widehat{\Sigma}}\) \(\newcommand{\lt}{<}\) \(\newcommand{\gt}{>}\) \(\newcommand{\amp}{&}\) \(\definecolor{fillinmathshade}{gray}{0.9}\)- List several nutrients that serve as antioxidants.
The market is flooded with advertisements for “super antioxidant” supplements teeming with molecules that block free radical production, stimulate the immune system, prevent cancer, and reduce the signs of aging. Based on the antioxidant-supplement industry’s success, the general public appears to believe these health claims. However, these claims are not backed by scientific evidence; rather, there is some evidence suggesting supplements can actually cause harm. While scientists have found evidence supporting the consumption of antioxidant-rich foods as a method of reducing the risk of chronic disease, there is no “miracle cure”; no pill or supplement alone can provide the same benefits as a healthy diet. Remember, it is the combination of antioxidants and other nutrients in healthy foods that is beneficial. In this section, we will review how particular antioxidants function in the body, learn how they work together to protect the body against free radicals, and explore the best nutrient-rich dietary sources of antioxidants. One dietary source of antioxidants is vitamins. In our discussion of antioxidant vitamins, we will focus on vitamins E, C, and A.

Antioxidant Chemicals Obtained from the Diet
There are many different antioxidants in food, including selenium, which is one of the major antioxidants. However, the antioxidants you may be the most familiar with are vitamins. The “big three” vitamin antioxidants are vitamins E, A, and C, although it may be that they are called the “big three” only because they are the most studied.
| Antioxidant | Antioxidant Source | Antioxidant Function |
|---|---|---|
| Vitamin A | Karat banana, beef liver, chicken liver | Protects cellular membranes, prevents glutathione depletion, maintains free radical detoxifying enzyme systems, reduces inflammation |
| Vitamin E | Sunflower seeds, almonds, sunflower oil | Protects cellular membranes, prevents glutathione depletion |
| Vitamin C | Oranges, grapefruit | Protects DNA, RNA, proteins, and lipids, aids in regenerating vitamin E |
| Vitamin D | Swordfish, salmon, tuna fish canned in water and drained | Regulates blood calcium levels in concert with parathyroid hormone |
| Carotenoids | Pumpkin, carrots | Free radical scavenger |
What Are Antioxidants, and Why Do We Need Them?
Atoms are composed of a nucleus, which contains neutrons and protons, and electrons, which orbit the nucleus. Atoms are most stable when they have an even number of electrons, so that they can orbit the nucleus in pairs.
An atom or group of atoms with an unpaired electron is called a free radical. Free radicals are inherently unstable and highly reactive. They steal electrons from other molecules in order to stabilize themselves, but in doing so, they create additional free radicals. This electron-grabbing is called oxidation and can set up a chain reaction, creating new free radicals and damaging important molecules along the way, similar to how one falling domino can bring down countless more.
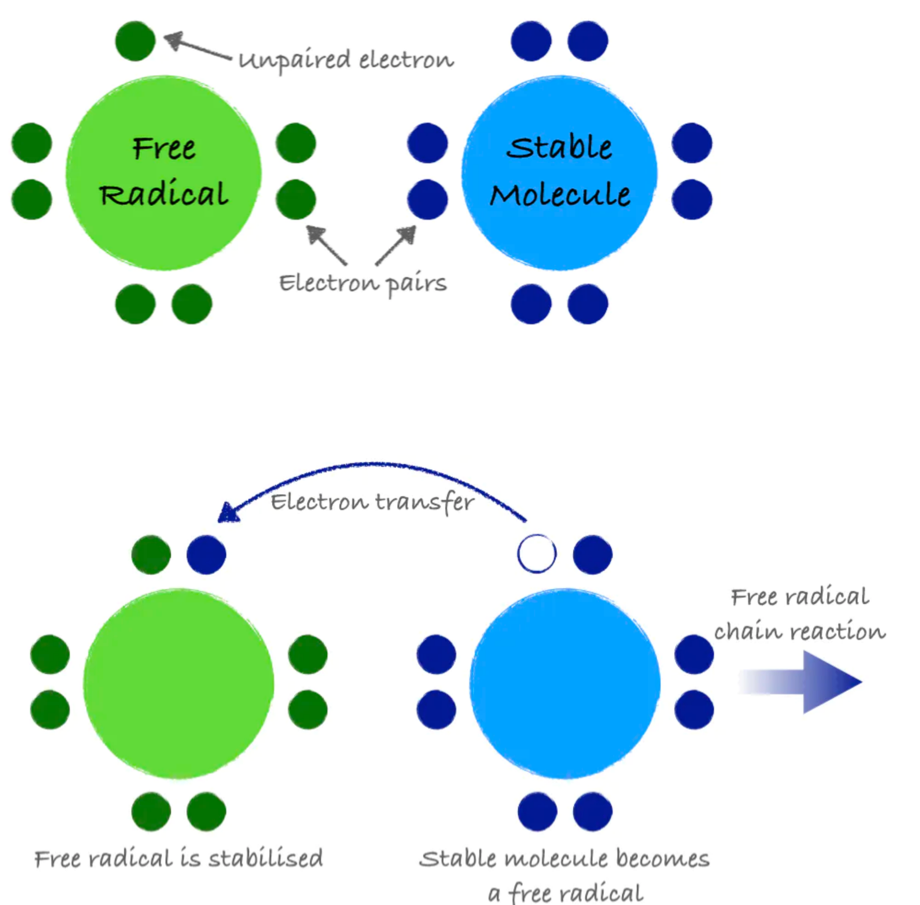
Antioxidants are molecules that can donate an electron to stabilize and neutralize free radicals. Like a domino that refuses to fall, an antioxidant can stop the free radical chain reaction in its tracks. In donating an electron, the antioxidant itself becomes a free radical. However, antioxidants are special in that they are not very reactive themselves and have processes for quick stabilization.
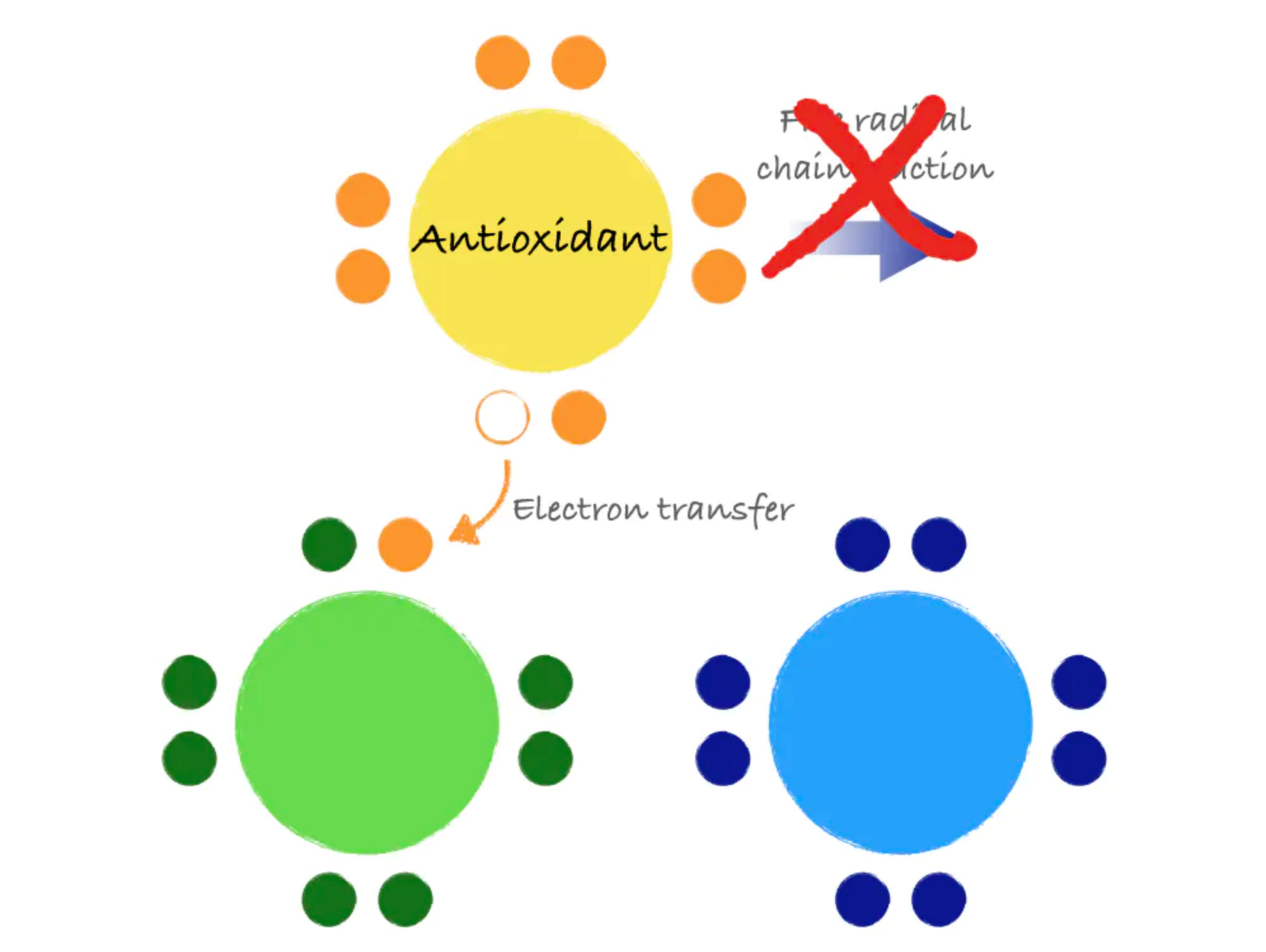
Some antioxidants are produced by the body, and some are consumed in the diet. Examples of dietary antioxidants include vitamins C and E, the mineral selenium, and phytochemicals, such as beta-carotene.
Free radicals are a natural byproduct of metabolic reactions and of exercise, and it’s normal to have low levels of free radicals in the body. In fact, free radicals play a role in normal functioning of the body, including the ability to fight off pathogens and to send signals from one cell to another. And with enough antioxidants present, free radicals can be kept in check so that they aren’t dangerous.1
However, too many free radicals and not enough protection from antioxidants creates a situation called oxidative stress. Free radical-induced damage, when left unrepaired, destroys lipids, proteins, RNA, and DNA, and can contribute to disease. Oxidative stress has been implicated as a contributing factor to cancer, cardiovascular disease, arthritis, diabetes, kidney disease, Alzheimer’s disease, Parkinson’s disease, and eye diseases such as cataracts.2
Substances and energy sources from the environment can add to or accelerate the production of free radicals within the body. Exposure to UV radiation (e.g., from sunlight), air pollution, tobacco smoke, heavy metals, ionizing radiation, asbestos, and other toxic chemicals increase the amount of free radicals in the body. They do so by being free radicals themselves or by adding energy that provokes electrons to move between atoms. Excessive exposure to environmental sources of free radicals can contribute to disease by overwhelming the free radical detoxifying systems and those processes involved in repairing oxidative damage.
Oxidative stress is associated with the development of chronic diseases, and eating a diet rich in antioxidant-containing foods like fruits, vegetables, and whole grains seems to protect against many of these same diseases. It was thus natural for researchers to hypothesize that taking antioxidants in supplement form might also offer protection from these diseases. However, several decades of research investigating this hypothesis have revealed disappointing results. Not only have these studies shown that antioxidant supplements generally aren’t beneficial, some have shown that they can cause health risks. For example, high doses of beta-carotene increased the risk of lung cancer in smokers, and high doses of vitamin E increased the risk of prostate cancer in men. Antioxidant supplements may also interact with other medications, further emphasizing the importance of talking with your doctor before taking supplements.2
Researchers aren’t sure why some antioxidant supplements have turned out to be dangerous. It may come back to the fact that free radicals play important roles in the body, and adding high doses of antioxidant supplements overwhelms the normal balance of free radicals and does more harm than good. It may also be that the benefits of eating antioxidant-rich foods come not just from the antioxidants but from the entire package of nutrients, like fiber and phytochemicals in the whole foods, a combination that simply can’t be replicated in a pill. Regardless, you can obtain adequate levels of dietary antioxidants simply by eating a healthy diet.1,2
References & Attributions
1 https://www.fda.gov/news-events/press-announcements/statement-fda-commissioner-scott-gottlieb-md-agencys-new-efforts-strengthen-regulation-dietary Accessed December 4, 2020
2 https://www.fda.gov/food/dietary-supplements. Accessed December 4, 2020
3 https://nutrition.bmj.com/content/ea...ph-2020-000150
4 Bjelakovic, G., D. Nikolova, and C. Gluud. 2013. Meta-regression analyses, meta-analyses, and trial sequential analyses of the effects of supplementation with beta-carotene, vitamin A, and vitamin E singly or in different combinations on all-cause mortality: Do we have evidence for lack of harm? PLoS ONE 8(9):e74558. doi: 10.1371/journal.pone.0074558
- Oregon
Attributions:
- Zimmerman, M., & Snow, B. Nutrients Important as Antioxidants. In An Introduction to Nutrition (v. 1.0). https://2012books.lardbucket.org/boo...ion/index.html, CC BY-NC-SA 3.0
References:
- 1Adcock, J. (2018, January 10). What are antioxidants? And are they truly good for us? The Conversation. https://theconversation.com/what-are-antioxidants-and-are-they-truly-good-for-us-86062
- 2National Center for Complementary and Integrative Health. (n.d.). Antioxidants: In Depth. Retrieved April 2, 2020, from https://www.nccih.nih.gov/health/antioxidants-in-depth
- 3National Institutes of Health Office of Dietary Supplements. (n.d.). Vitamin E – Health Professional Fact Sheet. Retrieved April 2, 2020, from https://ods.od.nih.gov/factsheets/VitaminE-HealthProfessional/
- 4National Institutes of Health Office of Dietary Supplements. (n.d.). Vitamin C – Health Professional Fact Sheet. Retrieved April 2, 2020, from https://ods.od.nih.gov/factsheets/VitaminC-HealthProfessional/
- 5Hemila, H., & Chalker, E. (2013). Vitamin C for preventing and treating the common cold. Cochrane Database of Systematic Reviews, CD000980. https://www.ncbi.nlm.nih.gov/pubmed/23440782
- 6National Institutes of Health Office of Dietary Supplements. (n.d.). Selenium- Health Professional Fact Sheet. Retrieved April 2, 2020, from https://ods.od.nih.gov/factsheets/Selenium-HealthProfessional/
Image Credits:
- Figure 8.12. “Free radical chain reaction” by Jacqui Adcock is licensed under CC BY-NC-ND 4.0
- Figure 8.13. “Antioxidant action” by Jacqui Adcock is licensed under CC BY-NC-ND 4.0
- Figure 8.14. “Food sources of Vitamin E” by Alice Callahan is licensed under CC BY 4.0, with images from top to bottom by Alice Callahan, CC BY 4.0; and Remi Yuan, Annie Spratt, and engin akyurt on Unsplash (license information)
- Figure 8.15. “Vitamin E/vitamin C antioxidant cycle” by Alice Callahan is licensed under CC BY 4.0
- Figure 8.16. “Food sources of Vitamin C” by Alice Callahan is licensed under CC BY 4.0, with images from top to bottom by Bruna Branco, Irene Kredenets, Kyaw Tun, and Forest Diver on Unsplash (license information)
- Figure 8.17. “38 year old man suffering from scurvy” by von K. H. Baumgärtner, Welcomme Library is licensed under CC BY 4.0
- “Emergen-C supplement photo” by Mike Mozart is licensed under CC BY 2.0
- Figure 8.18. “Food sources of selenium” by Alice Callahan is licensed under CC BY 4.0, with images from top to bottom by sunorwind, Сергей Орловский, Cake Tuxedo on Unsplash (license information), and “par cooked brown rice” by jules is licensed under CC BY-NC 2.0
AutoAttribution
- 8.6: Vitamins and Minerals as Antioxidants by Alice Callahan, Heather Leonard, & Tamberly Powell, is licensed CC BY-NC-SA

