5.20: Deltopectoral and Cervicodeltopectoral Fasciocutaneous Flaps for Head and Neck Reconstruction
- Page ID
- 52871
OPEN ACCESS ATLAS OF OTOLARYNGOLOGY, HEAD & NECK OPERATIVE SURGERY
DELTOPECTORAL AND CERVICODELTOPECTORAL FASCIOCUTANEOUS FLAPS FOR HEAD AND NECK RECONSTRUCTION
Johan Fagan, Ottie van Zy
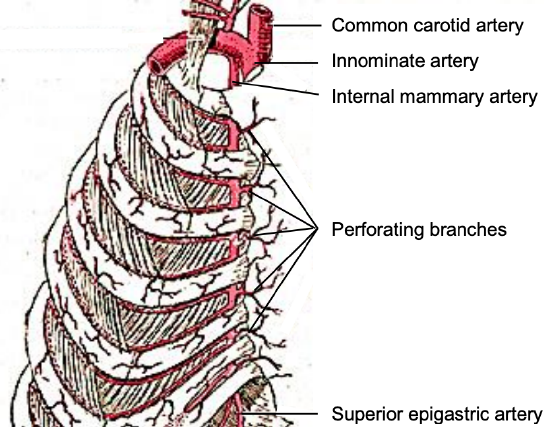
Figure 1: Perforators originate from the internal mammary artery
Fasciocutaneous flaps raised from the anterior chest wall below the clavicle are well suited to reconstructing skin defects in the head and neck region in terms of proximity, skin texture and skin colour. Bakamjian first described the deltopectoral (DP) flap in 1965 as a head and neck reconstructive option1. The DP flap is a pedicled axial fasciocutaneous flap based on the internal mammary artery perforator arteries (IMAP) (Figure 1) although the perforator(s) are usually not identified, dissected, nor mobilized. However, if the flap is raised as an island flap with complete division of the skin pedicle and the perforators are dissected out, it becomes an internal mammary artery perforator flap (IMAP flap); the IMAP flap will be discussed in a separate chapter.
Surgical anatomy
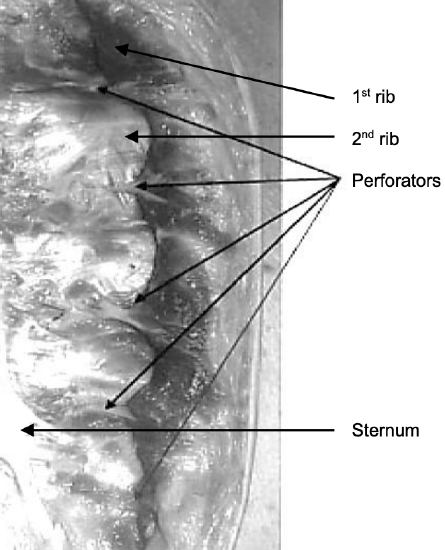
Figure 2: Left half of sternum: Perforators emerge approximately 7 mm lateral to sternum 2
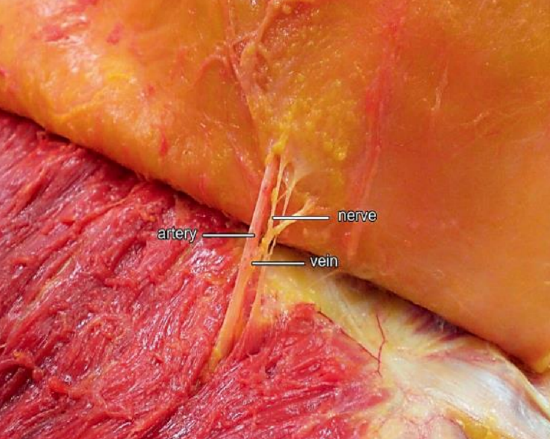
Figure 3: Perforator branch of IMA 3
Paired internal mammary (IMA) arteries arise from the subclavian artery and course inferiorly on the inside of the rib cage approximately 1 cm lateral to the sternum (Figure 1). The perforating branches of the IMA emerge about 7 mm lateral to the sternum through five or six intercostal spaces; they are 0.25-3.5mm in diameter (Figures 1, 2, 3). The so-called “principal perforator” traverses the 2nd intercostal space; the mean diameters of this artery and vein are just over 1.8 mm2. The vessels subsequently course laterally to provide blood supply to the pectoralis major muscle and overlying skin; the 2nd - 4th perforators send branches to the breast.
The blood vessels to the fasciocutaneous flap run in a plane superficial to the fascia that covers the pectoralis major and deltoid muscles; hence the importance of raising these flaps in a plane deep to the deltoid and pectoral fasciae.
The DP flap that is located medial to the deltopectoral groove (junction between pectoralis and deltoid muscles) has a reliable axial blood supply and can be transferred with a high probability of survival.
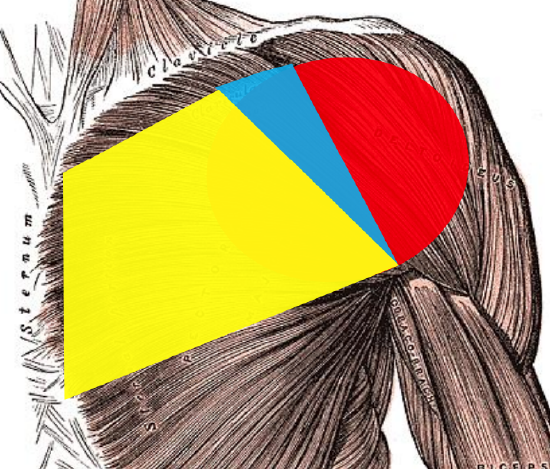
Figure 4a: 1st angiosome: yellow; 2nd angiosome: blue; 3rd angiosome: red
Three angiosomes (arterial/vascular territories) are included when extending a DP flap laterally over the deltoid (Figure 4a):
- 1st Angiosome: IMA perforators extend from the lateral border of the sternum up to close to the deltopectoral groove (This area also receives some blood supply from musculocutaneous perforators arising from the pectoralis major muscle)
- 2nd Angiosome: A small, but variable region of skin below the clavicle and medial to the deltopectoral groove is supplied by the thoracoacromial artery via a small direct cutaneous artery
- 3rd Angiosome: The area over the deltoid muscle is supplied by musculocutaneous perforators arising from the deltoid branch of the thoracoacromial artery and the anterior circumflex humeral artery
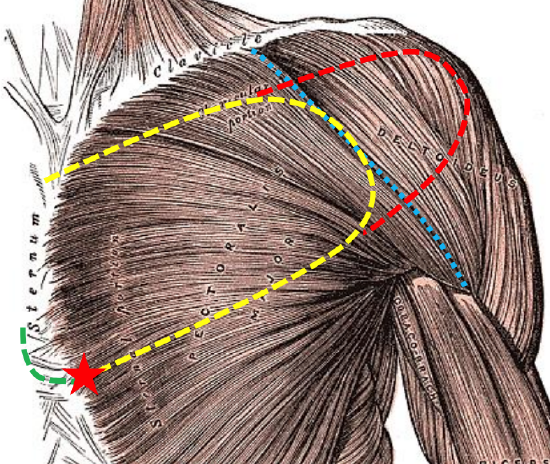
Figure 4b: Typical outline of a DP flap (yellow); deltopectoral groove (blue); lateral extension of flap (red); incision to gain additional length (green); axis of rotation (red star)
As one moves laterally and away from the IMA source-perforators the pressure gradient diminishes. Taylor 4 showed with the angiosome concept that the blood supply of the main angiosome and adjacent angiosome (2nd angiosome) is reliable, but that an additional angiosome, such as that over the deltoid muscle, is at risk of undergoing ischaemic necrosis. Once a DP flap is extended lateral to the deltopectoral groove, its reliability is therefore diminished (Figure 4b).
More dominant supply vessels to the 2nd and 3rd angiosomes increase the risk of necrosis in the 3rd/distal angiosome once these supply vessels are divided and the flap becomes dependent only on the vessels from the 1st angiosome. The further laterally the flap is raised the more random and unreliable the vascular supply and the less reliable the flap becomes. If very large deltoid perforators are therefore encountered when elevating the distal part of the flap, one should be more inclined to delay the flap as delaying or supercharging the flap lateral to the deltopectoral groove improves survival of its distal part (see later).
Deltopectoral Flap (Figure 4)
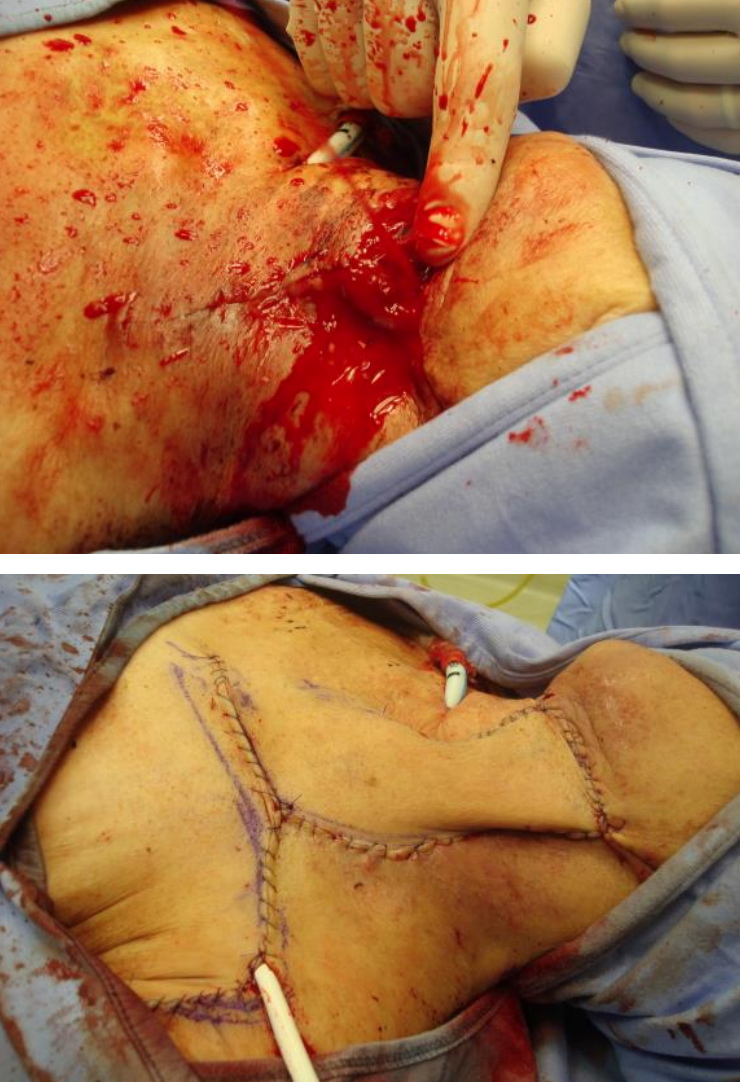
Figures 5a, b: DP flap used to cover skin defect that caused a carotid blowout
The DP flap has largely been supplanted by the pectoralis major, free tissue transfer and, to a lesser extent, latissimus dorsi flaps. It may however be useful in specific situations when other options are unavailable or have been exhausted e.g. reconstruction of cervical skin defects; skin cover of an exposed carotid artery (Figures 5a, b); closure of a pharyngocutaneous fistula; or (staged) hypopharynx reconstruction (very rarely). Contraindications include an internal mammary artery that has previously been used for coronary artery bypass surgery; and prior trauma or surgery (mastectomy, pacemaker, pectoralis major flap) to the anterior chest wall.
Advantages
- Provides large area of skin cover
- Better color and texture match compared to free tissue transfer flaps from distant sites
- Less bulky than pectoralis major flap
- Technical simplicity
Disadvantages
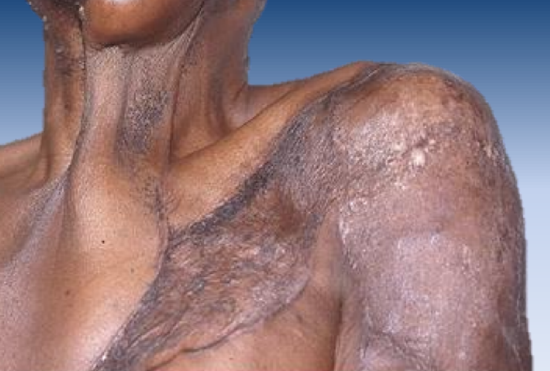
Figure 6: Scarring from skin grafts and flap contracture band of the neck
- Limited arc of rotation
- Fasciocutaneous pedicle limits the reach of the flap
- Unsightly donor defect especially if skin graft is required (Figure 6)
Anesthesia and preparation
- General anesthesia
- Paralysis acceptable as there are no major nerves are in the surgical field
- Supine position with padding/bag under the shoulders
- Prepare and drape the anterior chest wall, shoulder and neck
- Prepare and drape the thigh for a possible skin graft
Flap design (Figures 4, 7)
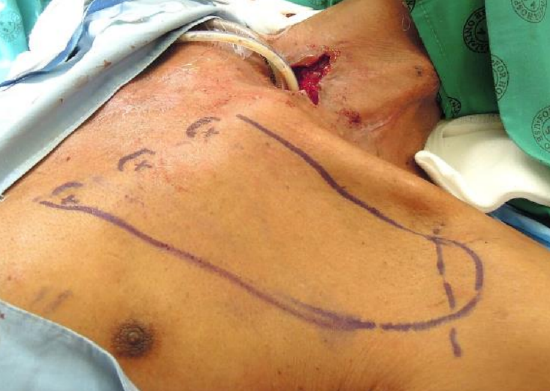
Figure 7: Closing a pharyngocutaneous defect after laryngectomy (pharyngeal mucosa has been closed); positions of the 1st three perforators and the deltopectoral groove have been marked on the skin
- Outline the flap over the anterior chest wall and shoulder (Figure 7)
- The DP flap was originally based on the 1st - 3rd perforators but is now is generally based only on the 2nd and 3rd perforators
- Mark the positions of the intercostal perforators on the skin in the intercostal spaces, 1 cm lateral to the sternum
- A Doppler probe may be used to define their positions more accurately (Figure 7)
- Measure the required length of the flap from the top of the defect in the neck to the point of rotation which is based at the most inferior perforator to be used
- Note that the point of rotation of the DP flap is located closer to the upper side of the base of the flap due to the more lax skin along the inferior border of the flap and along the anterior axillary fold
Limits of DP flap
- Above: clavicle (unless cervicodeltopectoral rotation flap is used)
- Below: 5th intercostal space
- Lateral: deltopectoral groove (extension over the deltoid muscle may require delaying the flap)
Flap elevation (Figure 8)
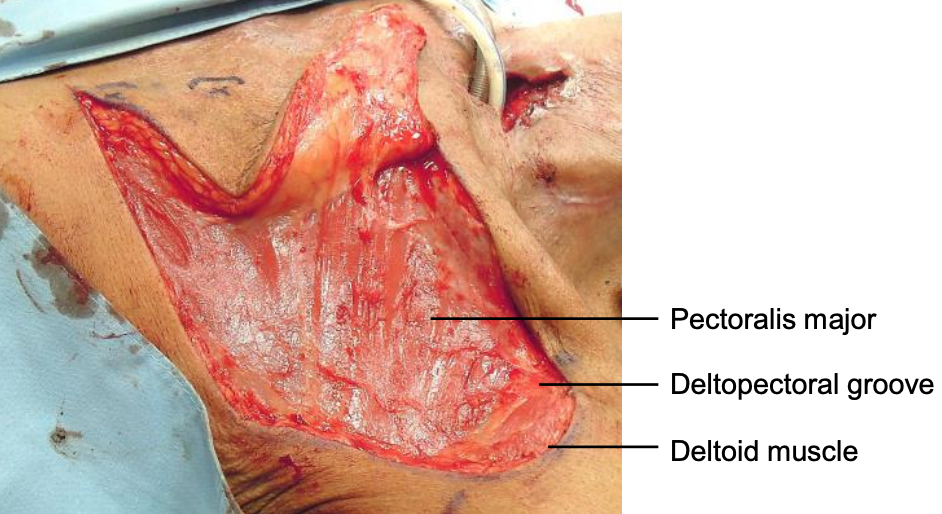
Figure 8: Flap has been elevated in a subfascial plane to expose pectoralis major and deltoid muscles
- Cut through skin and subcutaneous fat and breast tissue onto the (delto)pectoral fascia
- Cut through the (delto)pectoral fascia onto the muscle
- Suture the skin edges to the fascia
- Leave these sutures long at the distal end of flap and hold onto them when elevating the flap
- Use a scalpel (not diathermy) to elevate the pectoral fascia off the muscle in order to protect the suprafascial vascular network
- Elevate the flap from lateral to medial just deep to the (delto)pectoral fascia; this exposes the muscle fibers of the two muscles (Figure 8)
- Branches of the thoracoacromial artery which perforate the pectoralis major muscle laterally are severed during elevation
- Stop elevating the flap about 2 cm lateral to the sternum so as not to injure the perforators
Flap Inset
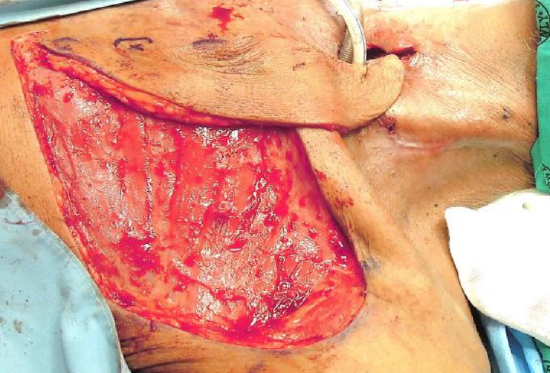
Figure 9a: Flap is positioned in its final position to cover the pharyngocutaneous defect; note the back-cut below the 3rd perforator to increase the length of the flap
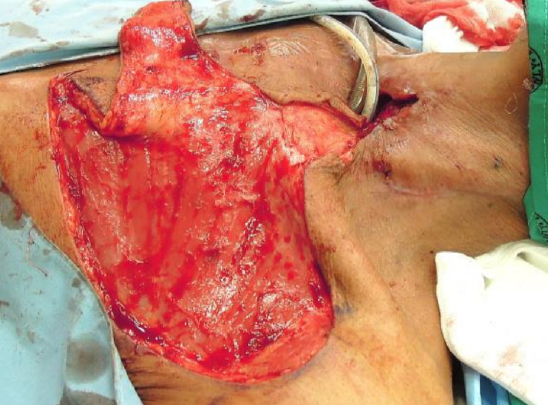
Figure 9b: The skin bridge between the defect and the flap has been divided and elevated to accommodate the flap’s pedicle
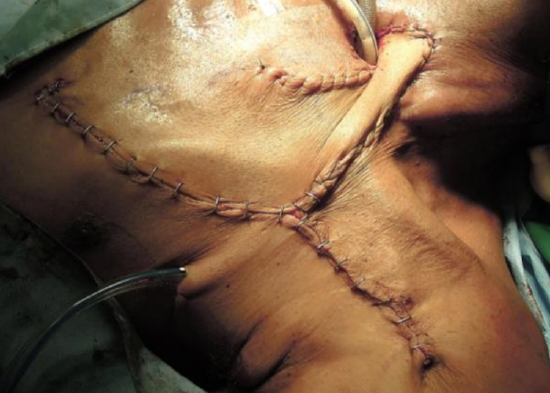
Figure 9c: Flap has been inset and the donor site has been closed primarily
- The entire flap may be used to cover adjacent defects (Figures 9a, b, c)
- The flap may also be tubed and passed over an intact bridge of skin, or fashioned as an island flap by separating the skin off the pedicle in a subcutaneous plane
Closure of donor defect
- Small donor sites can be closed primarily by undermining the surrounding skin and inserting a suction or pencil drain (Figure 9c)
- Larger donor sites are covered with a split thickness skin graft (Figure 6)
Maximizing flap survival
- Handle the flap atraumatically
- Use a scalpel (not diathermy) to raise the flap
- Use bipolar cautery for hemostasis
- Avoid injury to the perforating arteries
- Avoid tension on the flap during its rotation and inset
- If possible, avoid extending the flap beyond the deltopectoral groove
- Avoid tracheostomy ties around the neck as they may compress the vascular pedicle
Gaining additional length
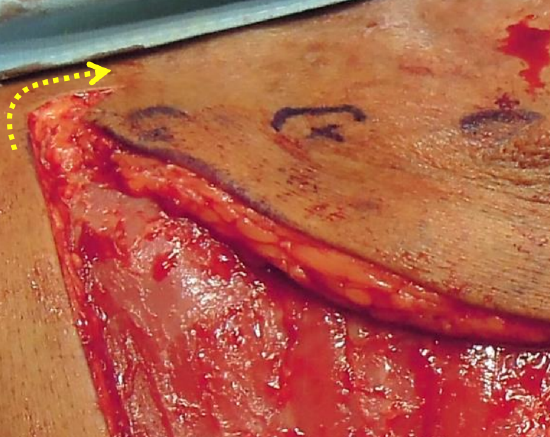
Figure 10: Additional reach obtained by cutting back below and medial to the most inferior IMA perforator
- Cut back over the sternum below and medial to the most inferior perforator (Figures 4, 10)
- Extend the flap over the deltoid muscle or even include an inferior 90º extension
- Risk of skin necrosis increases the further laterally the flap is extended over the shoulder
- A delay technique may be employed to improve survival of the distal flap (see below)
- The tip of the flap can be supercharged (see below)
- Use tissue expanders (see below)
Delay technique (Figures 11, 12)
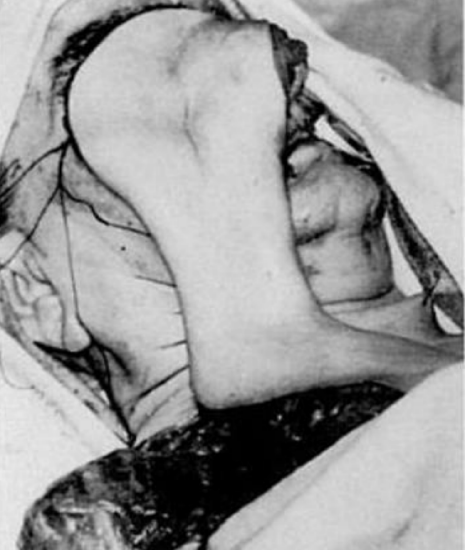
Figure 11: Additional reach obtained by delaying a DP flap (Bakmajian) 5
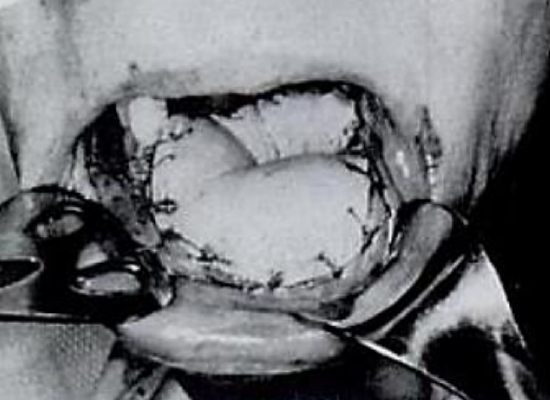
Figure 12: Oral cavity reconstruction is possible following delay of a DP flap (Bakmajian) 5
Delaying a DP flap may improve flap survival, especially with extension of the flap beyond the deltopectoral groove. Delay is thought to:
- Condition the tissue to ischemia
- Increase vascularity
- Sympathectomy
- Reactive hyperaemia
- Stimulates inflammatory cascade with release of vasodilators
- Vascular reorganization with opening of choke vessels, increasing vessels in the subdermal plexus and a more longitudinal reorganization of vessels
There are various methods to delay DP flaps
- Elevate the distal ½ of the flap ensuring division of the deltoid perforators as well as the contribution from the thoracoacromial artery
- Return and suture the flap back to its original position
- One may elect to apply a split thickness skin graft at the time of primary elevation to line the deep aspect of the flap to subsequently serve as the inner lining of a pharyngeal reconstruction
- Elevate the flap again 10-14 days later
Supercharging
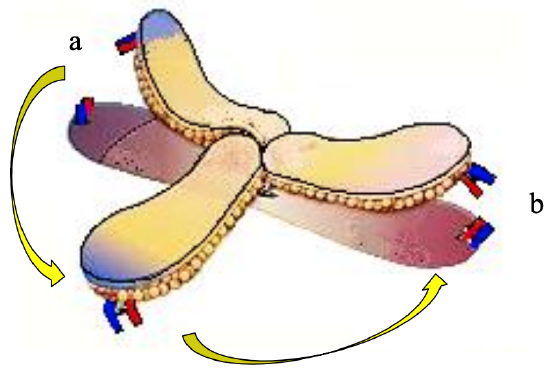
Figure 13: Supercharging tip of DP flap: Vessels at donor site (a) divided and anastomosed to vessels at recipient site (b)
Branches of the thoracoacromial vessels are identified at the superior part of the flap and anastomosed to vessels in the recipient site by microvascular technique (Figure 13).
Tissue expanders
Using tissue expanders placed deep to the pectoral fascia increases the area of skin available for reconstruction of both the recipient area and the donor defect. It permits reconstruction of defects as high up as the cheek and has the added benefit of delaying the flap.
Split skin graft for inner lining
When a DP flap is employed to repair an oro- or pharyngocutaneous fistula, then a split skin graft can be applied to the deep aspect of the flap either at the time of delaying a flap or at the time of definitive repair. This can also be achieved by applying a split skin graft over a tissue expander which is inserted endoscopically, hence allowing the flap to be elevated and inset into a pharyngeal defect with the skin graft already having taken.
Crossing an intact skin bridge
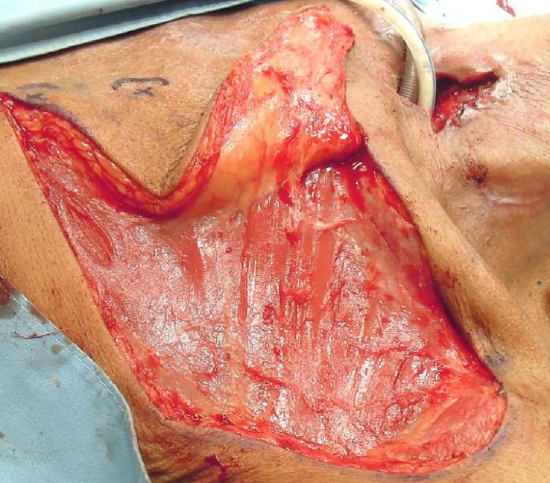
Figure 14: Skin bridge between donor and recipient sites
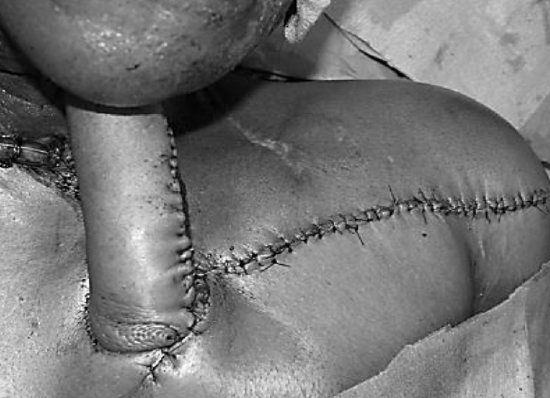
Figure 15: Staged repair of orocutaneous fistula; the pedicle is subsequently divided, and the proximal end is returned to the donor site 6
When skin bridge is interposed between the donor and recipient sites (Figure 14), the surgeon can opt to divide the skin bridge (Figure 9b), discard the interposing skin, de-epithelialize and tunnel the flap below the skin bridge, or tube the flap and subsequently divide the pedicle and return the proximal portion of the flap to the chest wall donor defect after 3-6 weeks (Figure 15).
Cervicodeltopectoral rotation flap
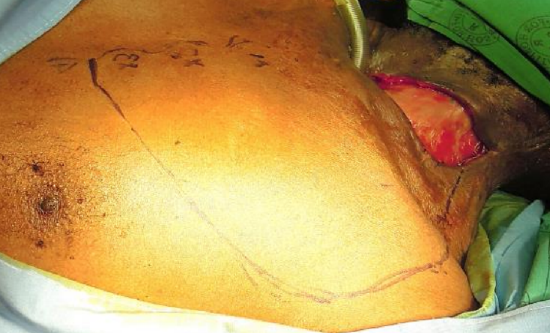
Figure 16a: Cervical skin defect; flap has been based on the 1st 3 perforators
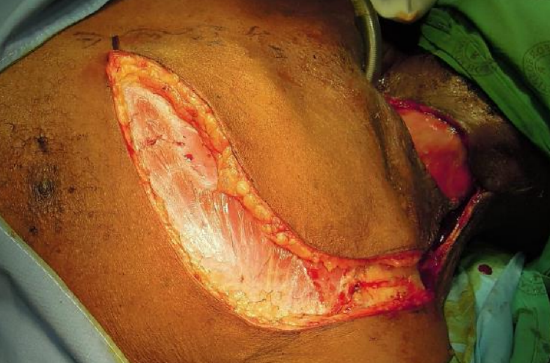
Figure 16b: Incise skin, fat, and pectoralis fascia
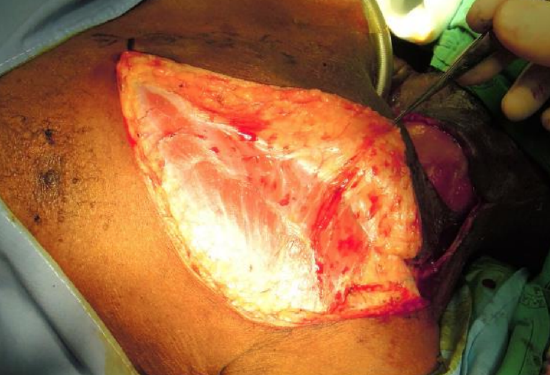
Figure 16c: Elevate DP flap deep to pectoralis fascia
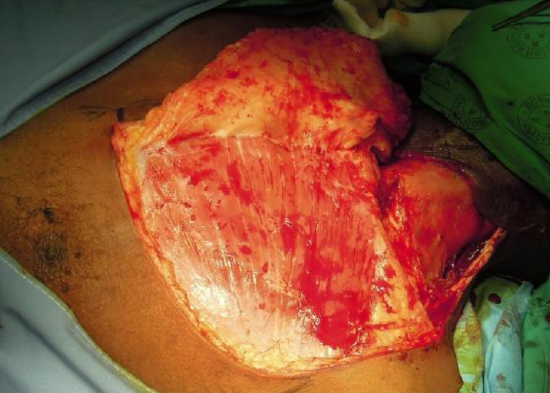
Figure 16d: Fully elevated cervicodeltopectoral flap
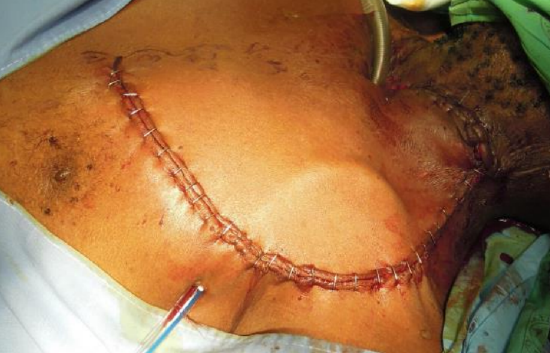
Figure 16e: Cervicodeltopectoral flap rotated into cervical defect and donor site closed primarily
When a cervical defect abuts or is in close proximity to the superior margin of a DP flap, then the defect can be covered with a cervicodeltopectoral rotation flap based on the IMA perforators (Figures 16a-e).
Combined DP & Pectoralis major flaps
When raising a pectoralis major flap one can elect to retain the option of using an ipsilateral DP flap (either at the same operation or later). This requires that when raising the pectoralis major flap, the dissection plane is kept deep to its fascial covering to preserve the blood supply to the DP flap. Either the DP flap is first raised as described above and, following pectoralis major flap transposition, is resutured in its original position; or the DP flap is raised through parallel superior and inferior incisions, maintaining its lateral attachment by branches of the acromiothoracic vessels at the deltopectoral groove.
Deltopectoral island flap 7
A skin island is designed that is placed laterally within the confines of a DP flap. Skin flaps are carefully elevated in a subdermal plane (protecting the underlying deeper suprafascial vascular supply to the skin island) via superior and inferior horizontal incisions. Then elevate the flap in the usual manner and transpose it underneath the intervening skin bridge to reach the defect and thus (usually) avoid a donor site skin graft.
Internal mammary artery perforator island flaps (IMAP flaps)
A DP flap can be converted into an island flap based on one or two perforating branches of the internal mammary artery. It may then be used as a pedicled or a free microvascular tissue transfer flap. This increases its versatility, provides a variety of axes of rotation and additional length3. IAMP flaps are discussed in a separate chapter.
Summary
- The most important part of the DP flap for reconstruction is invariably its distal tip; it is this area that may become ischemic and undergo necrosis
- The DP flap was the workhorse of head and neck reconstruction during the 1970’s and 80’s but has since been replaced by microvascular free flaps
- Although the DP flap may be 1st choice in some situations of external defects in the neck, tubed DP flap to reconstruct internal pharyngoesophageal defects has become obsolete in modern head and neck surgery
References
- Bakamjian,VY. A two-stage method for pharyngoesophageal reconstruction with a primary pectoral skin flap. Plast Reconstr Surg. 1965;36:173
- Gedge DR, Holton LH, Silverman RP, Singh NK, Nahabedian MY. Internal mammary perforators: a cadaver study. J Recon Microsurg. 2005;21(4):239-42
- Schellekens PPA. Proefschrift: Internal Mammary Artery Perforator flap: 2012
- Taylor GI, Palmer JH. The vascular territories (angiosomes) of the body: experimental study and clinical applications. Br J Plast Surg. 1987; 40(2); 113-41
- Lore JM. General purpose flaps. In: Lore JM, ed. An Atlas of Head and Neck Surgery. 3rd ed. Philadelphia, Pa: WB Saunders Co. 1988:344-57
- Balakrishnan C. Closure of orocuta nous fistula using a pedicled expanded deltopectoral flap Can J Plast Surg. 2008; 16(3): 178–80
- Portnoy, WM, Arena S. Deltopectoral island flap. Otolaryngol Head Neck Surg 1994;111:63-9
Other flaps described in The Open Access Atlas of Otolaryngology Head & Neck Operative Surgery
- Pectoralis major flap
- Buccinator myomucosal flap
- Buccal fat pad flap
- Nasolabial flap
- Temporalis muscle flap
- Paramedian forehead flap
- Upper and lower trapezius flaps
- Cervicofacial flaps
- Submental artery island flap
- Supraclavicular flap
- Latissimus dorsi flap
- Local flaps for facial reconstruction
- Radial free forearm flap
- Free fibula flap
- Rectus abdominis flap
- Anterolateral free thigh flap
- Thoracodorsal artery scapular tip (TDAST) flap
- Principles and technique of microvascular anastomosis for free tissue transfer flaps in head and neck reconstructive surgery
Author
JE (Ottie) Van Zyl MBChB, FCS
Plastic & Reconstructive Surgeon
Groote Schuur Hospital
Cape Town, South Africa
ottie@mweb.co.za
Editor & Author
Johan Fagan MBChB, FCS(ORL), MMed
Professor and Chairman
Division of Otolaryngology
University of Cape Town
Cape Town, South Africa
johannes.fagan@uct.ac.za


