5.8: Vascularized Free Fibula Flap (FFF) in Head and Neck Reconstruction
- Page ID
- 52896
OPEN ACCESS ATLAS OF OTOLARYNGOLOGY, HEAD & NECK OPERATIVE SURGERY
VASCULARISED FREE FIBULAR FLAPS (FFF) IN HEAD AND NECK RECONSTRUCTION
Ottie van Zyl, Johan Fagan
The vascularized free fibular flap (FFF) is the most commonly used flap in head and neck for bony reconstruction. It is a vascularized free composite flap containing bone and muscle, with or without skin and provides reliable single-stage reconstruction with excellent functional and aesthetic results. Other vascularized bone flaps used in the head and neck include iliac crest, scapula and radial forearm flaps.
Microvascular free tissue transfer allows one to transfer well-vascularized, bone-containing flaps for oromandibular and midface reconstruction. Compared to bone grafts or metal plates, well-vascularized bone is associated with a major reduction of surgical complications and has had an enormous impact on the success of such reconstructions. Vascularized flaps withstand irradiation much better than bone grafts or plates, and when reconstructing within a previous irradiated field, has the benefit of importing tissue with a new blood supply.
FFFs are commonly used for reconstruction of oromandibular and midface/maxillary defects; and of upper and lower limb long bones; the pelvis; and joints.
Benefits of FFF
- Large vessels
- Long vascular pedicle
- Well vascularized bone
- Long length of donor bone (>25 cm)
- Adequate bone stock
- Bony strength permits good screw fixation and solid reconstruction
- Bony reconstruction can be shaped with multiple segmental osteotomies
- Stable bicortical osseointegrated dental implant fixation is possible
- Thin, pliable overlying skin (usually)
- Skin island adequate for most head and neck reconstructions
- Very little soft tissue bulk (usually)
- Simultaneous cancer resection and harvesting of flap possible due to distant location of the donor site from head and neck resection
Caveats with FFF
- Donor site morbidity: Delayed wound healing and skin graft loss especially following peroneal tendon exposure; nerve injury; ankle instability; pseudocompartment syndrome, and muscle necrosis
- Preoperative vascular problems: Peripheral vascular disease, venous insufficiency, previous deep vein thrombosis, congenital absence of lower leg vessels (rarely)
- Poor skin quality: Obesity, stasis and ischemia
- Previous lower limb trauma: Fractures, vascular injury
Anatomy
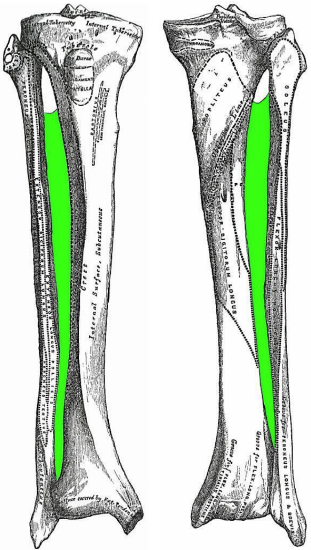
Figure 1: Anterior and posterior views of tibia, fibula and interosseus membrane (green) of right leg
The lower leg has two long bones i.e. tibia and fibula (Figure 1). The tibia is the principal weight-bearing bone and is much stronger than the slender fibula. The fibula is triangular in cross-section, but has 4 surfaces i.e. medial, lateral, and posterior with a narrow anterior surface. It serves as a bony pillar for the origins of eight muscles and insertion of one, as well as the attachment for ligaments and fascial septa. It contributes to stabilizing the knee by the insertions of the biceps femoris tendon and lateral collateral ligament onto the fibular head, and of the ankle where the lateral malleolus forms the lateral part of the mortise or socket, to hold the talus in place.
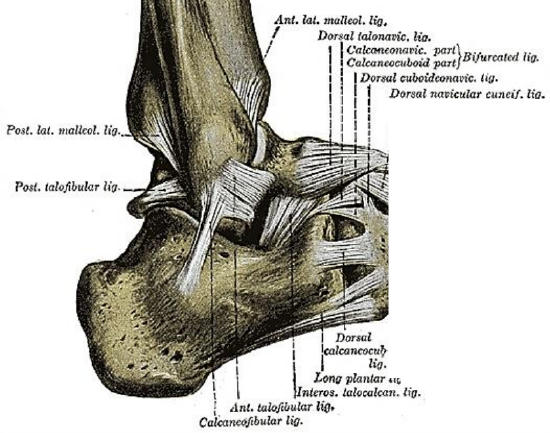
Figure 2: Ligaments attached to the distal fibula of right ankle
The fibula also stabilizes the ankle via the anterior and posterior tibiofibular ligaments, the anterior and posterior talofibular ligaments, and the calcaneofibular ligament (Figure 2). The stability and position of the lateral malleolus and distal fibula are further supported by the distal part of the very strong interosseous membrane which extends between the fibula and tibia (Figure 1). The interosseous membrane holds the two bones together and gives origin to some of the muscles of the lower leg. It has a defect proximally through which the anterior tibial artery gains access to the anterior compartment of the leg after the bifurcation of the popliteal artery (Figure 1).
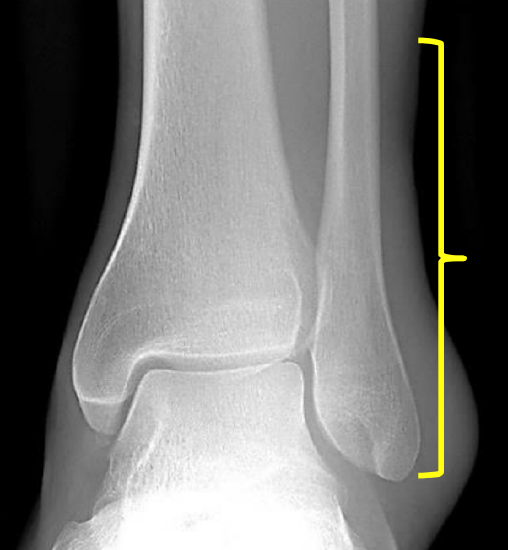
Figure 3: Preserve distal 5-6 cm of fibula to preserve stability of ankle
When harvesting a FFF it is of paramount importance to retain at least 5-6 cm of distal fibula to maintain the integrity of the distal interosseous membrane and of the ligaments mentioned in order to preserve the stability of the ankle (Figure 3).
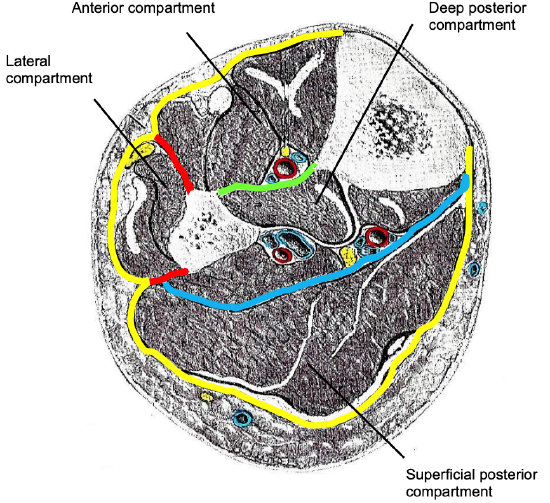
Figure 4: Fascial layers, compartments and cross-sectional anatomy of left leg: Investing layer (yellow); interosseus membrane (green); anterior and posterior crural intermuscular septa (red); transverse crural intermuscular septum (blue)
The investing deep fascia circumferentially encloses the muscles of the lower leg; it is attached to the anterior and medial borders of the tibia (Figure 4). The lower leg is separated into four fascial compartments i.e. anterior, lateral, and deep and superficial posterior (Figure 4). The interosseous membrane separates the anterior from the posterior compartment. The lateral compartment overlies the lateral fibula and is separated from the other two compartments by anterior and posterior crural intermuscular septa attached to the fibula. A transverse crural intermuscular septum divides the posterior compartment into superficial and deep compartments.
Contents of compartments (Figures 4, 5):
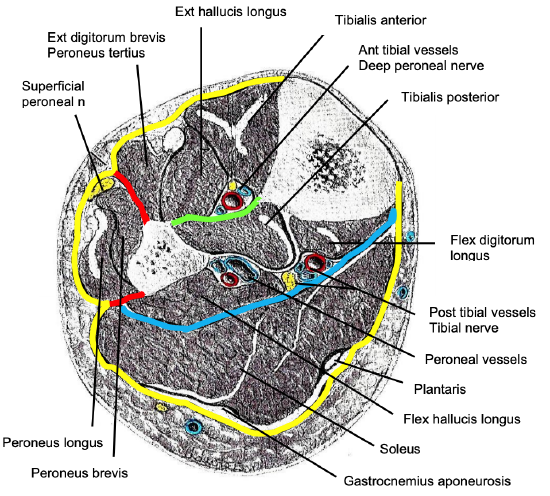
Figure 5: Contents of fascial compartments
Anterior compartment
- Tibialis anterior, extensor hallucis longus, extensor digitorum brevis, peroneus tertius
- Tibialis anterior artery & vein
- Deep peroneal nerve
Lateral compartment
- Peroneus longus, peroneus brevis
- Superficial peroneal nerve
- Blood supply to peroneal muscles are branches of the peroneal artery which lie within the posterior compartment
Superficial posterior compartment
- Gastrocnemius (medial & lateral), soleus, plantaris
Deep posterior compartment
- Tibialis posterior, flexor hallucis longus, flexor digitorum longus
- Tibial nerve
- Tibial & peroneal arteries and veins
Muscles attached to fibula (Figs 1, 4, 5, 6)
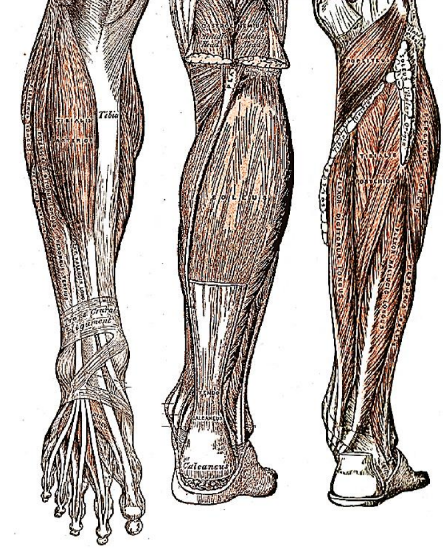
Figure 6: Anterior and posterior views of muscles of leg
- Soleus
- Peroneus longus
- Peroneus brevis
- Peroneus tertius
- Tibialis posterior
- Flexor hallucis longus
- Extensor hallucis longus
- Extensor digitorum longus
Muscles encountered when raising a FFF
- Peroneus longus
- Peroneus brevis
- Extensor digitorum longus
- Tibialis posterior
- Soleus
- Flexor hallucis longus
(The muscles subsequently reattach to scar tissue and the divided interosseous membrane)
Vasculature
The FFF is based on the peroneal artery and its venae comitantes.
Perforators supplying the skin run around the fibula via the posterior crural septum (Figure 4). The fibula has both endosteal and periosteal blood supply. The endosteal supply is via a branch of the peroneal artery which usually enters the bone at the junction of its proximal and middle thirds; this supply plays a role in long bone reconstruction where no osteotomies are performed. Periosteal blood supply is critical to vascularity of the bone when the fibula is shaped by performing osteotomies. Therefore, as much periosteum as possible, as well as a cuff of muscle, is preserved around the fibula.
The periosteal supply to the distal two thirds of the bone is via the peroneal vessels.
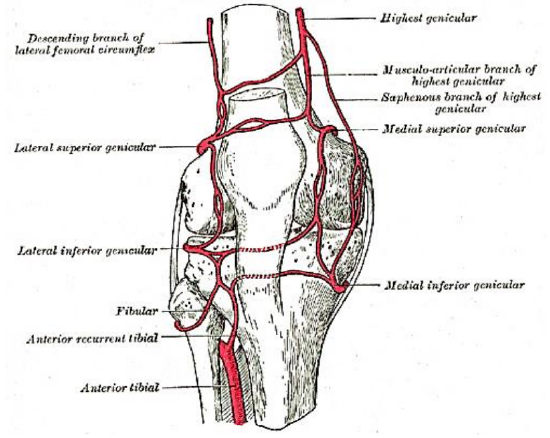
Figure 7: Blood supply to proximal fibula
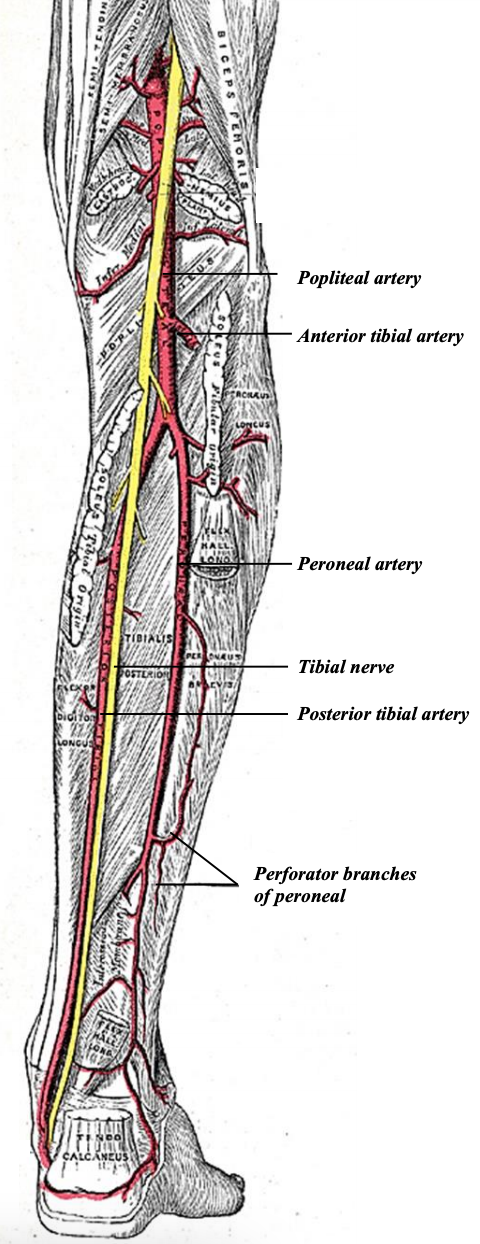
Figure 8: Course of peroneal artery
The head of the fibula and a variable length of the proximal third of the fibula are supplied by the anterior tibial vessels via the anterior recurrent tibial and/or lateral inferior genicular recurrent branches (Figures 7, 8).
The tibioperoneal trunk in turn bifurcates into the posterior tibial and peroneal arteries. The posterior tibial artery courses deeply along the lateral aspect of the leg and can be palpated as the posterior tibial pulse (Figures 5, 8).
The peroneal artery originates as a large branch of the posterior tibial artery, just below the popliteal fossa, 2-3 cm below the tendinous arch of the soleus (Figure 8). The artery is accompanied by paired venae comitantes.
The peroneal vessels are in close proximity to the fibula as they course along the entire length of the bone (Figures 5, 8). The artery descends along the medial side of the fibula where it lies posterior to the tibialis posterior muscle and anterior to the flexor hallucis longus muscle (Figure 5). It is closely applied to the fibula, but veers away, and more medially, towards its origin from the posterior tibial artery at the upper third of the fibula. Variations of lower leg vascular supply are uncommon but are important to consider. With a dominant peroneal artery, the pedal circulation is more dependent on this artery; sacrificing the artery may render the foot susceptible to ischemia.
Nerves (Figures 5, 8, 9, 10)
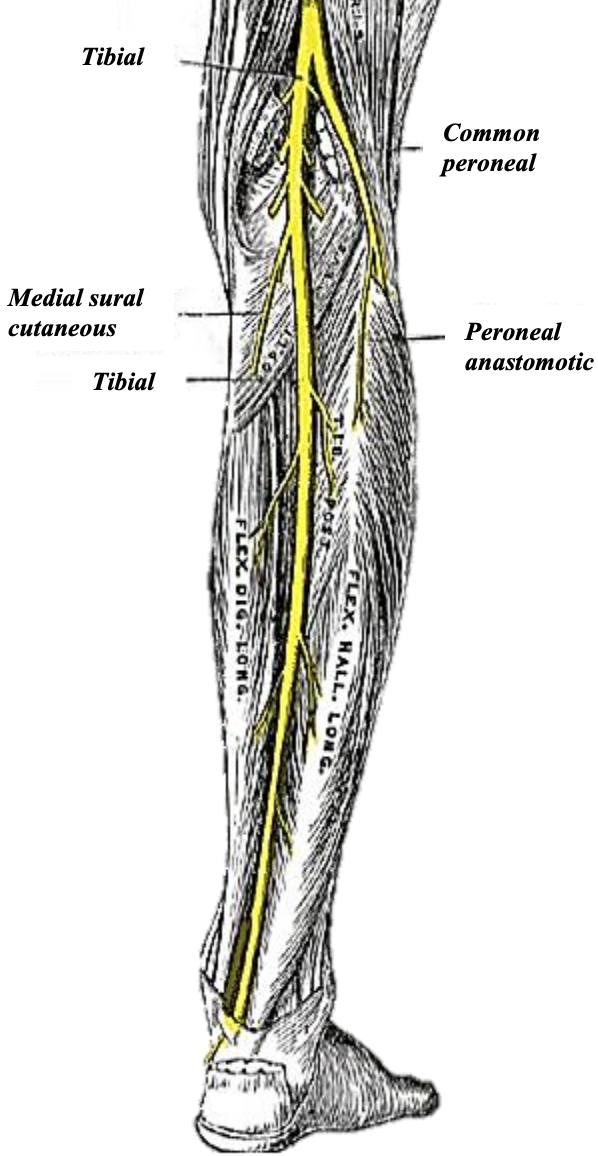
Figure 9a: Nerves of lower leg
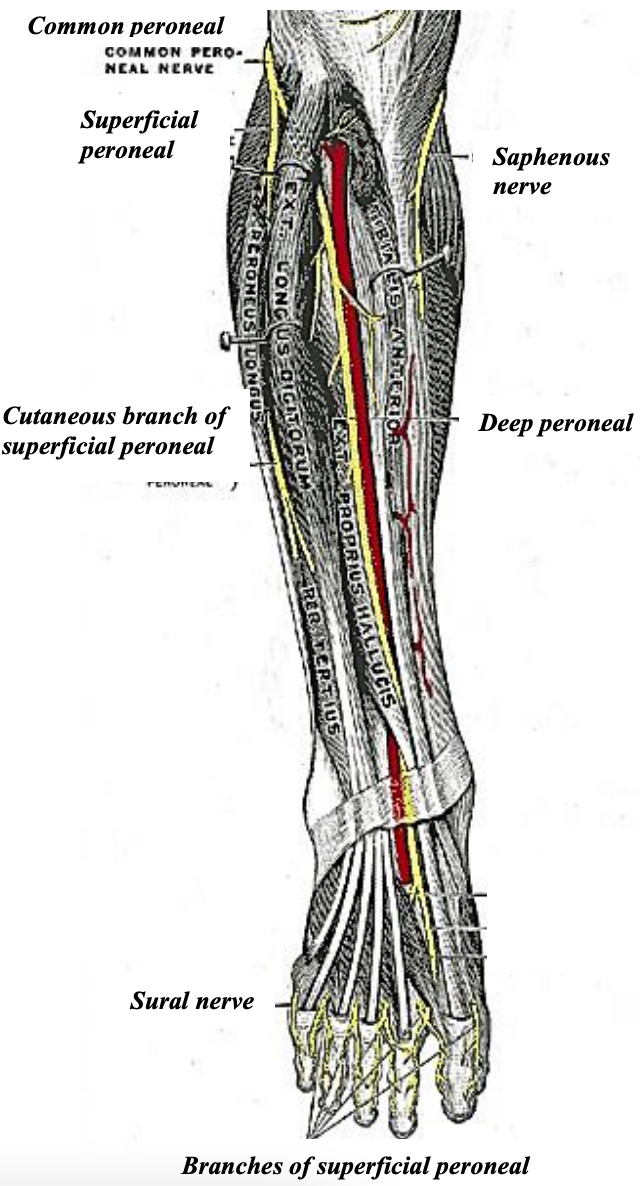
Figure 9b: Nerves of lower leg
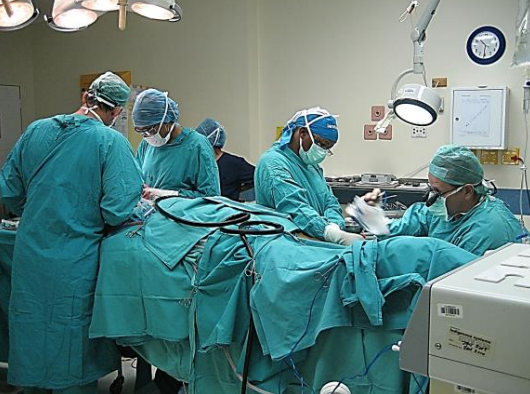
Figure 10: Two-team approach with anesthetic equipment at foot of bed
The sural, common peroneal and superficial peroneal nerves are vulnerable during elevation of a FFF. The sciatic nerve divides into the tibial and common peroneal nerves in the superior popliteal fossa. The tibial nerve gives off the medial sural cutaneous nerve which descends between the two heads of the gastrocnemius, pierces the deep fascia at about the middle of the back of the leg, to innervate the medial calf (Figure 9). It subsequently joins the communicating branch of the common peroneal to form the sural nerve. The sural nerve innervates the lateral leg. It is vulnerable at the posterior border of FFF skin elevation. It lies in close proximity to the small saphenous vein and passes distally near the lateral margin of the calcaneus tendon. It perforates the deep fascia in the midcalf to continue subcutaneously, passing midway between the lateral malleolus and the Achilles tendon to supply sensation to the lateral foot and little toe.
The common peroneal nerve, after giving off the sural communicating branch to the sural nerve and the lateral sural cutaneous nerve which supplies sensation to the lateral calf (may be used as sensory input to the FFF skin island), exits the popliteal fossa by passing superficial to the lateral head of the gastrocnemius muscle, lying just posterior to the biceps femoris tendon (Figures 5, 9). It winds from posteriorly around the lateral surface of the neck of the fibula and then beneath peroneus longus where it divides into the deep and superficial peroneal nerves (Figures 5, 9b). The common peroneal nerve is vulnerable when the proximal fibula and its head are harvested.
The deep peroneal nerve pierces the anterior crural intermuscular septum and travels deep to the extensor digitorum longus to join the anterior tibial artery in the anterior compartment, where it lies between tibialis anterior and extensor hallucis longus (Figures 5, 9b). It innervates these muscles and provides sensory innervation to the web between the 1st two toes and an area proximal to this on the dorsum of the foot.
The superficial peroneal nerve travels beneath the peroneus longus muscle in the upper third of the lateral leg and descends beneath the deep fascia in the anterior part of the lateral compartment or within the anterior crural intermuscular septum (Figures 5, 9b). It pierces the deep fascia to lie superficially in the distal leg. The nerve is vulnerable during elevation of the anterior aspect of the skin flap. It supplies sensation to the anterior surface of the distal leg and to most of the dorsum of the foot and toes.
Preoperative Evaluation
Preoperative evaluation is directed at determining the vascular status of the FFF. Take note of a history of intermittent claudication, deep vein thrombosis, lower limb trauma and the presence of varicose veins. Previous lower limb trauma or fractures may warrant X-rays. Assess the circulation of the foot and quality of the skin. Only if there is concern about possible arterial or venous insufficiency are further investigations indicated. Duplex color flow Doppler will usually suffice. Only very rarely is CT angiography or invasive angiography indicated. Should there be concern about vascularity an alternative donor site should be considered.
Informed consent
This should include a discussion about incisions, donor site morbidity including the possibility of a skin graft being required, failure rates, and dental rehabilitation.
Surgical setup
Resection of the primary tumor and elevation of the FFF are done simultaneously as a 2-team approach in order to minimize the duration of surgery (Figure 10). To create working space around the head and torso, the anesthetic equipment is placed at the foot of the bed with extensions for intravenous lines and anesthetic tubing. Two electrocautery systems are required. Cautery and other equipment are positioned at the foot of the bed contralateral to the operated leg. A tourniquet is placed around the thigh and a 3-liter infusion bag is placed under the buttock on the side of the FFF to internally rotate the leg as much as possible. The lower leg is shaved if necessary. A warming blanket covers the lower torso and opposite leg; alternatively, it may be placed under the patient. Intermittent calf compression is applied to the opposite leg. The table and/or chair heights are adjusted so that the surgeon and assistant are seated while raising the FFF.
Which donor leg to use?
Factors to consider include:
- Is a skin paddle required?
- Is the skin paddle to be used for internal or external construction? (the pedicle(s) to the skin island passes from the peroneal vessels around the posterior aspect of the fibula and exits the posterior crural septum)
- What side of the neck will be used for the donor vessels?
Donor leg for mandible reconstruction (Table 1)
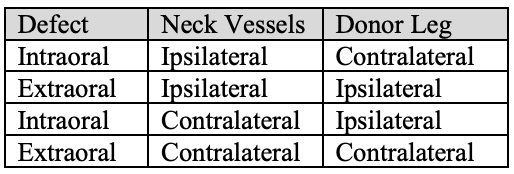
Table 1: Choice of leg with mandible reconstruction
- The lateral surface of the fibula becomes the outer surface of the “neomandible” and is the surface to which the reconstruction plate is applied
- For intraoral soft tissue reconstruction, the skin paddle needs to lie above the “neomandible"; yet it lies inferiorly while the pedicle is still attached to the lower leg. The bone and flap therefore are flipped over in a longitudinal axis when the FFF is transposed to the defect with the lateral surface of the fibula remaining laterally
- For reconstructing extra-oral or cutaneous defects the skin paddle needs to be inferolateral to the “neomandible”
- Combined intra- and extra-oral defects can be reconstructed with a single skin paddle which is de-epithelialised between two skin islands. Depending on the relative sizes of each area to be reconstructed, the flap can be harvested from the ipsilateral or contralateral lower leg
Exceptions to Table 1
- The vascular pedicle usually exits from the lateral side of the “neo-mandible"; however, if the contralateral neck is used for donor vessels it needs to exit on the medial side of the flap
- When the ascending ramus is reconstructed up to the angle it may be preferable to have the pedicle exit inferomedially and taken to the ipsilateral neck to avoid kinking the pedicle as it would have to "double-back" towards the neck if it exits posterosuperiorly
Donor leg for midface/maxillary reconstruction
The ipsilateral leg is generally selected, and the ipsilateral neck is always used (or selected in bilateral maxillary reconstruction) for the vascular anastomosis. The palatal defect is the most important part of the reconstruction; this requires the skin paddle to exit below the bone. Again, the lateral surface of the fibula usually faces anteriorly.
Exsanguinating the leg
- Elevate the leg 600 without exsanguinating the leg
- Select the widest tourniquet cuff possible to occlude blood flow at a lower pressure and thus reduce risk of injury
- Insert soft padded protection between the skin and the cuff to prevent skin abrasions and blisters
- Inflate tourniquet to at least 100 mmHg above the patient’s systolic blood pressure (adults)
- Record the tourniquet inflation time; the surgeon should be informed when tourniquet has been inflated for 60 min
- There is no clear-cut rule as to how long a tourniquet may be safely inflated for
- A maximum tourniquet time of 90 min for the leg has been proposed; thereafter deflating the cuff for 15 minutes to allow reperfusion; then the cuff can be reinflated for another 90 minutes
Skin Markings
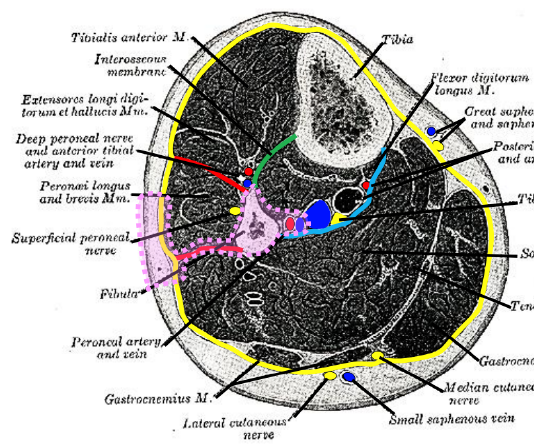
Figure 11: Cross-sectional outline of FFF (pink), based on peroneal artery, its venae comitantes and perforators that enter the skin via the posterior crural septum (red)
The FFF is based on the peroneal artery and its venae comitantes; perforators enter the skin via the posterior crural septum (Figures 4, 5, 8, 11).
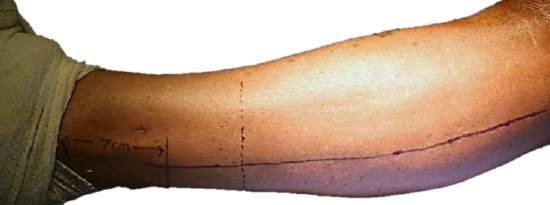
Figure 12: Skin markings
- Mark the head of the fibula and lateral malleolus (Figure 12)
- Draw a line along the posterior border of the fibula along the posterior crural intermuscular septum (Figures 4, 11, 12). This is visible as the indentation between the peroneus (superior to the line) and soleus muscles (posterior to the line)
- Mark the distal osteotomy site (Figure 12) by drawing a transverse line at the level of the planned osteotomy; at least 5 cm (preferably >7 cm if less bone is required) above the projection of the lateral malleolus to maintain ankle stability (Figure 3)
- Beware of the superficial peroneal nerve which is located 3-4 cm anterior to the longitudinal line in the lower half of the leg, and the sural nerve which is 3-4 cm posterior to this line
- Plan the dimensions of the skin flap
- Mark the width of the flap on the leg; it may be as narrow as 1 cm or as broad as 12 cm. When harvesting a narrow skin island, it is critical to determine the position of the posterior crural intermuscular septum as it carries the perforators; the safer option is to harvest a wider flap which is trimmed later
- Mark the position of the flap along the length of the leg; one does not know at this stage where along the length of the leg the skin perforators are
- If a long flap is required, center it over the junction of the distal and middle thirds of the leg
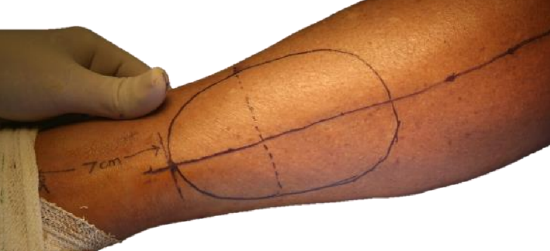
Figure 13: Flap centered over junction of the distal and middle thirds of leg
- With smaller flaps one runs the risk of not positioning it over a perforator; the safer option is first to incise the skin along the anterior margin of the flap in a gentle curve, permitting one to extend the length of the skin island if a perforator is situated outside the initial mark-up (Figure 13)
Raising the flap
- It is the author’s preference to keep the knee extended as the leg is more stable, less assistance is required, and it makes the operation easier; others prefer to raise the FFF with knee flexed
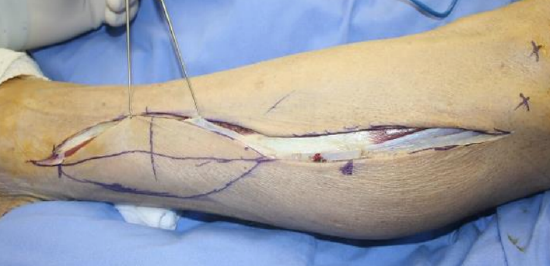
Figure 14: With smaller flaps the safer option may be 1st to incise the skin along the anterior margin of the flap and find the perforator
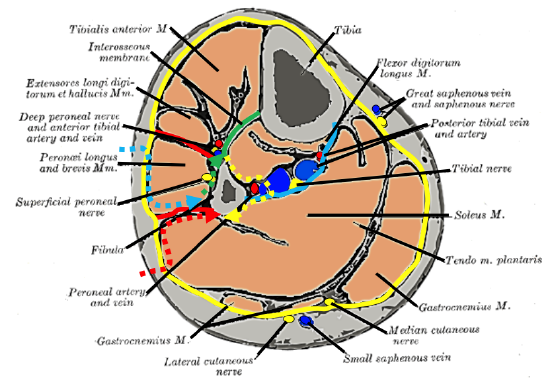
Figure 15: Sequence of surgery: blue arrow → red arrow → green arrow → yellow arrow
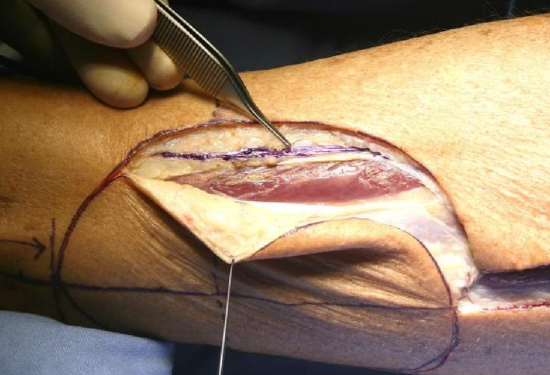
Figure 16: Avoid injury to the superficial peroneal nerve (inked)
- Make and incision along the anterior margin of the skin flap (Figures 14, 15, 16)
- Cut through deep dermis, fat and carefully through investing deep fascia with monopolar electrocautery (Figure 14, 16)
- Avoid the superficial peroneal nerve where it lies just beneath the deep fascia as it courses from lateral to a more anterior position and subcutaneously in the lower third of the lower leg (Figures 9, 11, 15, 16)
- The muscles in the lateral compartment (peronei longus & brevis) are now exposed (Figures 5, 11, 15, 16)
- Retract the anterior edge of the flap laterally with skin hooks placed on the deep fascia; cat’s paw retractors are used to pull the peroneal muscles anteriorly (Figure 16)
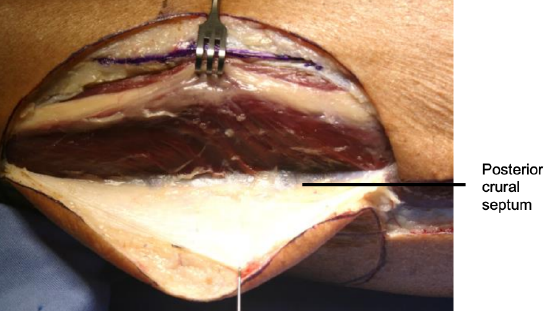
Figure 17: Freeing flap and deep fascia from lateral surface of peronei up to posterior crural intermuscular septum
- Use a #15 scalpel blade to separate the peroneal muscles from the deep fascia up to the posterior crural intermuscular septum (Figures 15, 17)
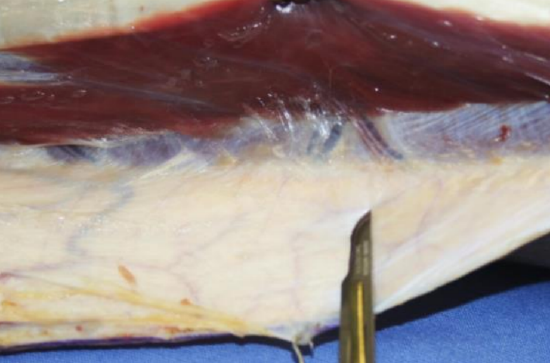
Figure 18: Perforators in view along posterior crural septum
- As sharp dissection is continued posteriorly along the septum, one or two perforators will come into view as they curl around the posterior margin of the fibula (Figure 18). Mark their positions on the skin and deep fascia with ink
- The flap may now be redesigned and centered over the perforator(s), especially if the skin island is to be rotated with the perforator as its pivot point
- If no perforators are seen, look more proximally, and if found, modify the flap to include them. One can also use a proximal musculocutaneous perforator to supply an additional island of skin. If no other source is available and the flap is to remain attached over an extended length of the fibula, the supply via minute capillaries may be adequate and can be assessed following deflation of the tourniquet. If necessary a second flap, e.g. radial free forearm or anterolateral thigh free flap may be harvested and anastomosed to the distal peroneal vessels sequentially or to separate vessels in the neck. A final option is to harvest fibula only with a thicker cuff of muscle which is left to granulate and epithelialize in the mouth without skin cover
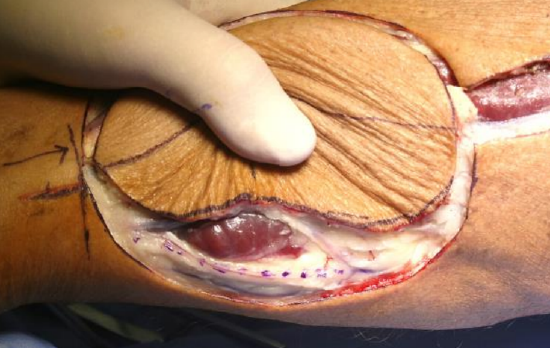
Figure 19: Dissection of posterior margin of flap taking care not to injure sural nerve (inked) and lesser saphenous vein
- Dissect the posterior margin of the flap taking care not to injure the sural nerve and lesser saphenous vein (Figures 15, 19)
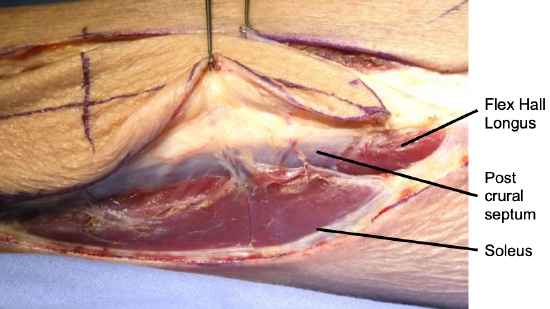
Figure 20: Deep fascia elevated off soleus up to posterior aspect of posterior crural intermuscular septum where perforators again come into view; note muscular branches
- Retract the posterior edge of the flap anterolaterally with skin hooks; use sharp dissection to elevate the deep fascia off the soleus muscle up to the posterior aspect of the posterior crural intermuscular septum where the perforator(s) again come into view (Figures 15, 20)
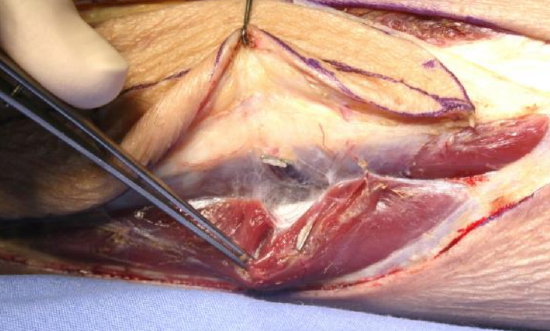
Figure 21: Muscular branches to soleus and flexor hallucis longus have been clipped and divided
- Small muscular branches to soleus and flexor hallucis longus may branch from the perforators (Figure 20); these are ligated with ligaclips and divided (Figure 21)
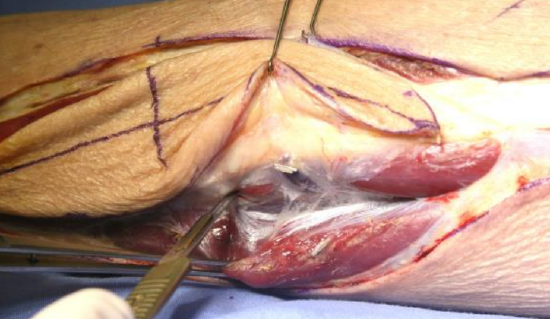
Figure 22: Identify flexor hallucis longus and divide the intermuscular septum
- Continue with sharp dissection deep to soleus; identify flexor hallucis longus and divide the intermuscular septum that covers it along its full length (Figures 15, 22)
- Now return to the anterior surgical dissection (Figure 15: Green arrow)
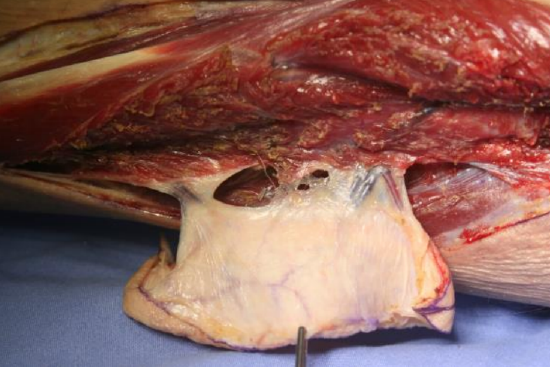
Figure 23: Posterior crural septum has been divided with scissors, leaving enough septum to protect perforators
- If mobility or propeller rotation of the skin island is required, the posterior crural septum may now be divided with scissors, leaving only enough septum to protect the perforator(s) from traction injury (Figure 23)
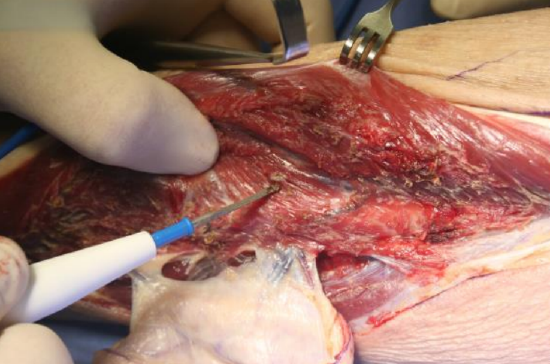
Figure 24: Release peronei, leaving a cuff of muscle on bone
- Release the peroneal muscles in the lateral compartment from the fibula with electrocautery, leaving a 5 mm cuff of muscle on the bone. First retract the muscles with cat’s paws and then with Langenbeck retractors as the muscle is mobilized, while the surgeon uses the non-dominant thumb to elevate and palpate the muscle over the bone. Palpating with the thumb helps to determine the amount of muscle cuff to be left on the bone (Figure 24)
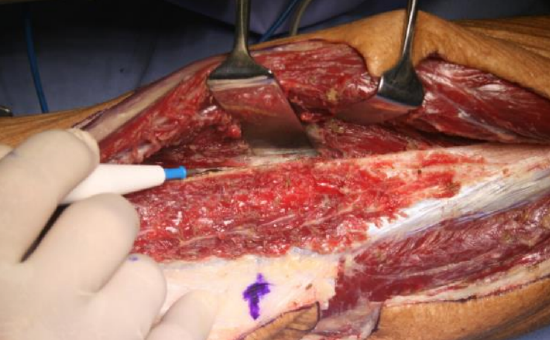
Figure 25: Incising the anterior crural intermuscular septum
- Use cautery to extend the dissection through the anterior crural intermuscular septum (Figures 15, 25)
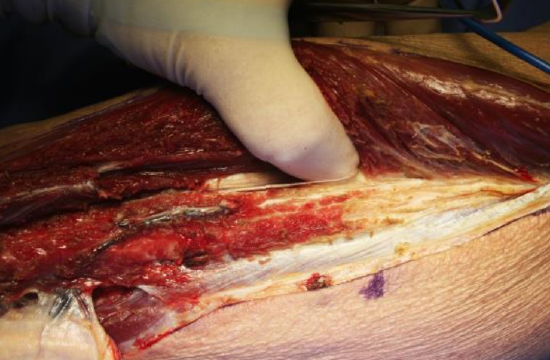
Figure 26: Stripping the proximal part of the peronei and anterior crural intermuscular septum off fibula with a finger
- Extend the dissection proximally; when dissecting proximally beyond the length of bone required for reconstruction, a finger is used to strip the proxymal part of the peronei and anterior crural intermuscular septum off the bone as no muscular cuff is required there (Figure 26)
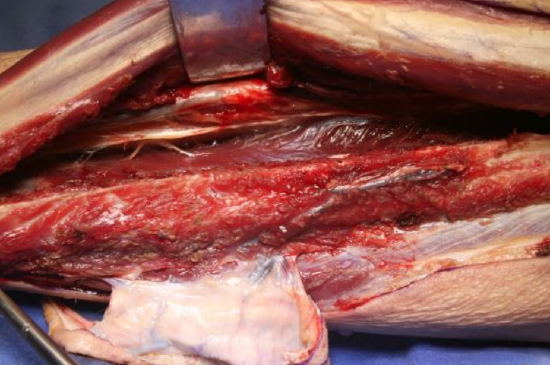
Figure 27: Retracting peronei anteriorly to expose extensor digitorum longus
- Deeper retractors may now be required to firmly retract the peronei anteriorly and the extensor digitorum longus medially (Figure 27)
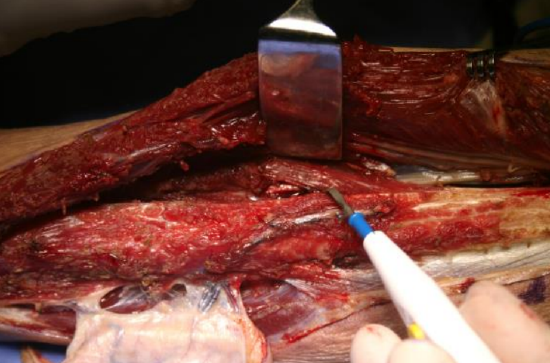
Figure 28: Dividing extensor digitorum longus fibers
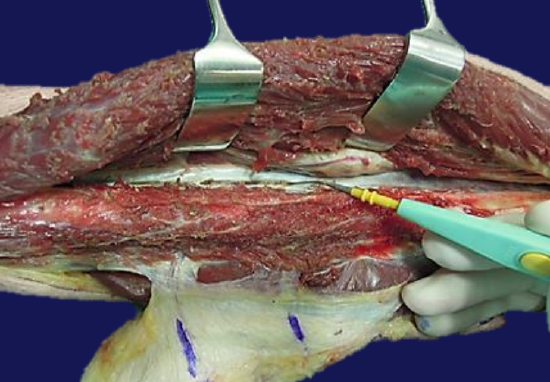
Figure 29: Dividing the interosseous membrane
- Divide the extensor digitorum longus and the deeper extensor hallucis longus fibers with cautery, a few millimeters from the free edge of the fibula (Figures 28)
- Expose and divide the interosseous membrane with electrocautery (Figure 29); this dissection is extended proximally to fully release the strong interosseous membrane; if not done it will prevent the fibula from being retracted laterally to expose the tibialis posterior muscle and peroneal vessels. Tibialis posterior protects the peroneal vessels while doing this
- The proximal and distal osteotomies are now performed. The length of fibula to be removed is the length of bone required for reconstruction plus a generous length of bone proximally as determined by the length of the vascular pedicle required
- Divide the muscle and periosteum transversely with a scalpel over the lateral surface of the fibula onto bone
- Do a subperiosteal dissection around the remainder of the fibula with a delicate dissector such as a Freer’s elevator or Mitchell’s trimmer, taking care not to injure the peroneal vessels or to excessively strip periosteum
- Perform the osteotomies. Use a metal plate to protect the vessels while performing the osteotomies with a high speed electric or air-driven oscillating or reciprocating microsaw
- Insert a bone or tendon hook in the marrow cavity at the distal osteotomy to retract the bone laterally while retracting the peroneal and anterior compartment muscles medially
- This exposes the tibialis posterior muscle; the anterior tibial vessels and deep peroneal nerve are seen lying medially on the cut interosseous membrane, lateral to the tibialis anterior muscle
- This exposure may be difficult; failure to have fully divided the interosseous membrane will not permit the fibula to swing laterally. Additional mobility can be achieved by retracting the proximal segment of the fibula laterally and carefully dividing the flexor hallucis fibers and strong intermuscular fascia between the peroneal vessels and the fibula by staying on the bone
- The segment of fibula is now released laterally
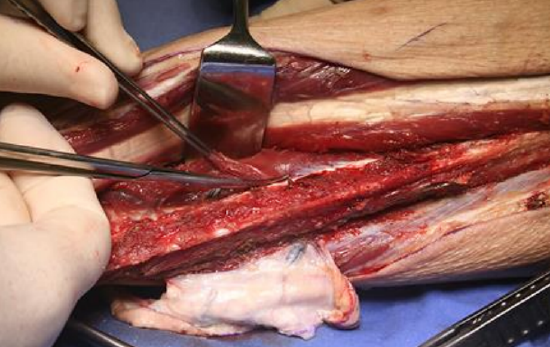
Figure 30: Tibialis posterior
- Retracting the distal end of the bone laterally, divide the tibialis posterior muscle from distally-to-proximally with Metzenbaum scissors (Figure 30)
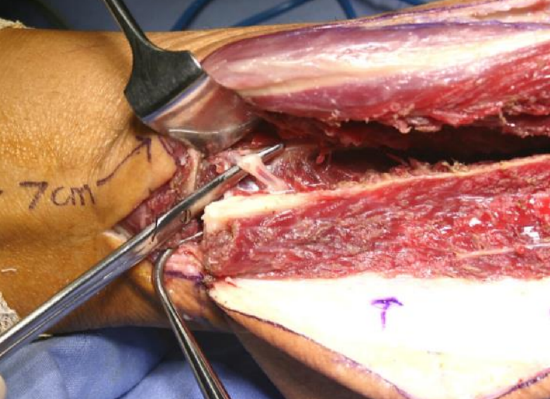
Figure 31: Distal peroneal vessels
- The peroneal vessels are now seen sloping away from the bone medially and are ligated adjacent to the distal osteotomy (Figure 31)
- As the tibialis posterior muscle is divided more proximally, the intermuscular septum under the muscle and covering the peroneal vessels is exposed
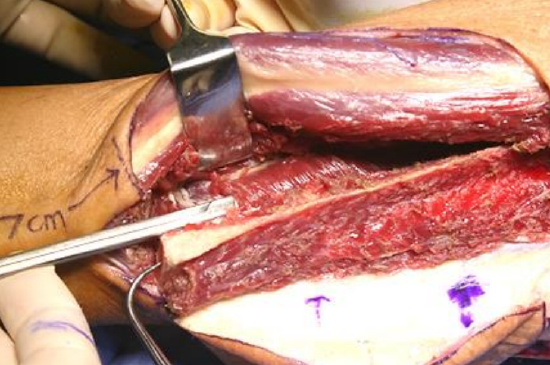
Figure 32: Scissors advanced along the avascular plane between intermuscular septum and peroneal vessels
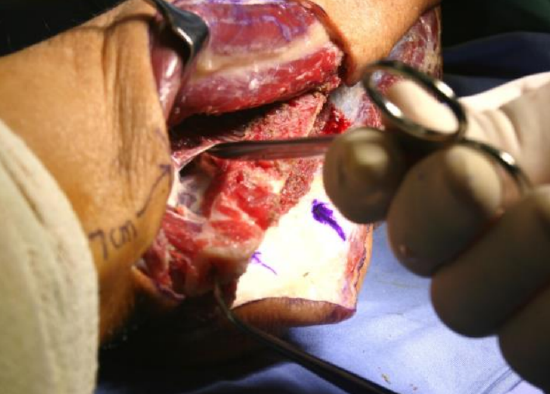
Figure 33: Avascular plane
- A relatively avascular plane exists between the intermuscular septum and the peroneal vessels; establish a dissection plane by advancing the closed tips of the scissors beneath the septum (Figures 32, 33)
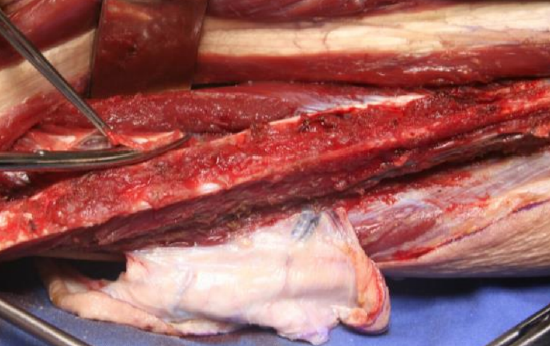
Figure 34: Dividing the interosseous membrane
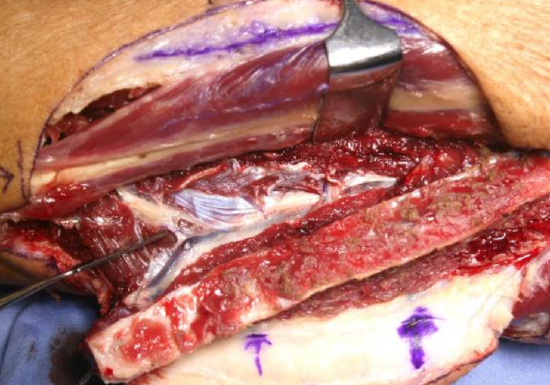
Figure 35: Peroneal vessels exposed after dividing intermuscular septum
- Divide the septum over the medial aspect of the vessels with scissors. Continue this division proximally until the peroneal vessels are exposed along their entire lengths; the large vein(s) lie anterior to the artery (Figures 34, 35)
- A few small side branches emerge, mainly medially, to supply tibialis posterior and are ligated with ligaclips and divided. More proximally, larger branches supplying soleus and other muscles are also ligated and divided
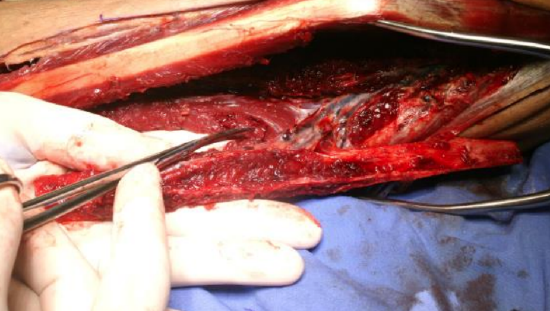
Figure 36: Flap being detached from flexor hallucis longus; index finger protects perforators
- Once all vascular side branches have been divided, detach the flap from its only remaining posteromedial attachment to the flexor hallucis longus. This is achieved by holding the fibula with the left hand and placing the index finger over the perforator to protect it and to remind the surgeon of its position. The muscle is divided from distally to proximally, leaving a generous cuff of muscle especially in the area of the perforating pedicle (Figure 36). Some surgeons prefer to harvest the whole flexor hallucis muscle with the flap because the muscle's blood supply originates mainly from the peroneal vessels; hence the viability of remaining muscle may be compromised
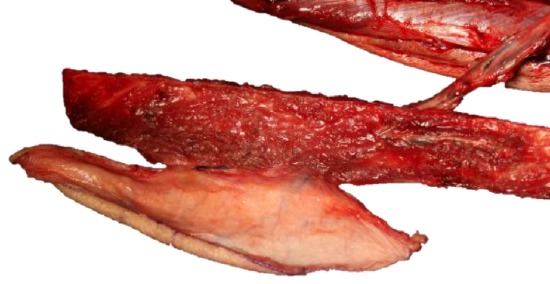
Figure 37: Skin island perforator, fibula, and vascular pedicle ready to be detached
- Once the flap (other than vascular pedicle) is completely free, the peroneal vessels are inspected (Figure 37)
- There are usually two and occasionally three veins. The most suitable vein is selected and the others are ligated as only one vein is needed for the anastomosis
- Carefully separate the vein and artery from each other for a few centimeters to permit the vascular anastomoses to be made some distance from one other
- The tourniquet is usually deflated following flap elevation, but if it has been elevated within an hour, one may continue with 3-dimensional shaping of the fibular osteotomies while the tourniquet is kept inflated
- Control bleeders from the flap and in the surgical bed with bipolar cautery, ligaclips and ties
Plating systems (Figures 38, 39)
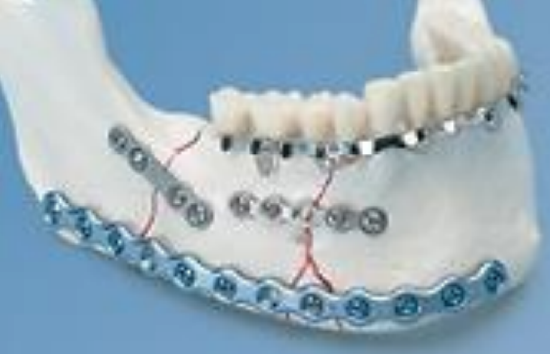
Figure 38: Reconstruction (blue) vs. miniplates (grey)
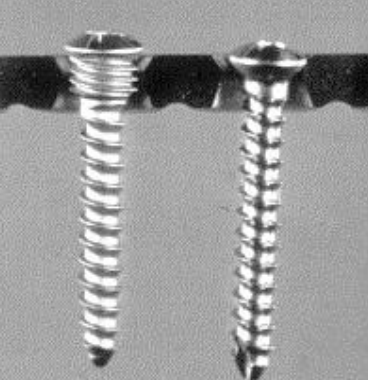
Figure 39: Locking vs. compression screw
Reconstruction plates simplify planning and allow for accurate preplating. They are strong and withstand the muscle forces and torque exerted on the reconstruction, give an extremely stable reconstruction and do not require intermaxillary fixation (IMF) postoperatively. The sizes of the plate and screws, and difficulty to remove them (not normally required) can however prove problematic.
Miniplates (load sharing) are used for maxillary reconstruction; although they may be used for mandibular reconstruction, the plates are more likely to fracture and there is more mobility of the bony segments, necessitating IMF. Miniplates cause less radiation scatter and inserting dental implants is simpler as the plates are smaller and are easily removed. Because fixation is less rigid it permits molding into proper dental occlusion with IMF.
Locking titanium (loadbearing) reconstruction plate systems are preferred so as to avoid compressing the plate against the bone and causing subcortical bone resorption and loosening of screws (Figure 39).
Three-dimensional shaping of fibula
A variety of methods are employed to achieve near-perfect mandibular form and occlusion. Critical elements are to accurately bend/fashion the reconstruction plate and to use the plate(s) to plan the osteotomies. The plates can be shaped in the following ways:
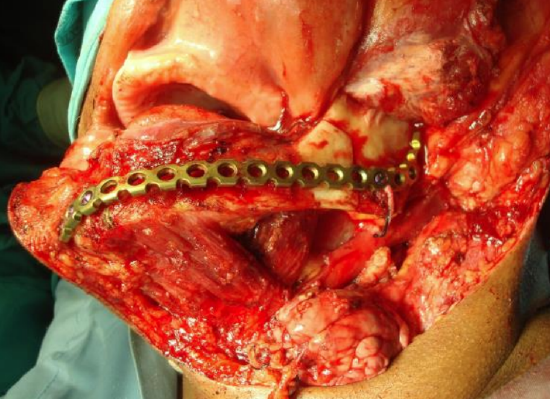
Figure 40: Preplating the mandible
- Preplating by contouring the plate on the exposed mandible prior to resection (authors’ preference) is done if tumor does not distort the outer surface of the mandible; if tumor needs to be elevated off the mandible to permit the plate to sit flush with the bone, the plate is then cleaned and autoclaved before using it. This method assures good form and occlusion (Figure 40)
- Preoperative computer-generated model plate contouring, especially when preplating is not possible due to distortion of the lateral surface of the mandible by tumor (expensive)
- Intermaxillary fixation (IMF) and "blind" plate contouring after tumor resection
- Intraoperative external fixation devices and "blind" plate contouring when IMF not possible
- "Blind" plate contouring after tumor resection
The description of FFF that follows is an anterior mandibular reconstruction with an intraoral flap and anastomosis to the ipsilateral neck vessels.
- The mandibular osteotomy sites are marked on the mandible with the saw
- A (locking) reconstruction plate is fashioned by contouring it on the outer surface of the mandible; the plate is positioned just above the lower border of the mandible
- At least three screw holes are drilled to either side of the planned mandibular osteotomies and the plate is temporarily fixed to the bone with bicortical locking screws
- The positions and angles of the planned osteotomies are precisely drawn on the external surface of the plate at both osteotomy sites. If the resection includes the angle of the mandible, the angle is marked on the plate
- The plate and screws are removed, and the plate is handed to the reconstructive team to use as a template to plan and design the fibular osteotomies
- The mandibulectomy is completed
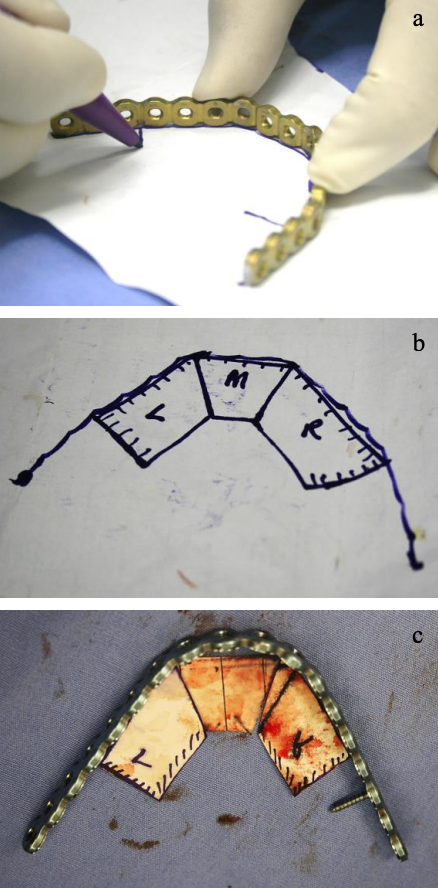
Figures 41a, b, c: Drawing angles of osteotomies
- Draw the profile of the required bony reconstruction on stiff paper (Figures 41a-c)
- There are two profiles to consider i.e. one viewed from above and the other viewed from anterolaterally
- Place the plate vertically on a firm sheet of paper and looking from superiorly, draw an ink line along the inner aspect of the plate to outline the horizontal segment of the mandible up to the angle of the mandible
- Accurately draw the angles of the osteotomies as viewed from above, using a ruler to outline the segments; the osteotomies are drawn at 900 to the plate (Figures 41a, b, c)
- Avoid unnecessary osteotomies and bony segments of <1.5 cm
- Two to three fibula bone segments are generally required with a unilateral segmental mandibulectomy, 3-4 following hemimandibulectomy and up to 7 for bilateral mandibulectomy The lateral (horizontal ramus of mandible) and vertical (ascending ramus) limbs of the plate, as viewed from anterolaterally, are drawn on paper up to the medial aspect of the lateral horizontal bone segment. Keep the plate flat by marking on the edge of an instrument table. Mark the point of the mandibular angle and the osteotomy angle between the horizontal and vertical segments; mark the uppermost osteostomy
- There are thus two sets of paper segments; one viewed from above and the other viewed from anterolaterally
- The segment medial to the angle will have a 900 angle to the plate at the mandibular angle as viewed from above, but approximately a 600 angle on the anterolateral view
- Mark the individual paper segments to identify and to orientate them. They may be redrawn on a firmer material such as sterilized X-ray plates

Figure 42: Paper templates
- By positioning the segments in a straight line, one can draw V-shaped templates to be used for V-shaped osteotomies, if preferred (Figure 42)
- While attached to the leg, the fibula is usually upside down with the pedicle on the wrong side for intraoral reconstruction. To avoid confusion, it is easier to place the fibula in the correct orientation as it would lie in the mouth by flipping it over in its longitudinal axis while still attached by its vascular pedicle
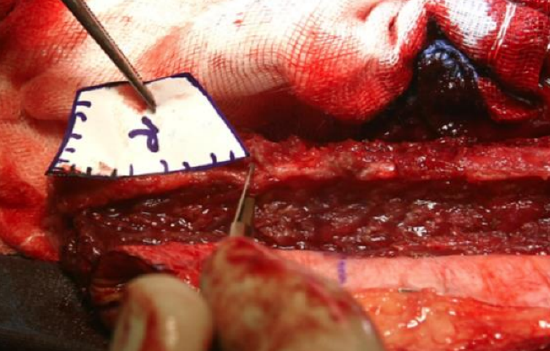
Figure 43: Transferring paper segment to the fibula
- Transfer the paper segments to the fibula to plan the osteotomies starting at the distal end of the fibula and using the superior-view templates first (Figure 43)
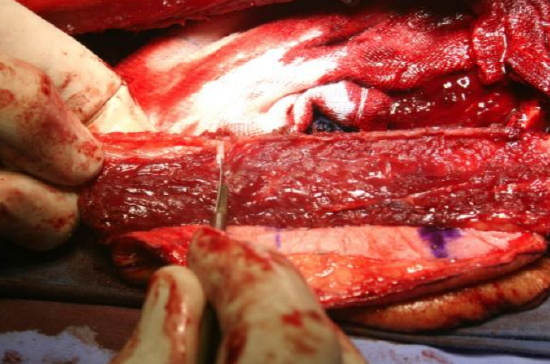
Figure 44: Incising periosteum on lateral surface of fibula for osteotomies
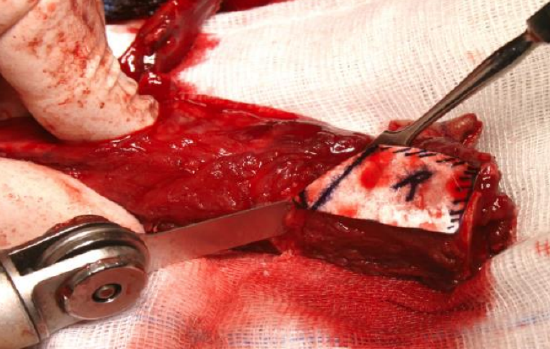
Figure 45: Osteotomy cuts
- Make scalpel cuts at precise angles on the lateral surface of the fibula, stripping only enough periosteum to make the saw cuts (Figures 44, 45)
- Hold the fibula with Kocher’s or bone-holding forceps and make the osteotomies

Figure 46: Attaching bone with flap still attached to leg
- The bony segments may be fixed to the plate and the mandible reconstruction virtually completed with the vascular pedicle still attached to the leg (Figure 46); alternatively, the final fixation of the bony segments to the plate is done after transposing the flap to the bony defect
- Only after the recipient vessels in the neck have been prepared, is the pedicle divided
- The individual bony segments are fixed to the plate with unicortical screws, while the plate is fixed to the mandible with bicortical screws. Use only one or two screws per segment to facilitate placing dental implants
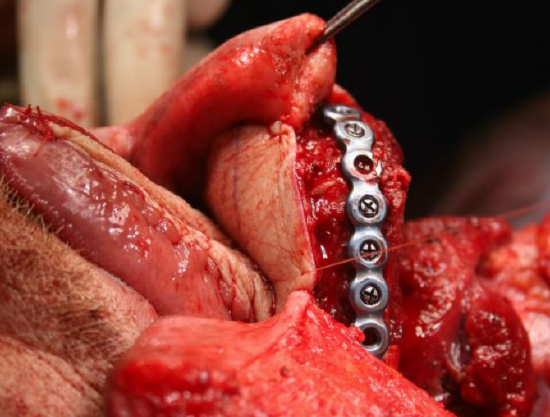
Figure 47: Flap fixed into place
- The flap is screwed into place, and at least part of the skin flap sutured to the mucosa or skin to stabilize the flap while doing the microvascular anastomosis (Figure 47)
Microvascular anastomosis
Readers are referred to the chapter Principles and technique of microvascular anastomosis for free tissue transfer flaps in head and neck reconstructive surgery for a detailed description of the surgical technique. Key points include the following:
- Standard microvascular anastomoses are performed with strict attention to technique
- Avoid kinking or rotating the vascular pedicle
- The artery is generally anastomosed end-to-end either to the facial or the superior thyroid artery. Other options are the external carotid artery or the transverse cervical artery
- One vein is anastomosed end-to-side to the internal jugular vein. Occasionally it may be anastomosed either end-to-side or end-to-end to a major tributary of the internal jugular vein or to the external jugular vein
- Beware of kinking of the vessels at the point where they depart from the bone as this lies deep to the bone and is not visible once bony fixation is completed
- A palpable pulsating artery does not necessarily imply that there is adequate flap perfusion. One should feel confident when seeing bleeding from the muscle or skin edges
Wound closure
- A Valsalva maneuver is done and the neck wound checked for bleeding and haemostasis achieved
- A suction drain is inserted into the neck, taking care for it not to cross over the microvascular repair or over the internal jugular vein (may thrombose the vein)
- Suture the tracheostomy to the skin; avoid ties around the neck as this may occlude the internal jugular vein and cause venous outflow obstruction and flap failure
- Repair the fibula donor site by suturing the lateral compartment muscles to the soleus muscle over a suction drain. Avoid suturing the wound under tension as this may cause skin necrosis, delayed wound healing and pseudocompartment syndrome. Skin grafting is done if required; if possible, cover exposed tendon or fascia with muscle before applying the skin graft
- Apply a posterior lower limb foot gutter splint for one week with the ankle at 900. Full weight bearing is commenced after one to two weeks
- The lower limb should be elevated as much as possible during the first 6 weeks to avoid swelling and edema.
- Physiotherapy to the lower limbs is commenced as early possible or as soon as the skin graft will permit
Examples of FFF reconstructions
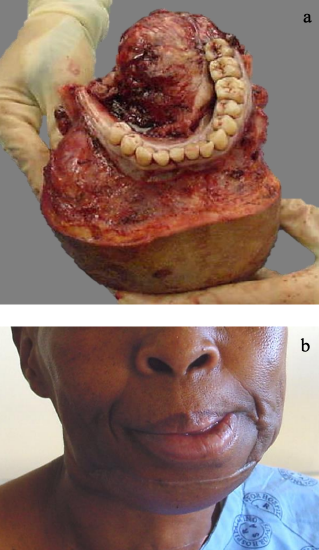
Figures 48a, b: Resection of mandible and FFF reconstruction
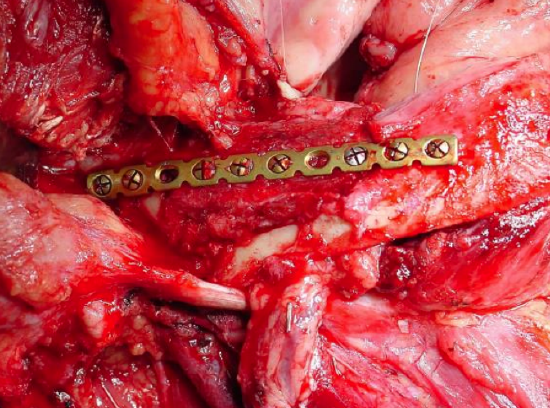
Figure 49: FFF onlay osseocutaneous flap following marginal mandibulectomy
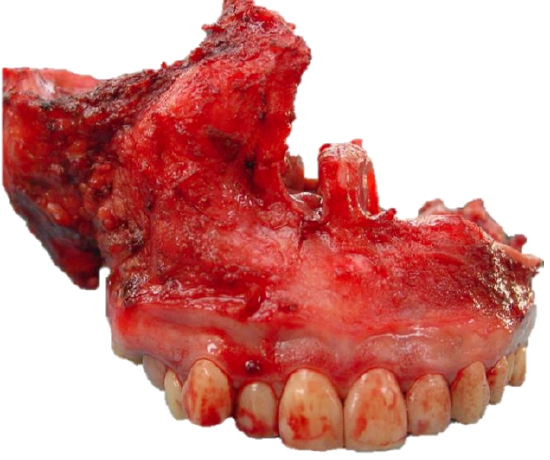
Figure 50a: Bilateral maxillectomy
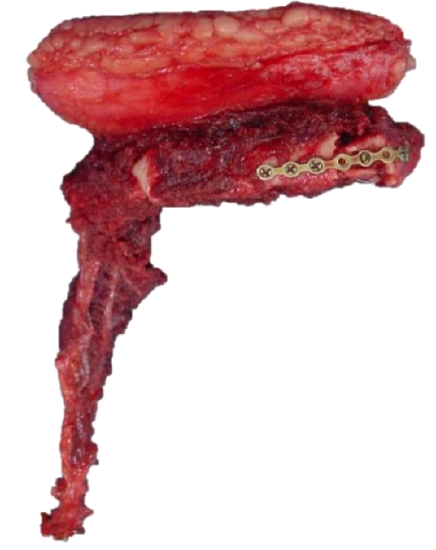
Figure 50b: FFF ready to be inset
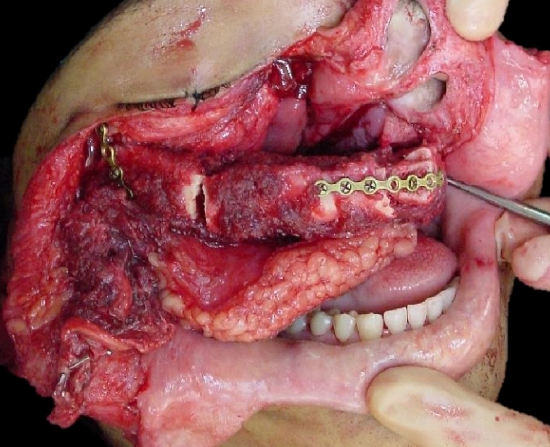
Figure 50c: Inset of FFF
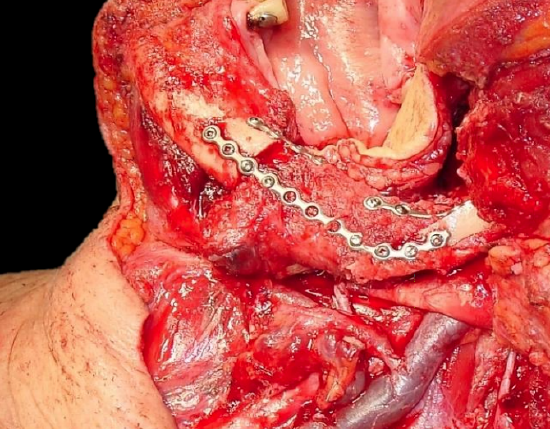
Figure 51: FFF reconstruction of lateral segmental mandibulectomy using miniplates
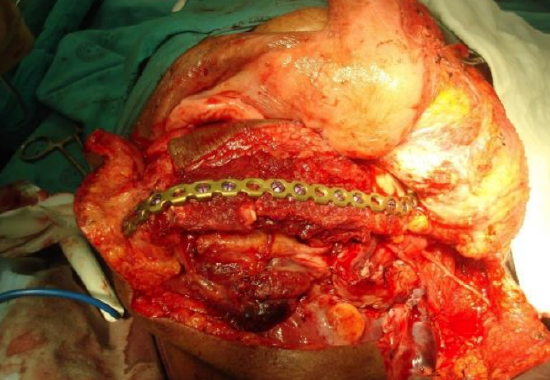
Figure 52: FFF reconstruction using a reconstruction plate following segmental mandibulectomy
Other flaps described in The Open Access Atlas of Otolaryngology Head & Neck Operative Surgery
- Pectoralis major flap
- Buccinator myomucosal flap
- Buccal fat pad flap
- Nasolabial flap
- Temporalis muscle flap
- Deltopectoral flap
- Paramedian forehead flap
- Upper and lower trapezius flaps
- Cervicofacial flaps
- Submental artery island flap
- Supraclavicular flap
- Latissimus dorsi flap
- Local flaps for facial reconstruction
- Radial free forearm flap
- Rectus abdominis flap
- Anterolateral free thigh flap
- Thoracodorsal artery scapular tip (TDAST) flap
- Principles and technique of microvascular anastomosis for free tissue transfer flaps in head and neck reconstructive surgery
Author
Ottie (JE) Van Zyl MBChB, FCS
Plastic & Reconstructive Surgeon
Groote Schuur Hospital
Cape Town, South Africa
ottie@mweb.co.za
Author and Editor
Johan Fagan MBChB, FCS(ORL), MMed
Professor and Chairman
Division of Otolaryngology
University of Cape Town
Cape Town, South Africa
johannes.fagan@uct.ac.za


