5.2: Cervicofacial Flap for Head and Neck Reconstruction
- Page ID
- 15455
OPEN ACCESS ATLAS OF OTOLARYNGOLOGY, HEAD & NECK OPERATIVE SURGERY
CERVICOFACIAL FLAPS IN HEAD AND NECK RECONSTRUCTION
Wayne Manana, Johan Fagan
Despite the advent of vascularized free tissue transfer flaps, cervicofacial flaps remain a ‘‘workhorse’’ for reconstruction of large cheek defects, especially in many regions where economic or infrastructural facilities limit the use of free tissue transfer flaps.
First described by Esser in 1918, the cervicofacial flap was first described in its modern form by Juri and Juri in 19791. It is a random flap comprising skin, subcutaneous fat, superficial neck veins, and platysma muscle. It includes the delicate superficial cervical fascia just deep to skin that envelopes the platysma and muscles of facial expression and extends from the epicranium above to the axilla and upper chest below; over the face it is represented by the superficial musculoaponeurotic system (SMAS).
The cervicofacial flap has a wide pedicle and can be employed to cover large anterolateral craniofacial defects. Superiorly it can reach the supraorbital margin, laterally the postauricular area, and medially up to the midline.
The cervicofacial flap provides an excellent color match as well as thickness and texture, can be performed under local anesthesia, and provides concomitant exposure for neck dissection and parotidectomy, has very acceptable donor site scars, scars are camouflaged in the cheek borders, and requires minimal surgical time and has minimal postoperative morbidity.
As it is a thin flap, additional bulk may be provided with a pectoralis major muscle flap placed deep to the cervicofacial flap. It may also be an option for patients not suitable for free tissue transfer flaps e.g. elderly of medically unfit patients who cannot tolerate prolonged operations.
Indications
- Superficial defects up to 14 x 10 cm
- All zones of the cheek, external ear
- Temporo-frontal and brow defects
- Orbital exenteration defects
- To provide skin cover in combination with muscle flaps e.g. pectoralis major and temporalis flaps
Contraindications
- Poor skin laxity
- Active wound infection
- Smoking: stop 2 weeks pre- and 1 week postoperatively
- Previous radiation to surgical site
- High risk of keloid
- Through-and-through defects particularly when the following structures are missing: masseter, mandible, maxilla and temporalis muscle
Advantages
- Technically simple
- Reliable, with low failure rate i.e. “very forgiving” even with inexperienced surgeons
- Single team approach
- Shorter anesthetic and surgical time than for free flaps with cost-saving and favoring unfit patients
- Can be performed under local anesthesia
- No postoperative ICU monitoring required
- Single-stage procedure
- Few perioperative complications and morbidity
- Versatile with multiple applications
- Proximity to the defect
- Good arc of rotation
- Covers defects up to 14 x 10 cm
- Excellent color and texture match with facial skin
- Camouflage scars in the cheek borders (inconspicuous and follows borders of facial subunits)
- Reduced costs: short operating time and hospital stay
- Simultaneous exposure for parotidectomy and neck dissection (Figure 1)
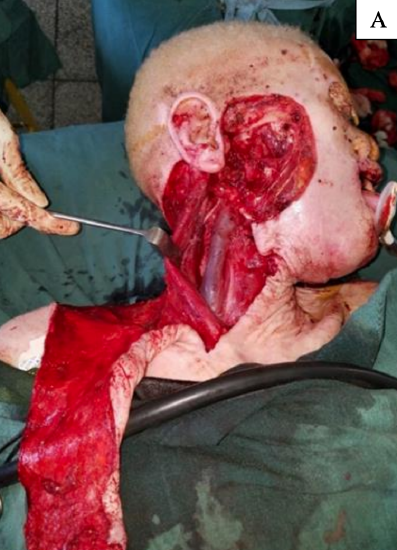
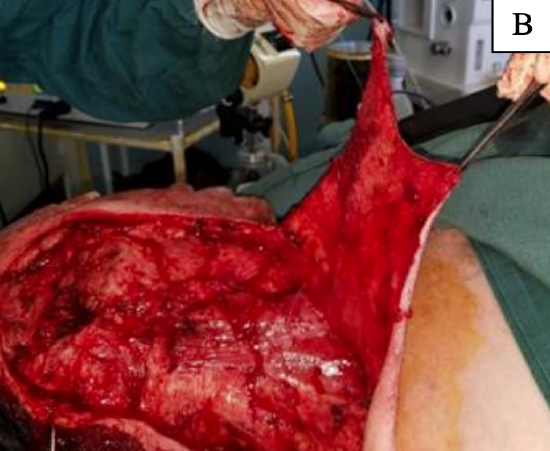
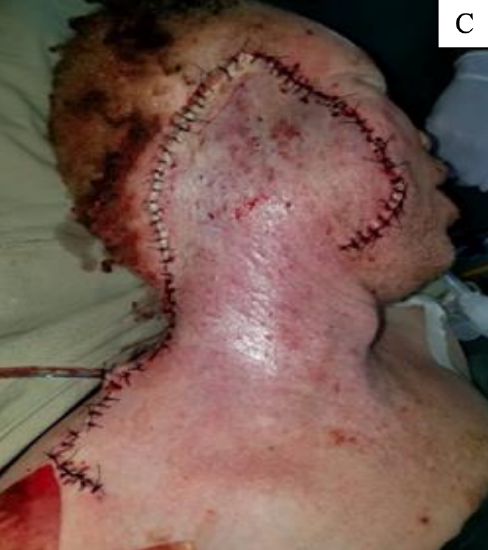
Figure 1: A, B demonstrate excellent exposure of cervicofacial flap for parotidectomy and neck dissection. C shows immediate postoperative view of cervicofacial flap for patient in image B
Relevant Anatomy
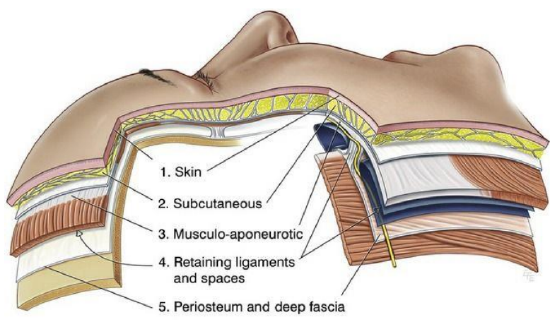
Figure 2: Layers of facial / cheek portion
The cervicofacial flap is divided into 2 portions, the facial (cheek) and the cervical portions. The cheek is a laminated structure (Figure 2) composed superficial-to-deep of:
- Epidermis
- Dermis
- Subcutaneous tissue
- Superficial muscular aponeurotic system (SMAS)
Immediately deep to the SMAS are branches of the facial nerve, the parotid gland, the buccal fat pad, and the mandible inferiorly. The branches of the facial nerve are protected beneath the SMAS in most locations. Thus, a surgeon who prefers to dissect beneath the SMAS layer must be familiar with facial nerve anatomy.
Identifying the borders of the cheek with other facial subunits is essential to camouflage the scar lines, including the infraorbital rim, nasolabial fold, preauricular crease, and inferior mandibular margin.
The SMAS was first described by Mitz and Peyronie in 19762. It encloses the platysma, risorius, triangularis, auricularis, occipitallis, and frontalis muscles. The SMAS is connected to the dermis by fibrous septae allowing movement of the skin for facial expression. It is confluent with the galea layer of the scalp and with the temporoparietal fascia in the temporal region. In the neck, the SMAS continues as the superficial cervical fascia. The surgical importance of the SMAS lies in its relationship with the facial nerve. The facial nerve branches are found deep to the fascia, whereas the sensory supply to the face is located superficial to it.
The vascularity of the SMAS has been debated for many years. Although anatomic studies report that the SMAS is an avascular layer, there is clinical evidence suggesting otherwise. Schaverien 3 used sequential dying and 3-dimensional computed tomographic angiography and venography on 24 hemifaces2. They reported that the SMAS is perfused by the transverse facial artery perforator branches on their route to the subdermal plexus. This finding is supported by the observation that skin necrosisis more frequently encountered in subcutaneous face-lifts where the SMAS is not included in the flap.
Arterial supply
The blood supply of the cervicofacial flap can be divided into 2 portions i.e. facial and cervical.
The facial portion of the flap receives its blood supply via the subdermal plexus in a random pattern from the facial artery, transverse facial artery and the superficial temporal artery via its orbito-zygomatic branch. It is dissected in a subcutaneous plane. A modification of this portion of the flap is to incorporate the SMAS as proposed by Kroll4 to enhance the vascularity by providing the flap with an axial blood supply via perforators from the transverse facial artery.
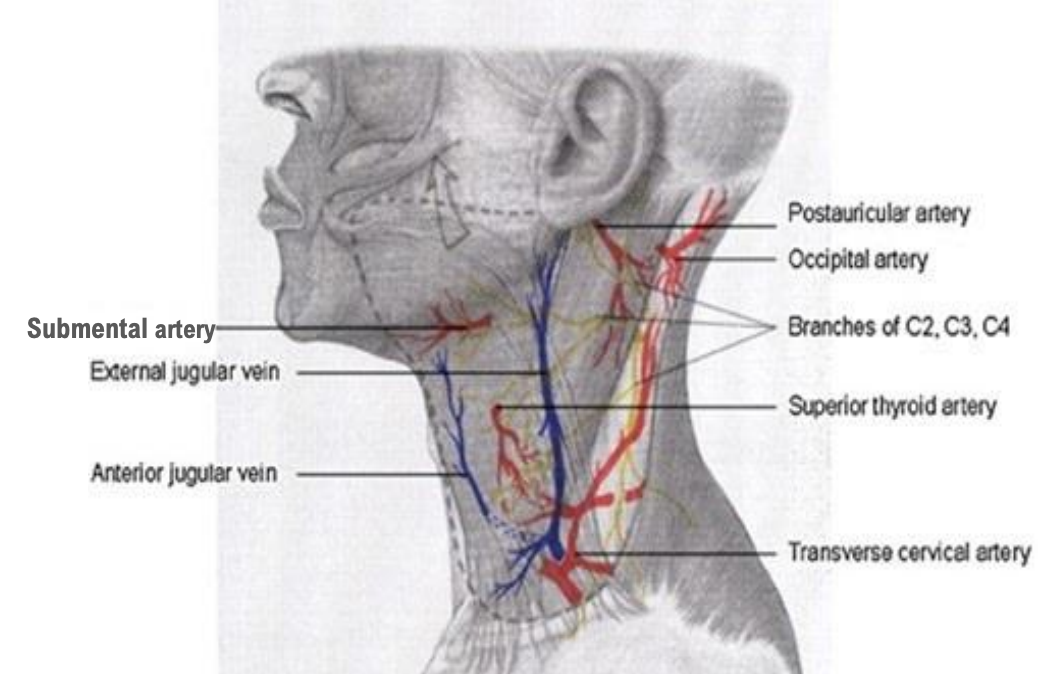
Figure 3: Neurovascular supply
The cervical portion of the flap, on the other hand, is dissected in a subplatysmal plane. By doing so, the platysma provides blood supply to this region. The platysma, as described by Hurwitz5, is supplied by the occipital and postauricular arteries posteriorly, the submental artery superiorly and medially, the superior thyroid artery centrally, and the transverse cervical artery inferiorly (Figure 3). However, during flap elevation, only the perforators from the submental artery are preserved.
Venous drainage
Both the facial and cervical portions have a venous system that accompanies the arteries. The venous drainage is however mainly provided by the external jugular vein and randomized dermal venous drainage (Figure 3). Enclosing the external jugular vein in the flap helps to reduce venous congestion and the risk of ischaemic necrosis.
Innervation
Sensory innervation is provided by branches of the greater auricular (C2-C3) and lesser occipital (C2) nerves. Careful dissection reduces the risk of paraesthesia, and patients should be informed of this potential complication.
Flap elevation
- The patient is placed in a supine position with the head turned to the opposite side and with a head ring to stabilize the head and shoulder roll to extend the neck
- Inject local anesthetic mixed with adrenaline in the subcutaneous plane
- Defects less than 3 x 3 cm can be closed under local anesthesia. Though supraSMAS dissection can easily be performed under local anesthesia, subplatysmal dissection is difficult to perform under local anesthesia because of the risk of bleeding and patient discomfort
- A deliberate attempt to camouflage the postoperative scars is achieved by placing incisions along the various grooves, facial subunits and shadows in the head and neck region
- The incisions generally follow an imaginary line drawn along the following landmarks (Figure 3)
- Nasolabial fold
- Infraorbital, subciliary or subtarsal
- Zygomatic arch/hairline
- Preauricular crease
- Retroauricular / posterior hair line
- Lateral border of trapezius
- Modification includes extension along deltopectoral groove
- The flap components depend on the anatomical location
- Superior to zygomatic arch: skin and subcutaneous tissue
- Inferior to zygomatic arch: skin, subcutaneous tissue, and SMAS
- Cervical portion: platysma and skin. The importance of including platysma in the cervical portion of the flap has been demonstrated by Hakim6
Reconstruction
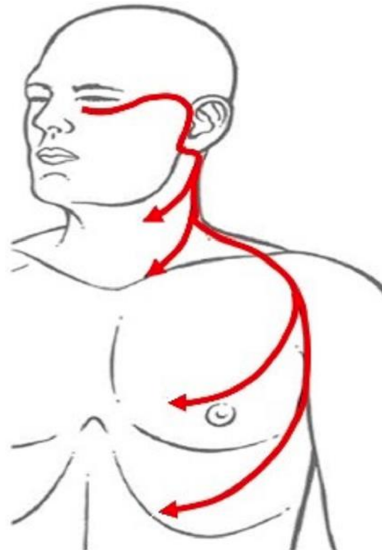
Figure 4: Incisions for cervicofacial flap
Roth7 divided the cheek into Zones to help determine the method of cheek reconstruction (Figure 4)
- Zone 1: Infraorbital and lower eyelids
- Zone 2: Preauricular and parotid
- Zone 3: Chin and antero-inferior cheek
The versatility of the cervicofacial flap is demonstrated by its ability to close defects in all the three zones.
Zone 1 presents a unique challenge because of its proximity to nearby vital structures. Inappropriate flap selection in Zone 1 reconstruction can cause ectropion and hence significant aesthetic and functional problems. Zones 2 and 3 provide the best opportunity for direct closure, especially in elderly patients. Zone 2 is reconstructed mainly with the cervical (platysmal) portion of the cervicofacial flap (Figures 7, 9, 11). The flap design for Zone 2 is also known as the Mustarde flap (Figures 5 and 6).
Examples of Zone 1 defect repairs
Case 1: Complete excision of lower eyelid for basal cell carcinoma and use of a cervicofacial flap to reconstruct cutaneous portion of lower eyelid (Figure 6)
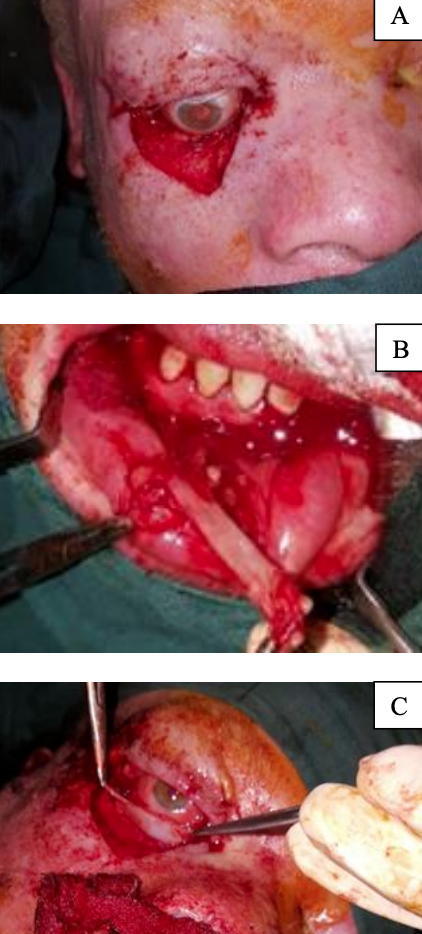
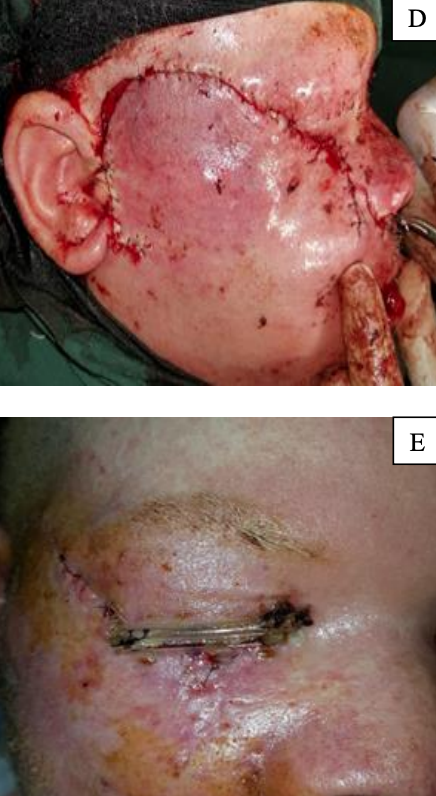
Figure 6: (A) Complete excision of lower eyelid in a patient with BCC; (B) Harvesting of labial mucosa to recreate conjunctiva; (C) Nasal septal cartilage used to recreate tarsal plate; (D) Cervicofacial flap to reconstruct cutaneous portion of lower eyelid; (E) Tarsorrhaphy to reduce risk of ectropion
Case 2: Excision of SCC lower eyelid and infraorbital region with reconstruction with cervicofacial flap (Figure 7)
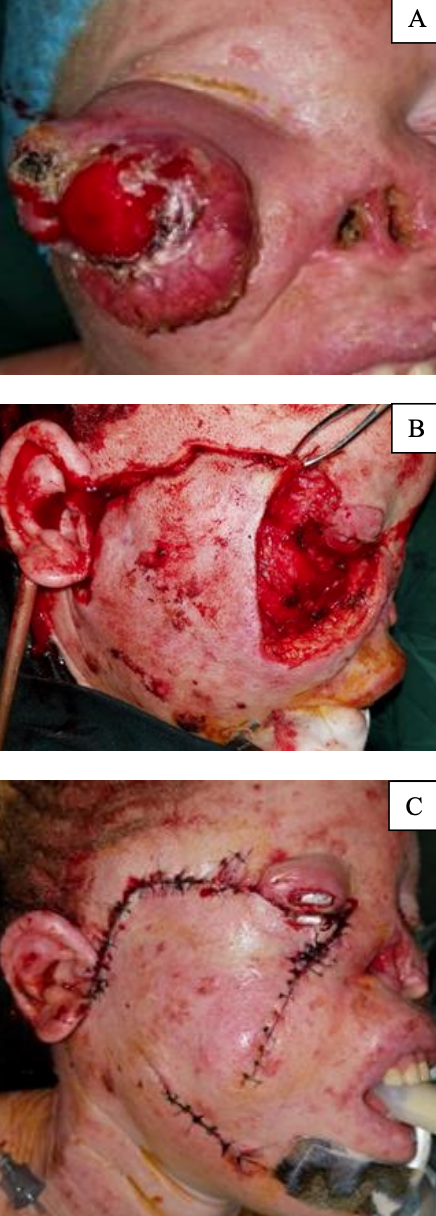
Figure 7: Excision of SCC of lower eyelid and infraorbital region with reconstruction with cervicofacial flap
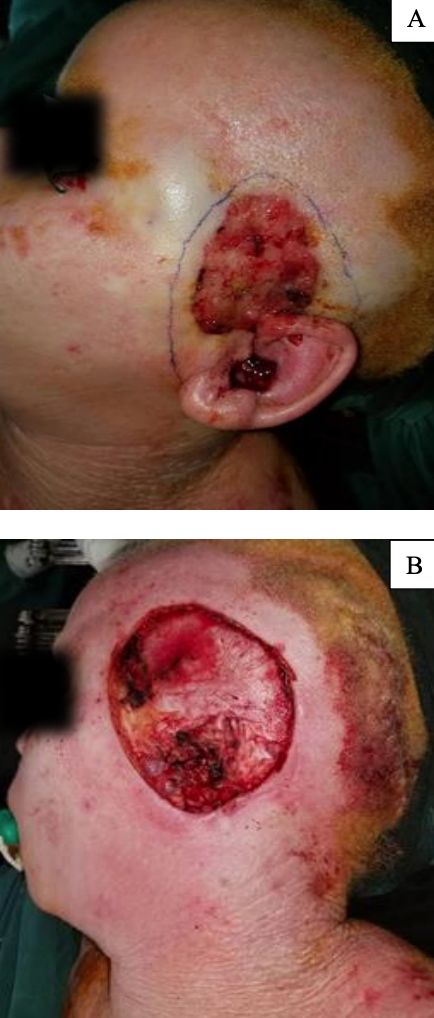
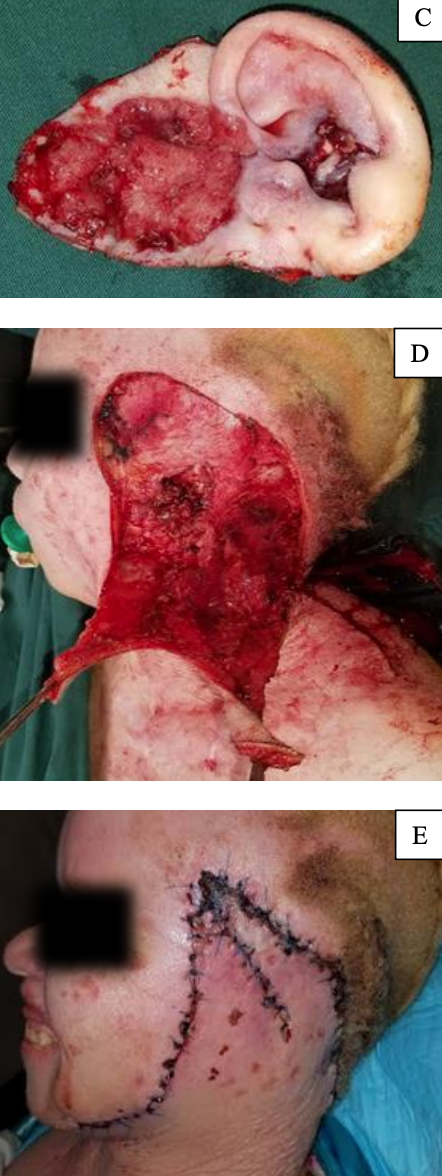
Figure 8: 10 cm Zone 2 defect reconstructed with a cervicofacial flap
Example of Zone 2 defect repair: 10 cm Zone 2 defect reconstructed with a cervicofacial flap. The underlying deep temporal fascia has been preserved. This demonstrates how the flap can be used for closure of temporal defects particularly when the ear has been sacrificed (Figure 8)
Modifications of Cervicofacial Flaps
Cervicothoracic flap: Inferior extension of over chest wall
The modern cervicofacial flap has facial and cervical portions; however, when a greater arc of rotation is needed, a pectoral extension can be used.
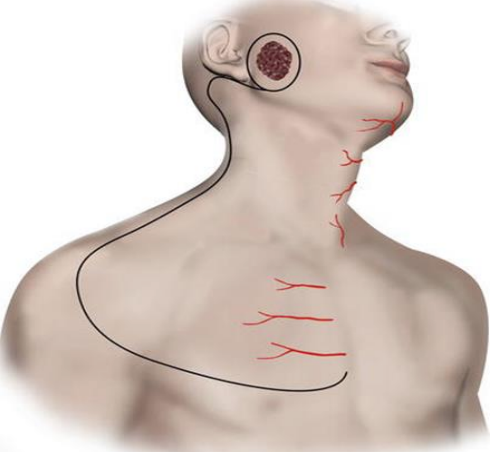
Figure 9: Cervicothoracic flap design, a modification of the cervicofacial flap that includes the deltopectoral flap 10
The cervicothoracic flap was first described by Conley 9 in 1960, and later by Garrett 10 in 1966 (Figure 9). In addition to the blood supply to the cervicofacial flap, the cervicothoracic flap also receives blood supply via perforators from the internal mammary artery. In order to preserve the internal mammary perforators, the medial extent of the flap elevation should not extend beyond 2 cm from the lateral sternal border.
The facial and cervical portions of the flap are raised in a similar manner to the conventional cervicofacial flap, with the pectoral portion raised in a subcutaneous plane. The cervicothoracic flap can be extended inferiorly to the costal margin, but most recommend not extending the flap beyond the nipple.
When comparing outcomes of the cervicothoracic and the conventional cervicofacial flap, Moore 11 and Liu 12 reported similar complication rates for the two flaps.
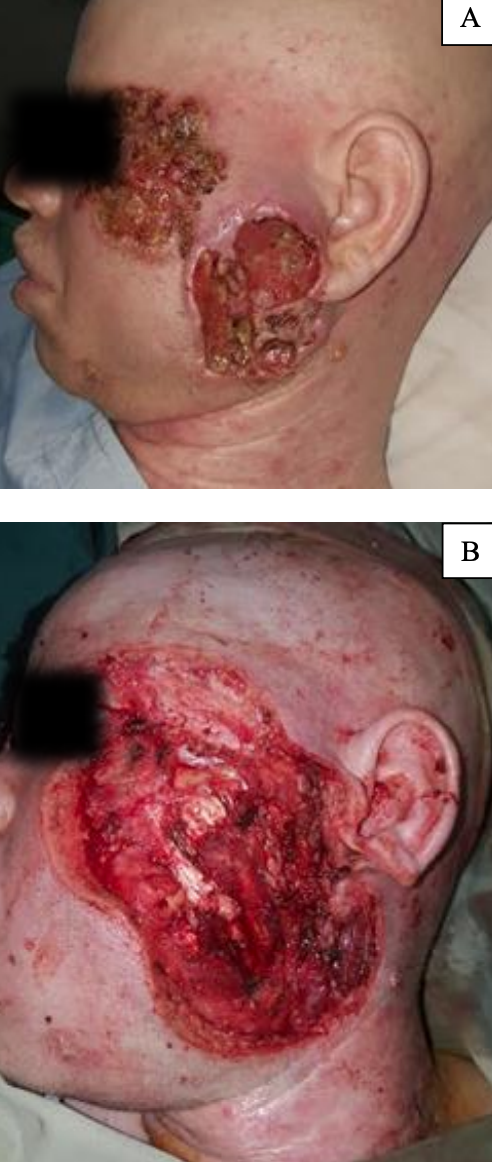
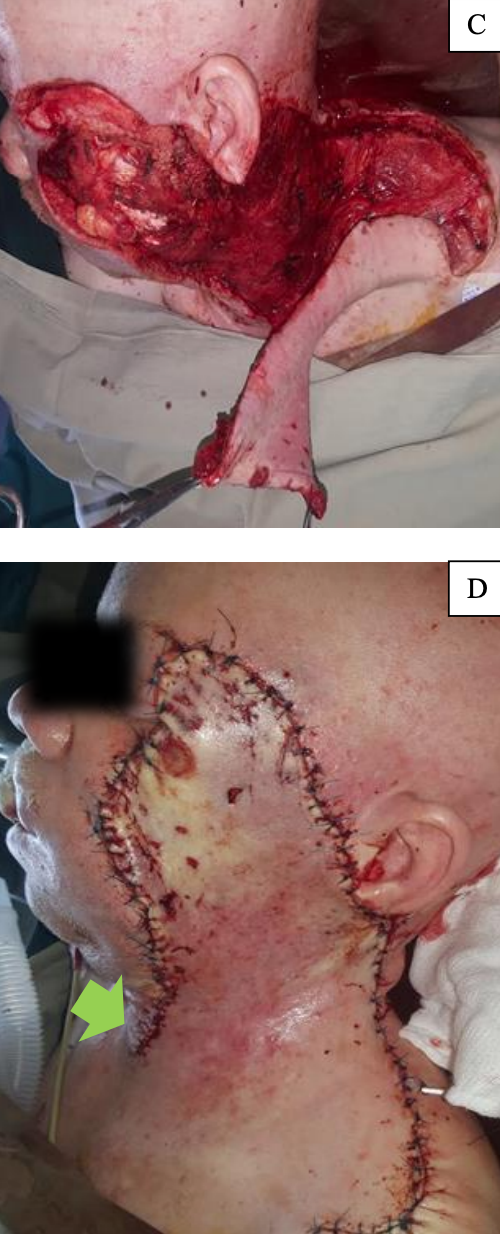
Figure 10: 16 x 10 cm Zone 2/3 defect closed with cervicothoracic flap. The anterior release incision (green arrow) greatly increases the flap coverage by combining advancement and rotational movements. The orange arrow show distal tip necrosis healing by secondary intention
Example of Zone 2 and 3 defect repair: Cervicothoracic flap used for 16 x 10 cm Zone 2/3 defect (Figure 10)
Subcutaneous vs. sub-SMAS dissection in facial portion of flap
The dissection plane in the facial portion of the flap can be either in the subcutaneous or sub-SMAS levels. The incorporation of the SMAS can theoretically change the blood supply of the flap from a random to an axial pattern via perforators from the transverse facial artery. This benefit was first advocated by Barton and Zilmer at the American Society of Plastic and Reconstructive Surgery meeting in 1982 and later published by Kroll 4 in 1994. However, this theoretical benefit was challenged in Whetzel and Stevenson's 13 cadaver study, in which they found no difference in blood supply based on ink-injection patterns between the two skin flaps. Jacono 14 conducted a retrospective clinical study to compare these 2 techniques, and reported no difference in skin necrosis, except in patients with a history of tobacco smoking. Moreover, dissection beneath the SMAS layer likely increases the risk of injuring the facial nerve. The intimacy of the nerve to the SMAS and the relatively high success rate of the subcutaneous dissection technique has many surgeons questioning the value of incorporating the SMAS.
Anterior release incision and posteriorly based cervicofacial flap
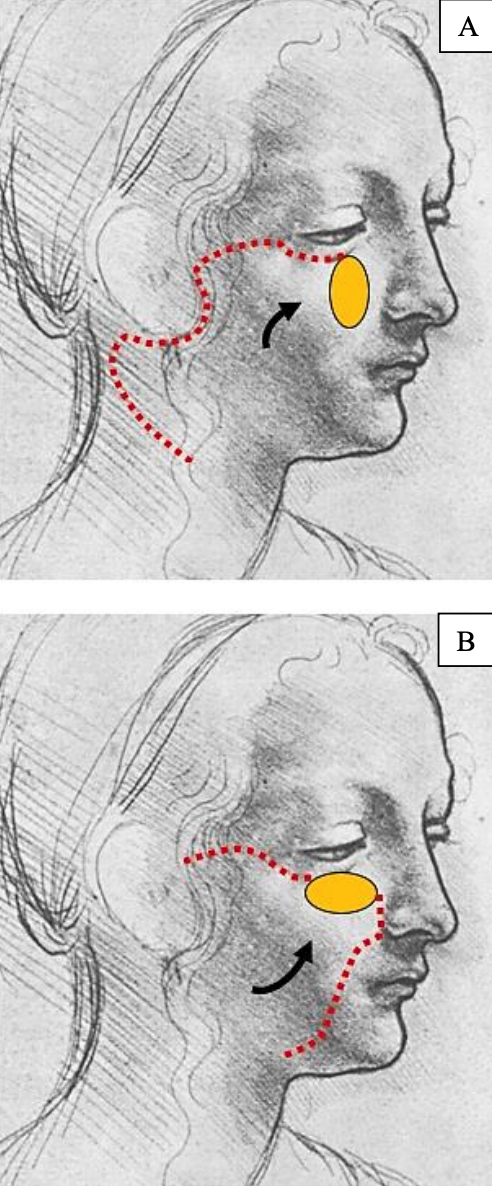
Figure 11: (A) illustrates the typical anteriorly based cervicofacial flap; note the vertical orientation of the infraorbital defect. (B) shows a reverse/posteriorly based cervicofacial flap; note the horizontal orientation of the infraorbital defect. The incision for the reverse cervicofacial flap follows the nasolabial groove
The flap can be based posteriorly. Boyette and Vural 15 advocate the use of the reverse flap for superomedial cheek defects in which the horizontal axis is greater than the vertical (Figure 11). They described its use in 6 cases with a mean flap size of 6 x 4.9 cm and reported no flap failures; 3 patients had ectropion not requiring surgical intervention. The investigators opine that a reverse cervicofacial flap for horizontally oriented defects helps to decrease flap tension and minimises excessive skin excision for standing cone deformities.
It is the 1st author’s (W.M.) experience that combining the anteriorly based cervicofacial flap with an anterior release incision greatly increases the arc of rotation and permits closure of bigger defects of up to 16 x 10 cm. This is however associated with a higher incidence of distal flap necrosis as the submental artery is usually sacrificed in the process. Distal flap necrosis has however not been a concern as the wounds eventually heal by secondary intention (Figures 10, 11, 12).
Example of Zone 2/3 defect repaired combining a conventional cervicofacial flap and anterior release incision (Figure 12)
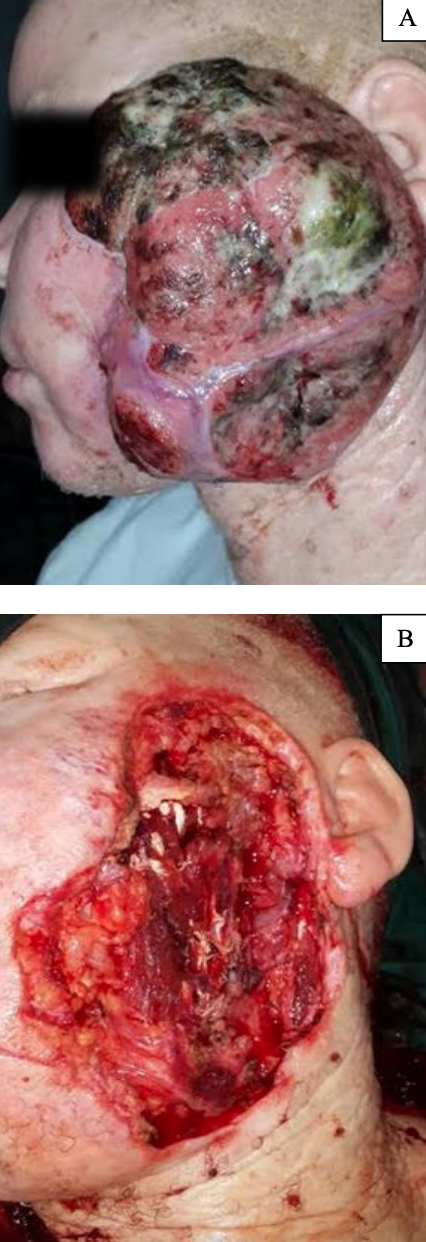
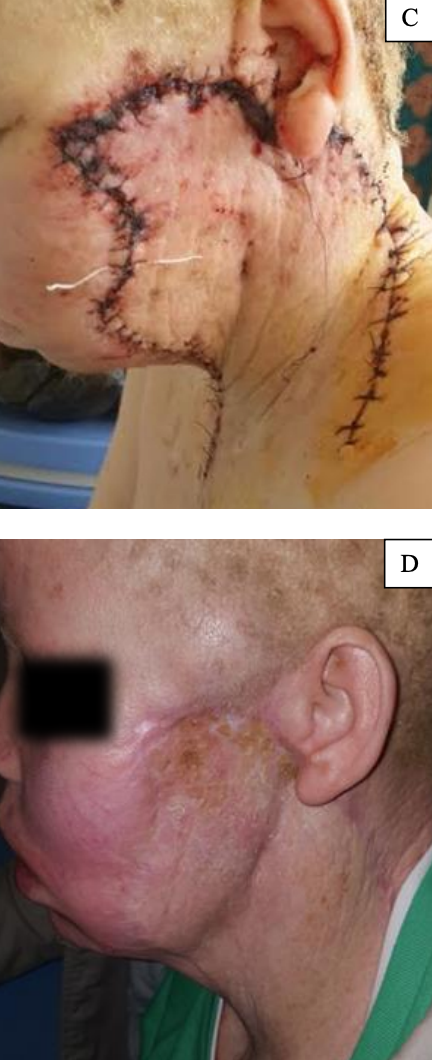
Figure 12: Large squamous cell carcinoma closed by modified cervicofacial flap, combining a conventional cervicofacial flap and an anterior release incision
Combination cervicofacial flap with other flaps for complex defects
Cervicofacial flaps generally do not provide much bulk. For deeper defects, additional bulk can be achieved by placing a free fat graft or a muscle flap e.g. temporalis or pectoralis major, deep to the flap. For complex reconstructions, a cervicofacial flap may also be used in combination with free flaps e.g. free fibula, anterolateral thigh or radial forearm flaps.
Tissue expansion
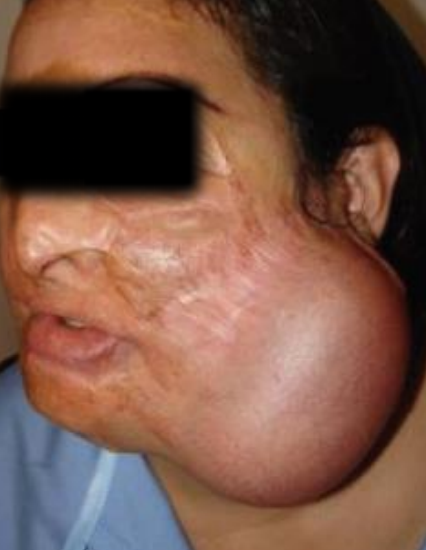
Figure 13: Tissue expander used to increase size of cervicofacial flap 16
Prior tissue expansion can be employed to increase the size of a cervicofacial flap for larger defects (Figure 13).
Postoperative care
- Keep the head elevated to reduce edema of the flap
- Remove drains when output is <50 mL/24 hrs
- Remove dressing 2 days postoperatively to inspect the wound
- Should distal tip necrosis occur, consider debridement followed by daily dressing until the wound heals by secondary intention
Complications
Patients with decreased skin laxity, sebaceous skin, higher Fitzpatrick grading, or a history of keloids may present a greater surgical challenge. Proper patient selection, gentle handling of the tissues, and good hemostasis minimize complications. Hematomas require immediate evacuation because they may jeopardize the blood supply to the flap, potentiating the risk of necrosis. Infections are treated with drainage and antibiotics.
Complications specifically related to cervicofacial flaps are standing cone deformity, long-term ectropion, distal tip necrosis, and nerve injuries.
Standing cone deformity or dog-ear cosmetic problem: This is best addressed at the time of the initial surgery.
Postoperative ectropion: This occurs when a flap is extended to the infraorbital region. Postoperative edema causes eversion of the lower eyelid, and as healing proceeds, scarring and fibrosis may ensue and maintain the eyelid in an everted position. Extending the flap superiorly to the lateral canthus and suturing it to the periosteum reduces the risk of persistent ectropion.
Another way to prevent this complication is the use of a temporary suspension suture (Frost suture). A 4-0 nylon suture is passed through the inferior tarsal plate and then secured to the supraorbital region. The free ends are then joined and form a sling counteracting caudal tension.
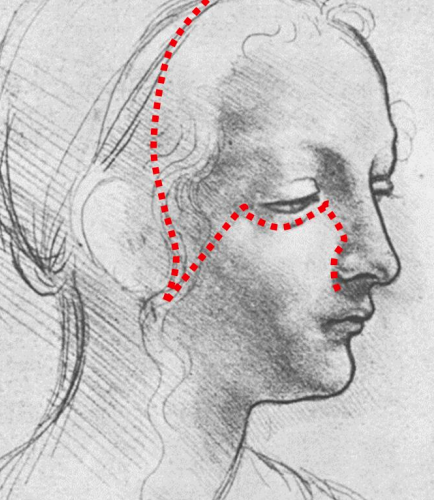
Figure 14: Yin-Yang rotation
Another way to prevent ectropion is to use the Yin-Yang rotation, as described by Belmahi, 17 for cases in which minimal skin laxity is appreciated (Figure 14). A temporoparietal scalp flap is raised and rotated in the opposite direction to the cervicofacial flap to close the preauricular cutaneous defect. Raising the additional temporoparietal scalp flap helps to minimize wound tension and provides cephalic anchorage to the facial flap, which reduces the incidence of ectropion.
Distal tip necrosis (Figure 11): This may occur due to unreliable random pattern vascularization of the flap. The reported incidence is about 15%. Risk factors include a history of smoking, radiation therapy, thinner skin in older patients and diabetes. Although raising the flap with the SMAS may theoretically increase the blood supply, this has not been borne out by clinical studies. Incorporating the SMAS for patients without risk factors may therefore not be beneficial. However, those with associated risk factors may find significant benefits as demonstrated in a study by Jacono14. Another measure to reduce distal tip necrosis is to include the external jugular vein with the flap to improve the venous drainage. When flap tension is unavoidable because of the size of the defect, additional grafts or flaps should be employed to avoid undue tension.
Facial nerve injury: This is a dreadful complication and has major functional and aesthetic consequences. A detailed knowledge of facial and facial nerve anatomy, use of blunt dissection and bipolar electrocautery, are mainstays of preventing injury. The incidence of facial nerve injury is low, and when it occurs, it is usually transient. Facial nerve injury should therefore initially be managed conservatively. Persistent paralysis without improvement may warrant facial reanimation surgery.
References
- Juri J, Juri C. Advancement and rotation of a large cervicofacial flap for cheek repairs. Plast Reconstr Surg 1979; 5:692-6
- Mitz V, Peyronie M. Plastic & Reconstructive Surgery 1976. 58(1):80-8
- Schaverien MV, Pessa JF, Saint-Cyr M, et al. The arterial and venous anatomies of the lateral face lift flap and the SMAS Plast Reconstr Surg. 2009; 123(5): 1581-7
- Close SS, Kroll GP, Reece G. et al. Deep-plane cervicofacial rotationadvancement flap for reconstruction of large cheek defects Plast Reconstr Surg. 1994;94(1):88-93
- Close DJ, Hurwitz JA, Rabson JW, Futrell JW. The anatomic basis for the platysma skin flap. Plast Reconstr Surg. 1983;72(3): 302-12
- Hakim SG, Jacobsen HC, Aschoff HH. et al. Including the platysma muscle in a cervicofacial skin rotation flap to enhance blood supply for reconstruction of vast orbital and cheek defects: anatomical considerations and surgical technique Int J Oral Maxillofac Surg. 2009;38(12):1316-9
- Roth DA, Longaker MT, Zide BM. Cheek surface reconstruction: best choices according to zones Operat Tech Plast Reconstr Surg. 1998;5(1):26-36
- Cabrera R, Zide BM, Cheek Reconstruction: In: Aston SJ, Beasley RW, Thorne CHM, Eds. Grabb and Smith’s Plastic Surgery, 5th ed. Philadelphia: Lippincott 1999: pp. 501-512
- Conley JJ. The use of regional flaps in head and neck surgery. Ann Otol Rhinol Laryngol. 1960; 69:1223-34
- Garrett WS, Giblin TR, Hoffman GW. Closure of skin defects of the face and neck by rotation and advancement of cervicopectoral flaps. Plast Reconstr Surg. 1966; 38:342–6
- Moore BA, Wine T, Netterville JL. Cervicofacial and cervicothoracic rotation flaps in head and neck reconstruction. Head Neck. 2005; 12:1092- 101
- Liu FY, Xu ZF. The versatile applications of cervicofacial and cervicothoracic rotation flaps in head and Neck surgery. World J Surg Oncol. 2011; 9:135
- Whetzel TP, Stevenson TR. The contribution of the SMAS to the blood supply in the lateral face lift flap. Plast Reconstr Surg. 1997; 100:1011-8
- Jacono AA, Rousso JJ, Lavin TJ. Comparing rates of distal edge necrosis in deep-plane vs subcutaneous cervicofacial rotation-advancement flaps for facial cutaneous Mohs defects. JAMA Facial Plast Surg. 2014;16(1):31
- Boyette JR, Vural E. Cervicofacial advancement-rotation flap in midface reconstruction: Forward or reverse? Otorhinolangol Head Neck Surg. 2011.144(2):196-200
- Ashab Yamin MR, Mozafari N, Mozafari M, Razi Z. Reconstructive surgery of extensive face and neck burn scars using tissue expanders. World J Plast Surg 2015;4(1):40-9
- Belmahi A, Oufkir A, Bron T, Ouezzani S. Reconstruction of cheek skin defects by the ‘Yin-Yang’ rotation of the Mustardé flap and the temporoparietal scalp. J Plast Reconstr Aesthet Surg. 2009 Apr;62(4):506-9
Other flaps described in The Open Access Atlas of Otolaryngology Head & Neck Operative Surgery
- Pectoralis major flap
- Buccinator myomucosal flap
- Buccal fat pad flap
- Nasolabial flap
- Temporalis muscle flap
- Deltopectoral flap
- Paramedian forehead flap
- Upper and lower trapezius flaps
- Submental artery island flap
- Supraclavicular flap
- Latissimus dorsi flap
- Local flaps for facial reconstruction
- Radial free forearm flap
- Free fibula flap
- Rectus abdominis flap
- Anterolateral free thigh flap
- Thoracodorsal artery scapular tip (TDAST) flap
- Principles and technique of microvascular anastomosis for free tissue transfer flaps in head and neck reconstructive surgery
Author
Wayne Manana BA (Zim), BDS (UZ), MDS OMFS (UoN)
Specialist Oral & Maxillofacial Surgeon
Department of Maxillofacial Surgery
University of Zimbabwe College of Health Sciences
Harare, Zimbabwe
docmanana@gmail.com
Editor and Author
Johan Fagan MBChB, FCS (ORL), MMed
Professor and Chairman
Division of Otolaryngology
University of Cape Town
Cape Town, South Africa
johannes.fagan@uct.ac.za


