17.3: Cardiac Muscle and Electrical Activity
- Page ID
- 22376
By the end of this section, you will be able to:
- Describe the structure of cardiac muscle tissue and cells
- Identify and describe the components of the conducting system that distributes electrical impulses through the heart
Recall that cardiac muscle shares a few characteristics with both skeletal muscle and smooth muscle, but it has some unique properties of its own. Not the least of these exceptional properties is its ability to initiate an electrical potential at a fixed rate that spreads rapidly from cell to cell to trigger the contractile mechanism. This property is known as autorhythmicity. Skeletal muscle can not do this. Even though cardiac muscle has autorhythmicity, heart rate is modulated by the endocrine and nervous systems.
Structure of Cardiac Muscle Tissue and Cells
Compared to the giant cylinders of skeletal muscle, cardiac muscle cells, or cardiomyocytes, are considerably shorter with much smaller diameters. Cardiac muscle also demonstrates striations, the alternating pattern of dark A bands and light I bands attributed to the precise arrangement of the myofilaments and fibrils that are organized in sarcomeres along the length of the cell (Figure \(\PageIndex{1}\).a). These contractile elements are virtually identical to skeletal muscle. T (transverse) tubules penetrate from the surface plasma membrane, the sarcolemma, to the interior of the cell, allowing the electrical impulse to reach the interior. The T tubules are only found at the Z discs, whereas in skeletal muscle, they are found at the junction of the A and I bands. Therefore, there are one-half as many T tubules in cardiac muscle as in skeletal muscle. In addition, the sarcoplasmic reticulum of cardiomyocytes stores few calcium ions, so most of the calcium ions must come from outside the cells. The result is a slower onset of contraction. Mitochondria are plentiful, providing energy for the cross-bridge cycling responsible for contractions of the heart. Typically, cardiomyocytes have a single, central nucleus, but two or more nuclei may be found in some cells. Cardiac muscle undergoes aerobic respiration patterns, primarily metabolizing lipids and carbohydrates. Myoglobin, lipids, and glycogen are all stored within the cytoplasm.
Cardiac muscle cells branch freely. A junction between two adjoining cells is marked by a critical structure called an intercalated disc, which helps support the synchronized contraction of the muscle and structurally links the cells together (Figure \(\PageIndex{1}\)). Intercalated discs are formed by regions of sarcolemma with specialized integral membrane proteins. These form desmosomes, fasciae adherens, and large numbers of gap junctions. Desmosomes and fasciae adherens anchor the cells together. Fasciae adherens also anchor actin filaments of the sarcomere directly to the sarcolemma. Gap junctions allow the passage of ions from cell-to-cell through their sarcoplasm, a feature essential to producing synchronized contractions (Figure \(\PageIndex{1}\).c). Intercellular connective tissue also helps to bind the cells together. The importance of strongly binding these cells together is necessitated by the forces exerted by contraction; unlike skeletal muscle in which cells are the length of the organ to pull collectively on a bone via collagenous connective tissue attachments, smaller cardiac muscle cells pull on each other and ultimately on the dense connective tissue of the cardiac skeleton when they contract.
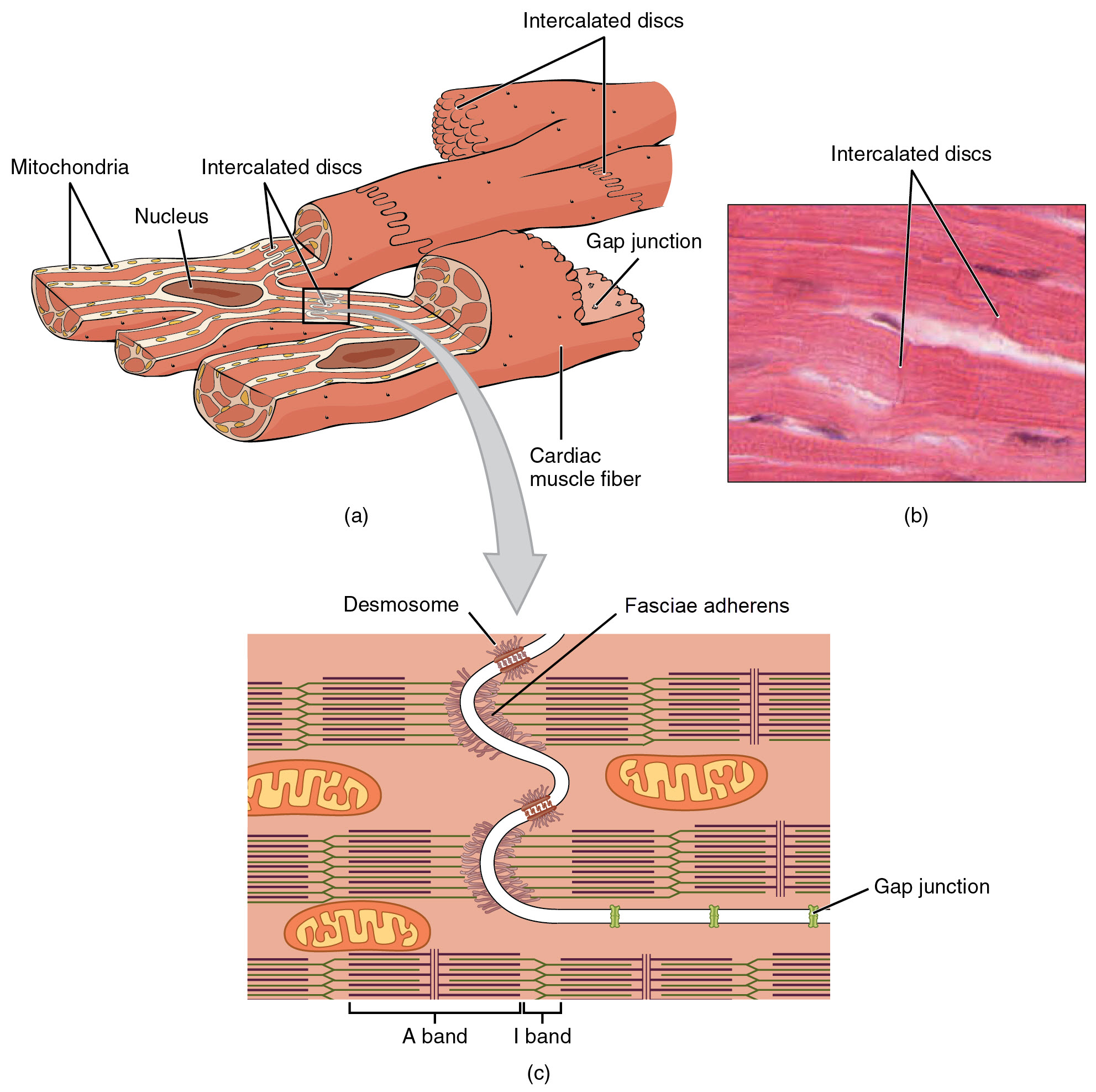 Figure \(\PageIndex{1}\): Cardiac Muscle. (a) Cardiac muscle cells have myofibrils composed of myofilaments arranged in sarcomeres, T tubules to transmit the impulse from the sarcolemma to the interior of the cell, numerous mitochondria for energy, and intercalated discs that are found at the junction of different cardiac muscle cells. (b) A photomicrograph of cardiac muscle cells shows the nuclei and intercalated discs. LM × 1600. (c) An intercalated disc connects cardiac muscle cells and consists of desmosomes, fasciae adherens, and gap junctions. (Image credit: "Cardiac Muscle" by Julie Jenks is licensed under CC BY 4.0 / A derivative from the original work / Micrograph provided by the Regents of the University of Michigan Medical School © 2012)
Figure \(\PageIndex{1}\): Cardiac Muscle. (a) Cardiac muscle cells have myofibrils composed of myofilaments arranged in sarcomeres, T tubules to transmit the impulse from the sarcolemma to the interior of the cell, numerous mitochondria for energy, and intercalated discs that are found at the junction of different cardiac muscle cells. (b) A photomicrograph of cardiac muscle cells shows the nuclei and intercalated discs. LM × 1600. (c) An intercalated disc connects cardiac muscle cells and consists of desmosomes, fasciae adherens, and gap junctions. (Image credit: "Cardiac Muscle" by Julie Jenks is licensed under CC BY 4.0 / A derivative from the original work / Micrograph provided by the Regents of the University of Michigan Medical School © 2012)There are two major types of cardiac muscle cells: myocardial contractile cells and myocardial conducting cells. The myocardial contractile cells constitute the bulk (99 percent) of the cells in the myocardium of the atria and ventricles. Myocardial contractile cells are autorhythmic, but they are primarily responsible for the strong, synchronized contractions necessary to pump blood through the body. The myocardial conducting cells (1 percent of the cells) are generally much smaller than the contractile cells and have fewer myofibrils for contraction. Myocardial conducting cells are specialized to initiate and propagate the electrical activity that travels throughout the heart and triggers the contractions of the myocardial contractile cells that propel the blood. If embryonic heart cells are separated into a Petri dish and kept alive, each is capable of generating its own electrical impulse (also referred to as a wave of depolarization or an action potential) that is followed by contraction (the capability that is described as autorhythmicity). When two independently beating embryonic cardiac muscle cells are placed together, the cell with the faster inherent rate sets the pace, and the impulse spreads from the faster to the slower cell to trigger a contraction in such rapid succession that they appear to contract in unison. As more cells are joined together, the fastest cell continues to assume control of the rate. A fully developed adult heart maintains the capability to generate its own electrical activity, triggered by the fastest cells. The fastest cells are the myocardial conducting cells. The slower cells (which are also stronger) are the myocardial contractile cells.
EVERYDAY CONNECTION
Repair and Replacement of Cardiac Muscle
Damaged cardiac muscle cells have extremely limited abilities to repair themselves or to replace dead cells via mitosis. Recent evidence indicates that at least some stem cells remain within the heart that continue to divide and at least potentially replace these dead cells. However, newly formed or repaired cells are rarely as functional as the original cells, and cardiac function is reduced. In the event of a heart attack or myocardial infarction (MI), dead cells are often replaced by patches of scar tissue in a process called fibrosis. Autopsies performed on individuals who had successfully received heart transplants show some proliferation of original cells. If researchers can unlock the mechanism that generates new cells and restore full mitotic capabilities to heart muscle, the prognosis for heart attack survivors will be greatly enhanced. To date, myocardial cells produced within the patient (in situ) by cardiac stem cells seem to be nonfunctional, although those grown in Petri dishes (in vitro) do beat. Perhaps soon this mystery will be solved, and new advances in treatment will be commonplace.
Conducting System of the Heart
The myocardial conducting cells are concentrated in specific areas of the heart that make up the Conducting System. The components of the cardiac conducting system include the sinoatrial node, the atrioventricular node, the atrioventricular bundle, the atrioventricular bundle branches, and the Purkinje fibers (Figure \(\PageIndex{2}\)).
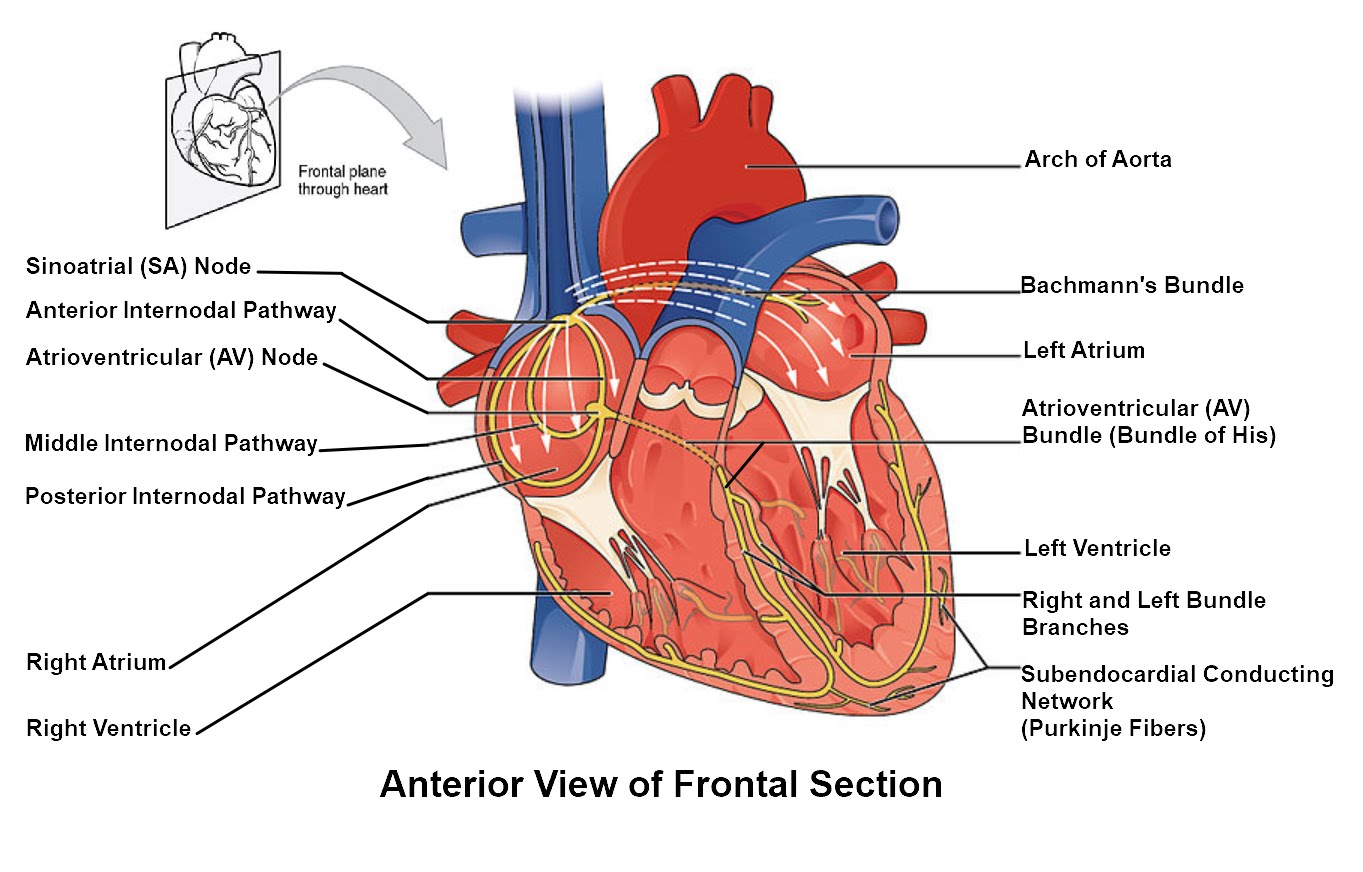 Figure \(\PageIndex{2}\): Conducting System of the Heart. Specialized conducting components of the heart include the sinoatrial node, the internodal pathways, the atrioventricular node, the atrioventricular bundle, the right and left bundle branches, and the Purkinje fibers. (Image credit: "Conducting System of the Heart" by Julie Jenks is licensed under CC BY 4.0 / A derivative from the original work)
Figure \(\PageIndex{2}\): Conducting System of the Heart. Specialized conducting components of the heart include the sinoatrial node, the internodal pathways, the atrioventricular node, the atrioventricular bundle, the right and left bundle branches, and the Purkinje fibers. (Image credit: "Conducting System of the Heart" by Julie Jenks is licensed under CC BY 4.0 / A derivative from the original work)Sinoatrial (SA) Node
Normal cardiac rhythm is established by the sinoatrial (SA) node, a specialized clump of myocardial conducting cells located in the posterosuperior walls of the right atrium in close proximity to the orifice of the superior vena cava. The myocardial conducting cells of the SA node have the highest inherent rate of electrical signal initiation, so the SA node is known as the pacemaker of the heart. It initiates the sinus rhythm, or normal electrical pattern followed by contraction of the heart.
This impulse spreads from its initiation in the SA node throughout the atria through specialized internodal pathways, to the atrial myocardial contractile cells and the atrioventricular node. The internodal pathways consist of three bands (anterior, middle, and posterior) of myocardial conducting cells that lead directly from the SA node to the next node in the conducting system, the atrioventricular node (see Figure \(\PageIndex{2}\)). The impulse takes approximately 50 ms (milliseconds, thousandths of a second) to travel between these two nodes. The relative importance of this pathway has been debated since the impulse would reach the atrioventricular node simply following the cell-by-cell pathway through the contractile cells of the myocardium in the atria. In addition, there is a specialized pathway called Bachmann’s bundle or the interatrial band that conducts the impulse directly from the right atrium to the left atrium. Regardless of the pathway, as the impulse reaches the atrioventricular septum, the insulating connective tissue of the cardiac skeleton prevents the impulse from spreading into the myocardial cells in the ventricles except at the atrioventricular node. Figure \(\PageIndex{3}\) illustrates the initiation of the impulse in the SA node that then spreads the impulse throughout the atria to the atrioventricular node.
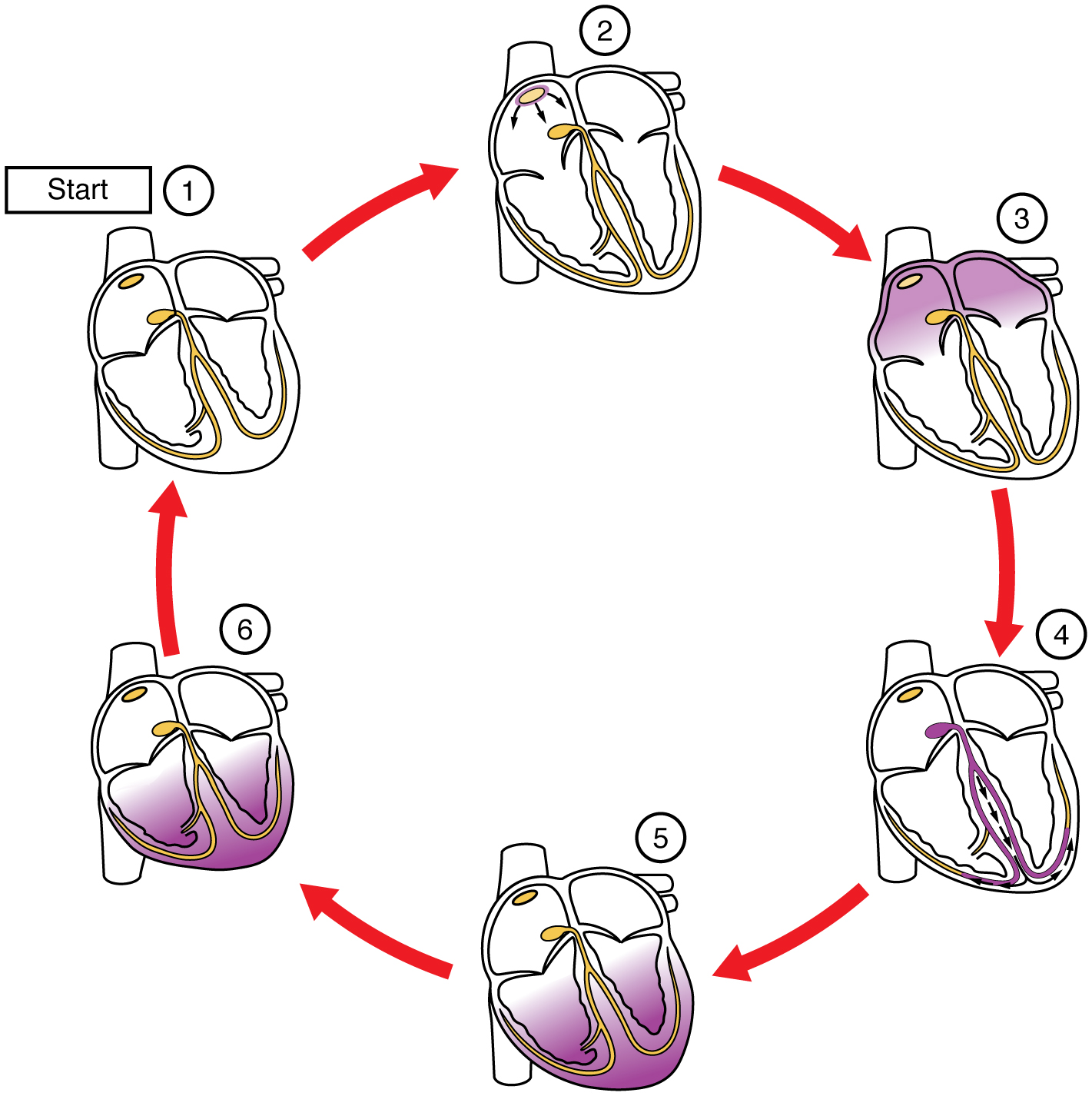
The electrical event (the wave of electrical signal) spreads the signal through the conducting system, but it is also the trigger for muscular contraction of the atria. The wave of electrical signal begins in the right atrium, and the impulse spreads across the superior portions of both atria and then down through the contractile cells. The contractile cells then begin contraction from the superior to the inferior portions of the atria, efficiently pumping blood into the ventricles.
Atrioventricular (AV) Node
The atrioventricular (AV) node is a second clump of specialized myocardial conducting cells, located in the inferior portion of the right atrium within the atrioventricular septum. The septum contains the dense irregular connective tissue of the cardiac skeleton that prevents the impulse from spreading directly to the ventricles without passing through the AV node. There is a critical pause before the AV node activates and transmits the electrical impulse to the atrioventricular bundle (see Figure \(\PageIndex{3}\), step 3). This delay in transmission is partially attributable to the small diameter of the cells of the node, which slow the impulse. Also, conduction between nodal cells is less efficient than between conducting cells. These factors mean that it takes the impulse approximately 100 ms to pass through the node. This pause is critical to heart function, as it allows the atrial cardiomyocytes to complete their contraction that pumps blood into the ventricles before the impulse is transmitted to the cells of the ventricle itself. With extreme stimulation by the SA node, the AV node can transmit impulses maximally at 220 per minute. This establishes the typical maximum heart rate in a healthy young individual. Damaged hearts or those stimulated by drugs can contract at higher rates, but at these rates, the heart can no longer effectively pump blood.
Atrioventricular Bundle (Bundle of His), Bundle Branches, and Subendocardial Conducting Network (Purkinje Fibers)
Arising from the AV node, the atrioventricular bundle, or bundle of His, proceeds into the interventricular septum before dividing into two atrioventricular bundle branches, commonly called the left and right bundle branches. The left bundle branch supplies the left ventricle, and the right bundle branch the right ventricle. Since the left ventricle is much larger than the right, the left bundle branch is also considerably larger than the right. Portions of the right bundle branch are found in the moderator band and supply the right papillary muscles. Because of this connection, each papillary muscle receives the impulse at approximately the same time, so they begin to contract simultaneously just prior to the remainder of the myocardial contractile cells of the ventricles. This is believed to allow tension to develop on the chordae tendineae prior to right ventricular contraction. There is no corresponding moderator band on the left. Both bundle branches descend and reach the apex of the heart where they connect with the subendocardial conducting network, or Purkinje fibers (see Figure \(\PageIndex{3}\), step 4). This passage takes approximately 25 ms.
The subendocardial conducting network, or Purkinje fibers, are additional myocardial conducting fibers that spread the impulse to the myocardial contractile cells in the ventricles. They extend throughout the myocardium from the apex of the heart toward the atrioventricular septum and the base of the heart at the top of the ventricles. The subendocardial conducting network has a fast inherent conduction rate, and the electrical impulse reaches all of the ventricular muscle cells in about 75 ms (see Figure \(\PageIndex{3}\), step 5). Since the electrical stimulus begins at the apex, the contraction also begins at the apex and travels toward the base of the heart, similar to squeezing a tube of toothpaste from the bottom. This allows the blood to be pumped out of the ventricles and into the aorta and pulmonary trunk. The total time elapsed from the initiation of the impulse in the SA node until initiation of impulse in the ventricles is approximately 225 ms.
Electrocardiogram
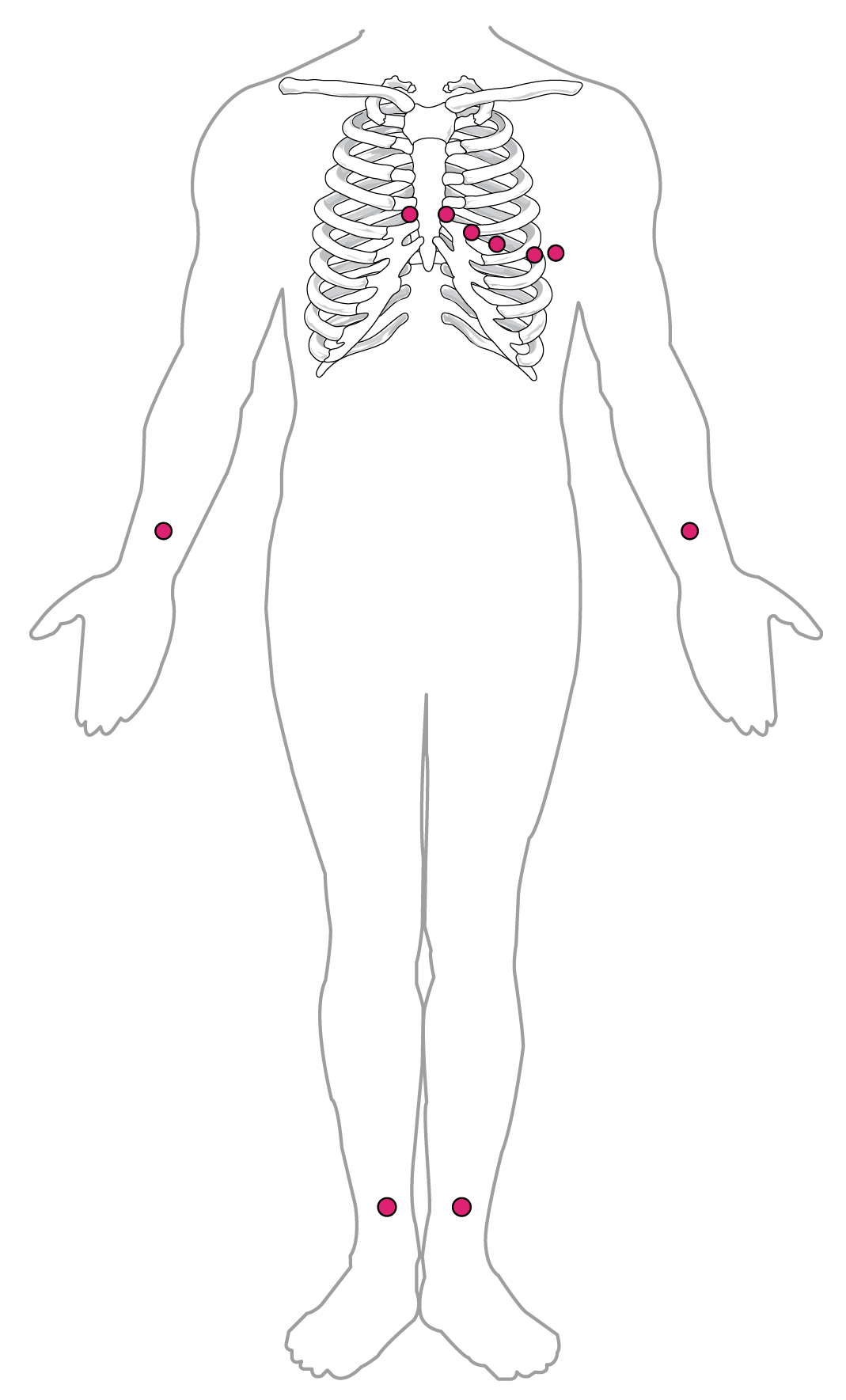
By careful placement of surface electrodes on the body, it is possible to record the complex, compound electrical signal of the heart. This tracing of the electrical signal is the electrocardiogram (ECG), also commonly abbreviated EKG (K coming from kardiology, the German spelling of cardiology). Careful analysis of the ECG reveals a detailed picture of both normal and abnormal heart function, and is an indispensable clinical diagnostic tool. The standard electrocardiograph (the instrument that generates an ECG) uses 3, 5, or 12 leads. The greater the number of leads an electrocardiograph uses, the more information the ECG provides. The term “lead” may be used to refer to the cable from the electrode to the electrical recorder, but it typically describes the voltage difference between two of the electrodes. The 12-lead electrocardiograph uses 10 electrodes placed in standard locations on the patient’s skin (Figure \(\PageIndex{4}\)). In continuous ambulatory electrocardiographs, the patient wears a small, portable, battery-operated device known as a Holter monitor, or simply a Holter, that continuously monitors heart electrical activity, typically for a period of 24 hours during the patient’s normal routine.
The ECG measures and graphs electrical activity over the time of each cardiac cycle that visually represents the spread of the electrical impulse through the conducting system (Figure \(\PageIndex{5}\)). The spread of electrical impulse from the SA node that initiates the cardiac cycle and causes atrial systole is visible in the P wave. The spread of electrical impulse from the AV node through the AV bundle and bundle branches is visible as the portion of the ECG with the largest amplitude (height), the QRS complex. The T wave (and a portion of the QRS complex) measure the electrical activity associated with repolarization, the physiological reset required to prepare for the next cardiac cycle.
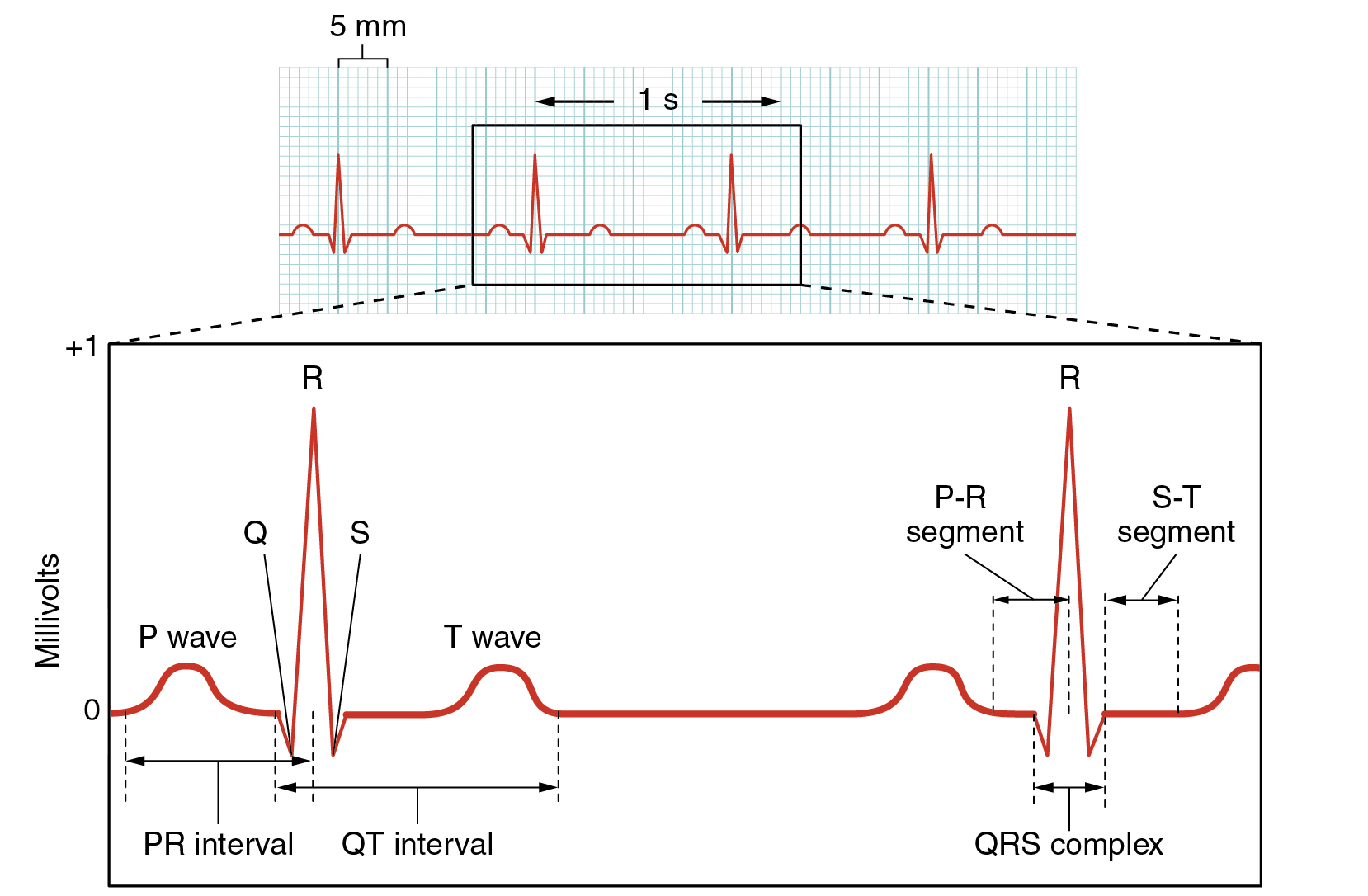
EVERYDAY CONNECTION
ECG Abnormalities
Occasionally, an area of the heart other than the SA node will initiate an impulse that will be followed by a premature contraction. Such an area, which may actually be a component of the conducting system or some other contractile cells, is known as an ectopic focus or ectopic pacemaker. An ectopic focus may be stimulated by localized ischemia (reduced blood flow to an area, depriving it of nutrients); exposure to certain drugs, including caffeine, digitalis, or acetylcholine; elevated stimulation by both sympathetic or parasympathetic divisions of the autonomic nervous system; or a number of disease or pathological conditions. Occasional occurrences are generally transitory and nonlife-threatening, but if the condition becomes chronic, it may lead to either an arrhythmia, a deviation from the normal pattern of impulse conduction and contraction, or to fibrillation, an uncoordinated beating of the heart.
While interpretation of an ECG is possible and extremely valuable after some training, a full understanding of the complexities and intricacies generally requires several years of experience. In general, the size of the electrical variations, the duration of the events, and detailed vector analysis provide the most comprehensive picture of cardiac function. For example, an amplified P wave may indicate enlargement of the atria, an enlarged Q wave may indicate a myocardial infarction (MI, heart attack), and an enlarged suppressed or inverted Q wave often indicates enlarged ventricles. T waves often appear flatter when insufficient oxygen is being delivered to the myocardium. An elevation of the ST segment above baseline is often seen in patients with an acute MI, and may appear depressed below the baseline when hypoxia is occurring.
As useful as analyzing these electrical recordings may be, there are limitations. For example, not all areas suffering a MI may be obvious on the ECG. Additionally, it will not reveal the effectiveness of the pumping, which requires further testing, such as an ultrasound test called an echocardiogram or nuclear medicine imaging. It is also possible for there to be pulseless electrical activity, which will show up on an ECG tracing, although there is no corresponding pumping action. Common abnormalities that may be detected by the ECGs are shown in Figure \(\PageIndex{6}\).
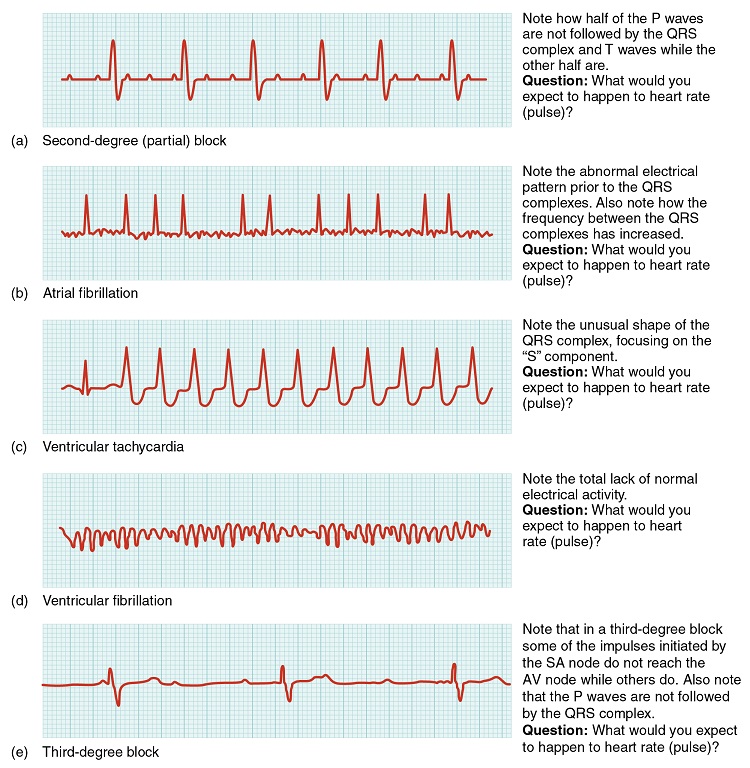
Figure \(\PageIndex{6}\): Common ECG Abnormalities. (a) In a second-degree or partial block, one-half of the P waves are not followed by the QRS complex and T waves while the other half are. (b) In atrial fibrillation, the electrical pattern is abnormal prior to the QRS complex, and the frequency between the QRS complexes has increased. (c) In ventricular tachycardia, the shape of the QRS complex is abnormal. (d) In ventricular fibrillation, there is no normal electrical activity. (e) In a third-degree block, there is no correlation between atrial activity (the P wave) and ventricular activity (the QRS complex). (Image credit: "Cardiac Arrhythmias" by OpenStax is licensed under CC BY 3.0)
EVERYDAY CONNECTION
Automated External Defibrillators (AEDs)
In the event that the electrical activity of the heart is severely disrupted, cessation of electrical activity or fibrillation may occur. In fibrillation, the heart beats in a wild, uncontrolled manner, which prevents it from being able to pump effectively. Atrial fibrillation (see Figure \(\PageIndex{6}\).b) is a serious condition, but as long as the ventricles continue to pump blood, the patient’s life may not be in immediate danger. Ventricular fibrillation (see Figure \(\PageIndex{6}\).d) is a medical emergency that requires life support, because the ventricles are not effectively pumping blood. In a hospital setting, it is often described as “code blue.” If untreated for as little as a few minutes, ventricular fibrillation may lead to brain death. The most common treatment is defibrillation, which uses special paddles to apply a charge to the heart from an external electrical source in an attempt to establish a normal sinus rhythm (Figure \(\PageIndex{7}\)). A defibrillator effectively stops the heart so that the SA node can trigger a normal conduction cycle. Because of their effectiveness in reestablishing a normal sinus rhythm, external automated defibrillators (EADs) are being placed in areas frequented by large numbers of people, such as schools, restaurants, and airports. These devices contain simple and direct verbal instructions that can be followed by nonmedical personnel in an attempt to save a life.

Disorders of the Conducting System
A heart block refers to an interruption in the normal conduction pathway. The nomenclature for these is very straightforward. SA nodal blocks occur within the SA node. AV nodal blocks occur within the AV node. Infra-Hisian blocks involve the bundle of His. Bundle branch blocks occur within either the left or right atrioventricular bundle branches. Hemiblocks are partial and occur within one or more fascicles of the atrioventricular bundle branch. Clinically, the most common types are the AV nodal and infra-Hisian blocks.
AV blocks are often described by degrees. A first-degree or partial block indicates a delay in conduction between the SA and AV nodes. This can be recognized on the ECG as an abnormally long PR interval. A second-degree or incomplete block occurs when some impulses from the SA node reach the AV node and continue, while others do not. In this instance, the ECG would reveal some P waves not followed by a QRS complex, while others would appear normal. In the third-degree or complete block, there is no correlation between atrial activity (the P wave) and ventricular activity (the QRS complex). Even in the event of a total SA block, the AV node will assume the role of pacemaker and continue initiating contractions at 40–60 contractions per minute, which is adequate to maintain consciousness. Second- and third-degree blocks are demonstrated on the ECG presented in Figure \(\PageIndex{6}\).
When arrhythmias become a chronic problem, the heart maintains a junctional rhythm, which originates in the AV node. In order to speed up the heart rate and restore full sinus rhythm, a cardiologist can implant an artificial pacemaker, which delivers electrical impulses to the heart muscle to ensure that the heart continues to contract and pump blood effectively. These artificial pacemakers are programmable by the cardiologists and can either provide stimulation temporarily upon demand or on a continuous basis. Some devices also contain built-in defibrillators.
Heart Rates
Heart rates (HRs) vary considerably, not only with exercise and fitness levels, but also with age. Newborn resting HRs may be 120 bpm. Resting HR gradually decreases until young adulthood and then gradually increases again with age.
Maximum HRs are normally in the range of 200–220 bpm, although there are some extreme cases in which they may reach higher levels. As one ages, the ability to generate maximum rates decreases. This may be estimated by taking the maximal value of 220 bpm and subtracting the individual’s age. So a 40-year-old individual would be expected to hit a maximum rate of approximately 180 bpm, and a 60-year-old person would achieve 160 bpm.
Heart rate is adjusted by chemical signals from the endocrine system as well as nervous signals that emerge from the medulla oblongata in the brainstem and travel to the heart via the vagus nerve (parasympathetic nervous system) to slow down the heart rate or via the sympathetic cardiac nerve (sympathetic nervous system) to speed up the heart rate. Both nerve pathways send branches to both the SA and AV nodes, and the sympathetic nervous system also sends neurons to portions of both the atria and ventricles. Without any nervous stimulation, the SA node would establish a sinus rhythm of approximately 100 bpm. Since resting rates are considerably less than this, it becomes evident that parasympathetic stimulation normally slows HR. Reducing the parasympathetic stimulation allows the heart rate to approach 100 bpm. Increasing the heart rate above 100 bpm involves sympathetic stimulation.
DISORDERS OF THE...
Heart: Abnormal Heart Rates
For an adult, normal resting HR will be in the range of 60–100 bpm. Bradycardia is the condition in which resting rate drops below 60 bpm, and tachycardia is the condition in which the resting rate is above 100 bpm. Trained athletes typically have very low HRs. If the patient is not exhibiting other symptoms, such as weakness, fatigue, dizziness, fainting, chest discomfort, palpitations, or respiratory distress, bradycardia is not considered clinically significant. However, if any of these symptoms are present, they may indicate that the heart is not providing sufficient oxygenated blood to the tissues. The term relative bradycardia may be used with a patient who has a HR in the normal range but is still suffering from these symptoms. Most patients remain asymptomatic as long as the HR remains above 50 bpm.
Bradycardia may be caused by either inherent factors or causes external to the heart. While the condition may be inherited, typically it is acquired in older individuals. Inherent causes include abnormalities in either the SA or AV node. If the condition is serious, a pacemaker may be required. Other causes include ischemia to the heart muscle or diseases of the heart vessels or valves. External causes include metabolic disorders, pathologies of the endocrine system often involving the thyroid, electrolyte imbalances, neurological disorders including inappropriate autonomic responses, autoimmune pathologies, over-prescription of beta blocker drugs that reduce HR, recreational drug use, or even prolonged bed rest. Treatment relies upon establishing the underlying cause of the disorder and may necessitate supplemental oxygen.
Tachycardia is not normal in a resting patient but may be detected in pregnant women or individuals experiencing extreme stress. In the latter case, it would likely be triggered by stimulation from the limbic system or disorders of the autonomic nervous system. In some cases, tachycardia may involve only the atria. Some individuals may remain asymptomatic, but when present, symptoms may include dizziness, shortness of breath, lightheadedness, rapid pulse, heart palpitations, chest pain, or fainting (syncope). While tachycardia is defined as a HR above 100 bpm, there is considerable variation among people. Further, the normal resting HRs of children are often above 100 bpm, but this is not considered to be tachycardia. Many causes of tachycardia may be benign, but the condition may also be correlated with fever, anemia, hypoxia, hyperthyroidism, hypersecretion of catecholamines, some cardiomyopathies, some disorders of the valves, and acute exposure to radiation. Elevated rates in an exercising or resting patient are normal and expected. Resting rate should always be taken after recovery from exercise. Treatment depends upon the underlying cause but may include medications, implantable cardioverter defibrillators, ablation, or surgery.
Concept Review
The heart is regulated by both neural and endocrine control, yet it initiates its own electrical impulse at an intrinsic rate followed by muscular contraction. The myocardial conducting cells within the heart establish the heart rate and transmit it through the myocardium. The myocardial contractile cells contract and propel the blood. The normal path of transmission for the conductive cells is the sinoatrial (SA) node, internodal pathways, atrioventricular (AV) node, atrioventricular (AV) bundle, bundle branches, and Purkinje fibers. Normal resting heart rate is in the range of 60 to 100 bpm.
Review Questions
Q. Which of the following distinguishes cardiac muscle cells from skeletal muscle cells?
A. Only cardiac muscle contains a sarcoplasmic reticulum.
B. Only cardiac muscle has striations.
C. Only cardiac muscle is capable of autorhythmicity.
D. Only cardiac muscle has a high concentration of mitochondria.
- Answer
-
Answer: C
Q. Which component of the heart's conducting system would have the slowest rate of firing?
A. atrioventricular node
B. atrioventricular bundle
C. bundle branches
D. subendocardial conducting network
- Answer
-
Answer: D
Critical Thinking Questions
Q. How does the delay of the impulse at the atrioventricular node contribute to cardiac function?
- Answer
-
A. It ensures sufficient time for the atrial muscle to contract and pump blood into the ventricles prior to the impulse being conducted into the lower chambers.
Q. How do intercalated discs aid contraction of the heart?
- Answer
-
A. Intercalated discs consist of desmosomes, fasciae adherens, and gap junctions. Desmosomes and fasciae adherens create strong connections between adjacent cardiac muscle cells that will contract against one another. Gap junctions within the intercalated discs allow impulses to spread from one cardiac muscle cell to another, allowing ions to flow between adjacent cells, propagating the electrical impulse to contract, and ensuring coordinated contractions.
Q. Why is it important that the conducting system initiates ventricular systole from the apex of the heart?
- Answer
-
A. Initiation of ventricular systole at the apex of heart allows the blood to be squeezed from the bottom-up toward the semilunar valves into the ascending aorta and pulmonary trunk.
Glossary
- artificial pacemaker
- medical device that transmits electrical signals to the heart to ensure that it contracts and pumps blood to the body
- atrioventricular bundle
- (also, bundle of His) group of specialized myocardial conductile cells that transmit the impulse from the AV node through the interventricular septum; form the left and right atrioventricular bundle branches
- atrioventricular bundle branches
- (also, left or right bundle branches) specialized myocardial conductile cells that arise from the bifurcation of the atrioventricular bundle and pass through the interventricular septum; lead to the Purkinje fibers and also to the right papillary muscle via the moderator band
- atrioventricular (AV) node
- clump of myocardial cells located in the inferior portion of the right atrium within the atrioventricular septum; receives the impulse from the SA node, pauses, and then transmits it into specialized conducting cells within the interventricular septum
- autorhythmicity
- ability of cardiac muscle to initiate its own electrical impulse that triggers the mechanical contraction that pumps blood at a fixed pace without nervous or endocrine control
- Bachmann’s bundle
- (also, interatrial band) group of specialized conducting cells that transmit the impulse directly from the SA node in the right atrium to the left atrium
- bundle of His
- (also, atrioventricular bundle) group of specialized myocardial conductile cells that transmit the impulse from the AV node through the interventricular septum; form the left and right atrioventricular bundle branches
- electrocardiogram (ECG)
- surface recording of the electrical activity of the heart that can be used for diagnosis of irregular heart function; also abbreviated as EKG
- heart block
- interruption in the normal conduction pathway
- interatrial band
- (also, Bachmann’s bundle) group of specialized conducting cells that transmit the impulse directly from the SA node in the right atrium to the left atrium
- intercalated disc
- physical junction between adjacent cardiac muscle cells; consisting of desmosomes, specialized linking proteoglycans, and gap junctions that allow passage of ions between the two cells
- internodal pathways
- specialized conductile cells within the atria that transmit the impulse from the SA node throughout the myocardial cells of the atrium and to the AV node
- myocardial conducting cells
- specialized cells that transmit electrical impulses throughout the heart and trigger contraction by the myocardial contractile cells
- myocardial contractile cells
- bulk of the cardiac muscle cells in the atria and ventricles that conduct impulses and contract to propel blood
- P wave
- component of the electrocardiogram that represents the initiation of electrical signal of the atria
- pacemaker
- cluster of specialized myocardial cells known as the SA node that initiates the sinus rhythm
- Purkinje fibers
- (also, subendocardial conducting network) specialized myocardial conducting fibers that arise from the bundle branches and spread the impulse to the myocardial contraction fibers of the ventricles
- QRS complex
- component of the electrocardiogram that represents the initiation of electrical signal of the ventricles
- sinoatrial (SA) node
- known as the pacemaker, a specialized clump of myocardial conducting cells located in the superior portion of the right atrium that has the highest inherent rate of depolarization that then spreads throughout the heart
- sinus rhythm
- normal contractile pattern of the heart
- subendocardial conducting network
- (also, Purkinje fibers) specialized myocardial conducting fibers that arise from the bundle branches and spread the impulse to the myocardial contraction fibers of the ventricles
- T wave
- component of the electrocardiogram that represents the repolarization of the ventricles
Contributors and Attributions
OpenStax Anatomy & Physiology (CC BY 4.0). Access for free at https://openstax.org/books/anatomy-and-physiology


